 Unacademy
Unacademy
Those thermodynamic properties which depend on initial and final state. Eg: Pressure volume
 LECTURE NOTES ON THERMODYNAMICS
LECTURE NOTES ON THERMODYNAMICS
31-Oct-2023 These are lecture notes for AME 20231 Thermodynamics
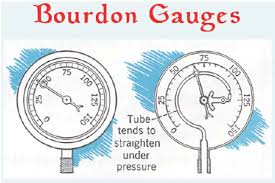 Basic Concepts of Thermodynamics Thermodynamics and Energy
Basic Concepts of Thermodynamics Thermodynamics and Energy
Thermodynamics can be defined as the study of energy energy transformations and its relation to matter. The analysis of thermal systems is achieved through the
 Lecture Notes on Thermodynamics
Lecture Notes on Thermodynamics
13-Feb-2019 Lecture Notes on Thermodynamics. Éric Brunet1 Thierry Hocquet2 ... These quantities are called thermodynamic variables. Frame 1.4 : Thermodynamic ...
 Thermodynamics Digital Notes.pdf
Thermodynamics Digital Notes.pdf
Limitations of the First Law - Thermal Reservoir Heat Engine
 THERMODYNAMICS
THERMODYNAMICS
We are interested in chemical reactions and the energy changes accompanying them. For this we need to know certain thermodynamic terms. These are discussed
 DIGITAL NOTES
DIGITAL NOTES
Zeroth Law of Thermodynamics – Principles of Thermometry –Constant. Volume gas Thermometer – Scales of Temperature – PMM I - Joule's Experiment – First law of
 NEET Revision Notes Chemistry Thermodynamics
NEET Revision Notes Chemistry Thermodynamics
For instance heat
 Hand Written Notes of Engineering Thermodynamics (PCME4201)
Hand Written Notes of Engineering Thermodynamics (PCME4201)
Hand Written Notes of Engineering Thermodynamics. (PCME4201). Dr Anand Gupta. Assistant Professor. Department of Mechanical Engineering. IGIT Sarang. Page 2
 THERMAL PHYSICS_(THERMODYNAMICS)_NOTES.indd
THERMAL PHYSICS_(THERMODYNAMICS)_NOTES.indd
d(PV) d(nRT). = PdV VdP 0. +. = VdP. PdV. = -. dP. P. dV. V. = -. 2. 10. 1. V. W 2.303nRTlog. V. ⎛. ⎞. ∆. = │. │. │. │. ⎝. ⎠. 1. 10. 2. P.
 Lecture Notes on Thermodynamics
Lecture Notes on Thermodynamics
13-Feb-2019 Thermodynamics is a branch of physics that studies macroscopic ... These lecture notes are intended for students who already have some ...
 LECTURE NOTES ON THERMODYNAMICS
LECTURE NOTES ON THERMODYNAMICS
14-Feb-2010 These are lecture notes for AME 20231 Thermodynamics
 Basic Concepts of Thermodynamics Thermodynamics and Energy
Basic Concepts of Thermodynamics Thermodynamics and Energy
Thermodynamics and Energy. Thermodynamics can be defined as the study of energy energy transformations and its relation to matter. The analysis of thermal
 DIGITAL NOTES
DIGITAL NOTES
DIGITAL NOTES. THERMODYNAMICS. (R18A0303). B.Tech II Year I Semester. DEPARTMENT OF MECHANICAL ENGINEERING. MALLA REDDY COLLEGE OF ENGINEERING &. TECHNOLOGY.
 Notes on Thermodynamics The topic for the last part of our physics
Notes on Thermodynamics The topic for the last part of our physics
This equation is called the first law of thermodynamics. It is a statement recognizing the existence of internal energy and its conservation. Some Heat
 LECTURE NOTES THERMODYNAMICS
LECTURE NOTES THERMODYNAMICS
Two copper blocks) in contact attain thermal equilibrium when the heat transfer between them stops. The equality of temperature is the only requirement for
 Lecture Notes on Thermodynamics and Statistical Mechanics (A
Lecture Notes on Thermodynamics and Statistical Mechanics (A
10-Dec-2019 Lecture Notes on Thermodynamics and Statistical Mechanics ... at http://astarte.csustan.edu/$sim$tom/SFI-CSSS/info-theory/info-lec.pdf.
