 Formulae For CHEMICAL KINETICS
Formulae For CHEMICAL KINETICS
It can be experimentally determined. XII Chemistry. CHAPTER 4 - CHEMICAL KINETICS. If rate law expression for a reaction is. Rate = k
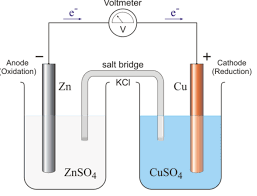 ELECTROCHEMISTRY ENGINEERING CHEMISTRY B.Tech 1 year
ELECTROCHEMISTRY ENGINEERING CHEMISTRY B.Tech 1 year
02-Apr-2020 All the plates are separated from adjacent plates by insulators like wood strips glass fiber etc. The entire combination is immersed in ...
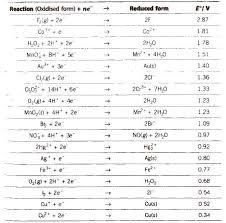 Chemistry Formula Electrochemistry
Chemistry Formula Electrochemistry
Chemistry Notes for class 12 Chapter 3. Electrochemistry. Electrochemistry is The conductivity of all the ions produced when 1 mole of an electrolyte is ...
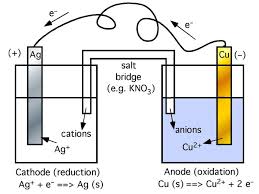 Chapter 18: Electrochemistry
Chapter 18: Electrochemistry
all other half-cells. Page 9. Reference half-cell : standard ... e.g. A constant current is passed through an electrolytic cell containing molten MgCl2 for 12 h.
 lech103.pdf
lech103.pdf
Species IO- is called as an intermediate since it is formed during the course of the reaction but not in the overall balanced equation. The first step
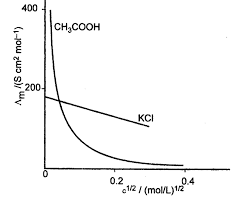 Revision Notes Class 12 Chemistry Chapter 3 – Electrochemistry
Revision Notes Class 12 Chemistry Chapter 3 – Electrochemistry
For this equation we take oxidation potential of anode and reduction potential of cathode. Since anode is put on left and cathode on right it follows therefore
 Electrochemistr ochemistr ochemistry Electrochemistr ochemistr
Electrochemistr ochemistr ochemistry Electrochemistr ochemistr
Sodium and magnesium metals are produced by the electrolysis of their fused chlorides and aluminium is produced (Class XII Unit 6) by all the materials that ...
 Unacademy
Unacademy
All redox reactions are exothermic. Why? Concept Ladder. According to classical concept oxidation is an addition of oxygen [or.
 Electrochemistry Formula Sheet
Electrochemistry Formula Sheet
Page 12. Gradeup CSIR-NET. Super Subscription. Features: 1. Memory Based Test All the CSIR NET Test Series based on the latest pattern and the trend that ...
 Formulae For ELECTROCHEMISTRY
Formulae For ELECTROCHEMISTRY
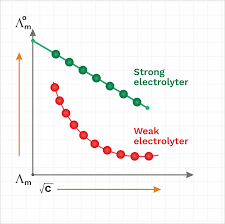
12. κ. Λ m x1000. = C. Remember: Unit of Λm in above formula is Scm2mol-1. 13. α c m. 0 m. ∧. = ∧. 14. 2 a cα. K = 1-α. XII Chemistry. CHAPTER 3 -
 Formulae For ELECTROCHEMISTRY
Formulae For ELECTROCHEMISTRY
Unit of ?m in above formula is Scm2mol-1. 13. ? c m. 0 m. ?. = ?. 14. 2 a c?. K = 1-?. XII Chemistry. CHAPTER 3 - ELECTROCHEMISTRY.
 Electrochemistry Formula Sheet
Electrochemistry Formula Sheet
Gradeup CSIR-NET. Super Subscription. Features: 1. Memory Based Test Series of the actual exam paper. 2. All the CSIR NET Test Series based on the latest
 Electrochemistry Electrochemistry
Electrochemistry Electrochemistry
In Class XI Unit 8
 Class -XII Chemistry Chapter-3 Electrochemistry Solved Examples
Class -XII Chemistry Chapter-3 Electrochemistry Solved Examples
The standard electrode potential of zinc ions is 0.76V. What will be the potential of a. 2M solution at 300K? Solution: The Nernst equation for the given
 Electrochemistr ochemistr ochemistry Electrochemistr ochemistr
Electrochemistr ochemistr ochemistry Electrochemistr ochemistr
65 Electrochemistry. As mentioned earlier (Class XI Unit 8) a galvanic cell is an electrochemical cell that converts the chemical energy of a spontaneous.
 Chemistry Notes for class 12 Chapter 3 Electrochemistry .pdf
Chemistry Notes for class 12 Chapter 3 Electrochemistry .pdf
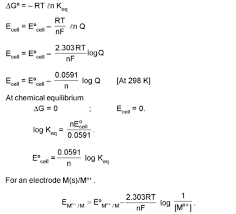
Nernst equation and Kc. At equilibrium. Page 9. 9
 Formulae For CHEMICAL KINETICS
Formulae For CHEMICAL KINETICS
chemical equation. It can be experimentally determined. XII Chemistry. CHAPTER 4 - CHEMICAL KINETICS. If rate law expression for a reaction is.
 Test4 ch19 Electrochemistry Practice-answers-Marked
Test4 ch19 Electrochemistry Practice-answers-Marked
p12. Predictable Oxidation and Reduction Strength Patterns Key Equations Given for Test: ... Determine all oxidation numbers and see which change!
 (968)-chemistry-gyan-sutra-jee-main.pdf
(968)-chemistry-gyan-sutra-jee-main.pdf
12. 6. Ionic Equilibrium. 15. 7. Electrochemistry Formula : Oxidation Number = number of electrons in the valence shell.
 Notes - 06 Electrochemistry - CIE Chemistry A Level
Notes - 06 Electrochemistry - CIE Chemistry A Level
6 : Electrochemistry In an ion the sum of the oxidation states of all the atoms is ?equal to the ... Therefore the balanced equation can be written as:.