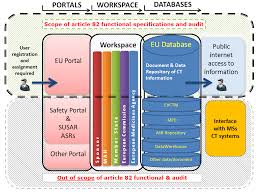
 Functional specifications for the EU portal and EU database to be
Functional specifications for the EU portal and EU database to be
25 мар. 2015 г. Legal requirements of the EU portal and EU database ... The functional specifications have to address the following systems requirements:.
 Guideline on good pharmacovigilance practices (GVP) Module I
Guideline on good pharmacovigilance practices (GVP) Module I
22 июн. 2012 г. ... database (or other systems to collate adverse reaction ... should ensure that the tasks are subject to a quality system compliant with the legal ...
 Guideline on veterinary good pharmacovigilance practices (VGVP
Guideline on veterinary good pharmacovigilance practices (VGVP
18 нояб. 2021 г. Audits of the pharmacovigilance system should ensure that it complies with the legal requirements the human ... systems or regulatory databases ...
 Guideline on good pharmacovigilance practices (GVP) - Module II
Guideline on good pharmacovigilance practices (GVP) - Module II
28 мар. 2017 г. The legal requirement for marketing authorisation holders to maintain and make available upon request a pharmacovigilance system master file ( ...
 The RIPE Database Requirements Task Force - Draft
The RIPE Database Requirements Task Force - Draft
“A core requirement of the Internet Numbers Registry System is to maintain a registry of conditions and legal requirements of the RIPE Database. The ...
 Requirements for the RIPE Database
Requirements for the RIPE Database
“A core requirement of the Internet Numbers Registry System is to maintain a registry of allocations legal requirements of the. RIPE Database. The requirement ...
 Functional specifications for the European Database on Medical
Functional specifications for the European Database on Medical
15 дек. 2022 г. • Point 8 covers the functional specification derived from the legal requirements for the EUDAMED Information system. The functional ...
 Guideline on computerised systems and electronic data in clinical
Guideline on computerised systems and electronic data in clinical
9 мар. 2023 г. system or database(s). In case of manual ... intended use is covered by local legal requirements and the in-house rules of the institution.
 Law Enforcement Database Systems
Law Enforcement Database Systems
Disposal methods must meet the requirements stated in PPP (Policies Practices and Procedures). Section 1.9.11. h. Information received from a CLETS terminal
 A NEW PERSPECTIVE IN DEVELOPING COMPREHENSIVE AND
A NEW PERSPECTIVE IN DEVELOPING COMPREHENSIVE AND
LEGAL DATABASE SYSTEM TO SUPPORT THE EASE OF DOING BUSINESS. By: Tyas Dian official Rules of Procedure of the. European Council; how the European. Council ...
 Guideline on good pharmacovigilance practices (GVP) - Module II
Guideline on good pharmacovigilance practices (GVP) - Module II
28 mar. 2017 PSMF section on computerised systems and databases . ... The legal requirement for marketing authorisation holders to maintain and make ...
 Functional specifications for the EU portal and EU database to be
Functional specifications for the EU portal and EU database to be
25 mar. 2015 Draft prepared with the clinical trials information system expert ... Legal requirements of the EU portal and EU database .
 Guideline on good pharmacovigilance practices (GVP) Module I
Guideline on good pharmacovigilance practices (GVP) Module I
22 jun. 2012 The legal requirement for quality systems was introduced by ... and to databases to authorised personnel respecting the medical and ...
 Requirements for a single database of beneficiaries
Requirements for a single database of beneficiaries
Ideally the legal entities obtaining EU funds would be linked to BRIS in their presentation on the reporting system's website
 Final report
Final report
20 dic. 2021 on draft regulatory technical standards under Article 9a (1) and (3) of. Regulation (EU) No 1093/2010 setting up an AML/CFT central database ...
 Guideline on veterinary good pharmacovigilance practices (VGVP
Guideline on veterinary good pharmacovigilance practices (VGVP
18 nov. 2021 complying with the legal requirements for pharmacovigilance tasks and responsibilities. The pharmacovigilance system of the marketing ...
 Database systems Level 2 Unit 20
Database systems Level 2 Unit 20
systems. P1 explain the principles of database systems. M1 explain the legal requirements for a database system. 2 Be able to create non-.
 Interim Report prepared by Milieu Ltd for the European Parliament
Interim Report prepared by Milieu Ltd for the European Parliament
Summary of legal requirements applying to EHRs The move towards centralised databases of electronic health records marks a fundamental shift in the.
 Draft guideline on computerised systems and electronic data in
Draft guideline on computerised systems and electronic data in
10 jun. 2021 Legal and regulatory background . ... Training requirements for computerised systems . ... Database decommissioning .
 A Practical Guide to Database Compliance
A Practical Guide to Database Compliance
Many regulatory compliance requirements focus on privileged insiders as well with special database system beyond his/her level of authorization.