 The Second Law of Thermodynamics
The Second Law of Thermodynamics
The second law of thermodynamics asserts that processes occur in a certain direction and that the energy has quality as well as quantity.
 1st Year Thermodynamic Lectures Dr Mark R. Wormald
1st Year Thermodynamic Lectures Dr Mark R. Wormald
Basic chemical thermodynamics :- Lecture 1. Introduction. Basic Definitions. 1st Law of thermodynamics. Lecture 2. 2nd Law and entropy. Free energy and
 Thermodynamics
Thermodynamics
Thermodynamics the study of the transformations of energy from one form into another. First Law: Heat and Work are both forms of Energy. in any process.
 Lecture 5: Thermodynamics - Scholars at Harvard
Lecture 5: Thermodynamics - Scholars at Harvard
Lecture5: Thermodynamics 1 Introduction Thermodynamicsis thestudyof heat andtemperature Onethingthat makes thermodynamicshard(andgenerallyunpopular) is all the damnvariables Everything is relatedandit's oftentoughtokeepstraight whatisanindependentandwhat is adependent variable
 Lecture Notes on Thermodynamics & Statistical Mechanics
Lecture Notes on Thermodynamics & Statistical Mechanics
Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects Through-out these lectures we will talk a lot about laws and models Models are a simpli?ed empirical description of a real system which generally develops overtime as our knowledge progresses
 Thermodynamics: The Three Laws of Thermodynamics - Equations
Thermodynamics: The Three Laws of Thermodynamics - Equations
We can apply thermodynamics to stable systems unstable systems or to each stage of a reaction mechanism Thermodynamics cannot give information on reaction rates directly (although the amount of energy required is going to be important in determining reaction rates - see Transition State Theory) FEATURES 1
 8044 Lecture Notes Chapter 3: Thermodynamics rst pass
8044 Lecture Notes Chapter 3: Thermodynamics rst pass
8 044 Lecture Notes Chapter 3: Thermodynamics rst pass Lecturer: McGreevy Reading: CJ Adkins Equilibrium Thermodynamics Chapters 1 2 3 1-3 7 Skip Adkins x3 5 4 R Baierlein Thermal Physics Chapter 1 The four laws of thermodynamics [according to Prof Yosi Avron]: 0th Law: You have to play the game 1st Law: You can’t win
 CHAPTER 2 LECTURE NOTES The First Law of Thermodynamics
CHAPTER 2 LECTURE NOTES The First Law of Thermodynamics
thermodynamic property that depends only on state of the system and noton how it got there is called a state function The internal energy U is a statefunction Therefore a change in the internal energy ?U can be calculated simplyby knowing the initial and final values of U: ?U = Uf Ui On the other hand q and w are path-dependent functions
 Searches related to thermodynamics notes pdf PDF
Searches related to thermodynamics notes pdf PDF
1 1 PRELIMINARY CONCEPTS – THE LANGUAGE OF THERMODYNAMICS In order to accurately and precisely discuss various aspects of thermodynamics it is essential to have a well-defined vernacular As such a list of some foundational concepts and their definitions are shown below: Universe – all measured space System – space of interest
What are the first three laws of thermodynamics?
There three laws are: The first law of thermodynamics is the law of the conservation of energy; it states that energy cannot be created nor destroyed. An example is when the chlorophyll absorbs light and transforms it into chemical energy.
What are the fundamentals of thermodynamics?
Fundamentals of Thermodynamics Thermodynamics is defined as the branch of science which deals with the study of different forms of energy along with their conversion. It is indeed an important branch of science because it provides an explanation of the macroscopic properties of matter in terms of the microscopic concept of matter.
What is the formula for thermodynamics?
Thermodynamics - Equations. ?Hrxn = ?H1 + ?H2 + ?H3 + ... k is the Boltzmann Constant with a value of 1.38 × 10 ?23 J/K which is the same as the universal gas constant, R, divided by Avogadro's number, NA . Note: S° is standard absolute entropy and is value you find in a thermodynamic table.