|
Lech103pdf
Chemistry by its very nature is concerned with change Substances with well defined properties are converted by chemical reactions into other substances |
|
CBSE NCERT Solutions for Class 12 Chemistry Chapter 3
Class- XII-CBSE-Chemistry Electrochemistry Practice more on Electrochemistry Page - 1 www embibe com CBSE NCERT Solutions for Class 12 Chemistry Chapter 3 |
Electrochemistry is the study of production of electricity from energy released during spontaneous chemical reactions and the use of electrical energy to bring about non-spontaneous chemical transformations.
The subject is of importance both for theoretical and practical considerations.
What is class 12 chemistry ch 3?
Electrochemistry is a fine combination of, or the relationship between electrical and chemical reactions.
The class 12 Chemistry chapter 3 notes cover a brief outline of electrochemical cells, Nernst equation, Gibbs energy of cell reaction, conductivity, Kohlrausch law and its applications, electrolysis, etc.
|
Electrochemistr ochemistr ochemistry Electrochemistr ochemistr
65 Electrochemistry. As mentioned earlier (Class XI Unit 8) a galvanic cell is an electrochemical cell that converts the chemical energy of a spontaneous. |
|
Electrochemistry Electrochemistry
reactions that do not proceed spontaneously. 3. Electrochemistry. Unit. 3 their fused chlorides and aluminium is produced (Class XII Unit 6) by. |
|
Unit 3.pmd
Exemplar Problems Chemistry. 3. Which of the following statement is correct? (i). E. Cell and Ar. G of cell reaction both are extensive properties. |
|
CHEMISTRY (043) Class XI
b) Study the shift in equilibrium between [Co(H2O)6]2+ and chloride ions by changing the concentration of either of theions. Page 3. CLASS -XII. S No. Unit. |
|
CHEMISTRY (CLASSES XI –XII)
3. CLASS XI (THEORY). (Total Periods 180). Unit I: Some Basic Concepts of Chemistry Unit XII: Organic Chemistry – Some Basic Principles and Techniques. |
|
CLASSIFICATION OF ELEMENTS AND PERIODICITY IN
study. UNIT 3. After studying this Unit you will be able to. • appreciate how the concept of grouping elements in accordance to their properties led to the. |
|
Chemistry Notes for class 12 Chapter 3 Electrochemistry .pdf
www.ncerthelp.com (Visit for all ncert solutions in text and videos CBSE syllabus |
|
NCERT Solutions for Class 12 Chemistry Chapter 3 - Electrochemistry
NCERT Solutions for Class 12 Chemistry Chapter 3. Electrochemistry. Q 3.1: Arrange the following metals in the order in which they displace each other from |
|
CBSE NCERT Solutions for Class 12 chemistry Chapter 3
Using the standard electrode potentials predict if the reaction between the following is feasible: Ags and Fe3+aq. NCERT Chemistry (Vol 1). Chapter 3 |
|
Class: XII Subject: CHEMISTRY TERM-1 NO. OF PERIOD TOPIC
Class: XII. Subject: CHEMISTRY EX-3:1. Learners will be able to understand the diffrences ... UNIT-12. ALDEHYDES KETONES preperation of carboxylic. |
|
Electrochemistry - NCERT
As mentioned earlier (Class XI, Unit 8) a galvanic cell is an electrochemical cell that converts the chemical energy of a spontaneous redox reaction into electrical |
|
Chemistry Notes for class 12 Chapter 3 Electrochemistry - Ncert Help
1 Page www ncerthelp com (Visit for all ncert solutions in text and videos, CBSE syllabus, note and many more) Chemistry Notes for class 12 Chapter 3 |
|
CHEMISTRY CLASS-XII - edudel
10 Unit II Solutions 10 Unit III Electrochemistry 12 23 Unit IV Chemical Kinetics 10 Unit V Students of class XII Chemistry wanted to prepare magnets |
|
Chemistry Class 12 Chapter 3 NCERT Solution - Mywayteaching
www mywayteaching com Class XII Chapter 3 – Electrochemistry Chemistry Question 3 1: Arrange the following metals in the order in which they displace |
|
Formulae For ELECTROCHEMISTRY - WordPresscom
