 Elements Compounds & Mixtures Worksheet
Elements Compounds & Mixtures Worksheet
Elements: •. A pure substance containing only one kind of atom. An element is always uniform all the way through (homogeneous). An element cannot nuclear
 Chemistry: Classifying Matter Name______________________
Chemistry: Classifying Matter Name______________________
If the material is a pure substance further classify it as either an element or compound in the right column. heterogeneous mixture? Give an example of each ...
 CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
MIXTURES. Classify the following as pure substances (element or compound) or mixtures. milk Mixture. 10. chicken noodle soup Mixture. 20. Soda Mixture. Page 2 ...
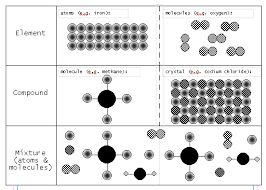
 The Nuts and Bolts of Elements Compounds and Mixtures
The Nuts and Bolts of Elements Compounds and Mixtures
Silica molecules always contain one silicon atom bonded to two oxygen atoms. In a molecule of table salt one sodium atom is bonded to one chlorine atom to
 ELEMENTS COMPOUNDS & MIXTURES
ELEMENTS COMPOUNDS & MIXTURES
An element is a simple pure substance which cannot be made simpler using chemical means. A pure substance is a form of matter that has the same composition
 Physical and Chemical Changes and Properties of Matter Worksheet
Physical and Chemical Changes and Properties of Matter Worksheet
When milk turns sour this is Classify each of the following substances as an element
 CHAP 2.pmd
CHAP 2.pmd
is a heterogeneous mixture for example
 Untitled
Untitled
Directions: Identify each of the following as an element compound
 K to 12 BASIC EDUCATION CURRICULUM SENIOR HIGH
K to 12 BASIC EDUCATION CURRICULUM SENIOR HIGH
elements and compounds. STEM_GC11S-Ie-. 28. 5. calculate the mass of a given reaction mixture when the following takes place: a. change in concentration of.
 Classification of Matter
Classification of Matter
Mixtures. Homogeneous. (Solutions). Heterogeneous. PURE. SUBSTANCE. COMPOUND. ELEMENT. Page 12. Pure Substances. An Element is a pure substances made of only
 Elements Compounds & Mixtures Worksheet
Elements Compounds & Mixtures Worksheet
Elements: • A pure substance containing only one kind of atom. An element is always uniform all the way through (homogeneous)
 iesc102.pdf
iesc102.pdf
of the matter around us exist as mixtures of is a heterogeneous mixture for example
 Elements Compounds
Elements Compounds
http://www.scasd.org/cms/lib5/PA01000006/Centricity/Domain/1441/WS2.1-ElCmp.pdf
 CHAP 2.pmd
CHAP 2.pmd
of the matter around us exist as mixtures of is a heterogeneous mixture for example
 Chemical Mixtures
Chemical Mixtures
entirely new substance called a compound. An alloy is a mixture of elements that has the characteristic of a metal. At least one of.
 CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
12. full fat milk heterogeneous MIXTURES. Classify the following as pure substances (element or compound) or ... 13. chocolate chip ice cream Mixture.
 Whats the Difference? - Atom Element
Whats the Difference? - Atom Element
Compound
 The Nuts and Bolts of Elements Compounds and Mixtures
The Nuts and Bolts of Elements Compounds and Mixtures
Silica molecules always contain one silicon atom bonded to two oxygen atoms. In a molecule of table salt one sodium atom is bonded to one chlorine atom to
 Chapter 15 Elements Compounds
Chapter 15 Elements Compounds
https://www.duxbury.k12.ma.us/cms/lib2/MA01001583/Centricity/Domain/462/Chapter%2015%20Elements%20and%20Compounds.pdf
 Untitled
Untitled
When milk turns sour this is a physical change because a change in odor does compound. 10. Oxygen (0?) element. 13. Raisin Bran heterogeneous mixture.
 [PDF] Elements Compounds & Mixtures Worksheet
[PDF] Elements Compounds & Mixtures Worksheet
M Milk M_Gasoline E_Iron (Fe) X Electricity E Uranium (U) element (E) a compound (C) a Heterogeneous Mixture (HM) or a Solution (S)
 [PDF] ELEMENTS COMPOUNDS & MIXTURES
[PDF] ELEMENTS COMPOUNDS & MIXTURES
A compound is a pure substance that consists of atoms of two or more elements joined together Compounds are formed when atoms of different elements react
 [PDF] Chemical Mixtures
[PDF] Chemical Mixtures
Milk is a mixture of liquid butterfat globules dispersed and suspended in water Colloids are generally considered heterogeneous mixtures but have some
 [PDF] MIXTURES ELEMENTS AND COMPOUNDS - Junta de Andalucia
[PDF] MIXTURES ELEMENTS AND COMPOUNDS - Junta de Andalucia
Mixtures contain two or more substances that are not chemically joined They can be made from both elements and compounds Be-
 [PDF] The Nuts and Bolts of Elements Compounds and Mixtures
[PDF] The Nuts and Bolts of Elements Compounds and Mixtures
Milk and mayonnaise are homogeneous mixtures Mixtures may also be heterogeneous – different parts of the mixture may look and act differently Asphalt and
 [PDF] Elements Compounds & Mixtures Notes & Practice
[PDF] Elements Compounds & Mixtures Notes & Practice
%2520compounds
 [PDF] Elements Compounds Substances and Mixtures © Van Der Sluys
[PDF] Elements Compounds Substances and Mixtures © Van Der Sluys
Indicate if the following common materials is a pure substance or a mixture 1 Milk is a 2 White sugar is 3 Brown sugar is 4 Tap water is
 [PDF] Classification of Matter
[PDF] Classification of Matter
Mixtures or Pure Substances Element Compound and give example Heterogeneous Mixture Milk toothpaste and mayonnaise are homogeneous mixtures
 [PDF] iesc102pdf - NCERT
[PDF] iesc102pdf - NCERT
actually a mixture of water fat proteins etc How do we judge whether milk ghee butter Pure substances can be elements or compounds
Is milk an element or compound Why?
Thus, milk is not a pure substance or a compound that is made up of one type of atom or molecule. But milk is a mixture of fats, proteins, sugar and water which are mixed irrationally. Thus, milk is a mixture. So, the given statement 'milk is a compound' is false.Why is milk a element?
Milk is not an element that is listed on the periodic table. Milk is not a single compound, but a mixture of compounds. The main compounds in milk include: water, lactose, fat, protein, and minerals. Milk can be purchased in different compositions, as well, like skim, whole, 2%, etc.Is milk a milk a mixture?
The main compounds of milk are lactose and casein and it is also called a colloidal mixture (i.e. in which one substance of microscopically dispersed insoluble or soluble particles is suspended throughout another substance). Therefore milk is a mixture, not a pure substance.- No, milk is not a compound or element.