3 Ecell = Eө cell - 2 303RT [Anode ion] log nF [Cathode ion] 4 Eө cell = c 0 059 logK 12 κ Λ m x1000 = C Remember: Unit of Λm in above formula is Scm2mol-1 13 α c m 0 m ∧ = ∧ 14 2 a cα K = 1-α XII Chemistry CHAPTER 3 - |
|
Class 12 Chemistry Ncert Intext Questions Electrochemistry
Class 12 Chemistry Chemical Kinetics NCERT Solutions for Class 12 Chemistry Chapter 3 NCERT Solutions For Class 12 Chemistry Chapter 3 NCERT |
|
Class 12 Chemistry Ncert Intext Questions Electrochemistry
NCERT Textbook Chapter 3 Electrochemistry Chemistry Class 12 Chemistry Chapter 3 Electrochemistry NCERT electrochemistry class 12 intext Archives |
|
Class 12 Chemistry Ncert Intext Questions ElectrochemistryConcept
26 déc 2020 · chemistry notes for class 12 chapter 3 electrochemistry electrochemistry is that branch of chemistry which deals with the study of production of electricity from |
|
Chemistry Project On Electrochemistry Class 12
part 1 NCERT unit 3 explained Electrochemistry Class 12 CBSE Notes Pdf Download Journal Project Report Electrochemical Cell Chemistry Project Chemistry |
|
[PDF] Electrochemistry - ncert
65 Electrochemistry As mentioned earlier (Class XI, Unit 8) a galvanic cell is an electrochemical cell that converts the chemical energy of a spontaneous |
|
[PDF] Chemistry Notes for class 12 Chapter 3 Electrochemistry
Standard electrode potential The potential difference developed between metal electrode and solution of ions of unit molarity (1M) at 1 atm pressure and 25°C ( |
|
[PDF] chemistry class-xii - edudel
Unit Page No 1 e Solid State 7 18 2 Solutions 19 33 3 Electrochemistry Ira Singh, a student of class XII Chemistry stated that normal saline solution |
|
[PDF] Grade 12 Chemistry - Manitoba Education
The Senior Years Student and the Science Learning Environment 3 Section 3 Assessment in Grade 12 Chemistry 1 Document Production Services Unit |
|
[PDF] Chemistry 12 - Notes on Unit 3 - Solubility - BC Learning Network
Chemistry 12 Notes on Unit 3 Solubility Chemistry 12 Unit 3 Solubility of Ionic Substances Page 3 CH3COOH(l) H+ rest of the Chemistry 11 class |
|
[Book] Electrochemistry Std 12th - incartw
Jan 30, 2020 · CBSE Class 12 Chemistry Electrochemistry Full Chapter By Shiksha Electrochemistry class 12 part 1 &#NCERT unit 3 explained in |
|
[PDF] 7 ELECTROCHEMISTRY
where, V is the volume of solution in cm3 containing 1 mole of electrolyte and C is the molar concentration Units 1 m 3 1000 S cm C mol |
|
[PDF] CHEMISTRY (Code No 043) 2019-20 - CBSE Academic
Total Periods (Theory 160+ Practical 60) Time 3 Hours Total Marks70 Unit No Title No of Periods Marks Unit I Some Basic Concepts of Chemistry 12 11 |
|
[PDF] Chemistry- Syallabus Class-XII-2020pmd
Chapters 3 Electrochemistry 12 Unit IV Chapters 4 Chemical Kinetics 10 Unit V CHEMISTRY CLASS XII (2020 21) Unit No Chapters Title Marks Total |
|
[PDF] Class 12 Chemistry Ncert Intext Questions Electrochemistry
Chemistry UNIT 3 ELECTROCHEMISTRY NCERT Chemistry Answers Class 12th Chapter 3 NCERT Solutions For Class 12 Chemistry Electrochemistry |
- electrochemistry class 12 pdf download
- grade 12 chemistry pdf
- grade 12 chemistry notes
- pradeep fundamental chemistry for class 12 pdf free download
- grade 12 chemistry lessons
- electrochemistry pdf notes for bsc
- electrochemistry class 12 notes ppt
- electrochemistry problems and solutions pdf
- class 12 chemistry unit 2
- class 12 chemistry unit 1 notes
- class 12 chemistry unit 6
- class 12 chemistry unit 2 notes
- class 12 chemistry unit 16
- class 12 chemistry unit 10
- class 12 chemistry unit 3 notes
- class 12 chemistry unit 1

10th Class Chemistry Notes All Chapters pdf - Rattapk
Source:https://sedinfo.net/wp-content/uploads/2018/05/9th-Chemistry-book-contents-page.jpg

Download 9th Class Chemistry Book
Source:https://www.vedantu.com/content-images/ncert-solutions/ncert-solutions-class-11-chemistry-chapter-2-structure-of-atom/1.png
NCERT Solutions for Class 11 Chemistry Chapter 2 Structure of Atom
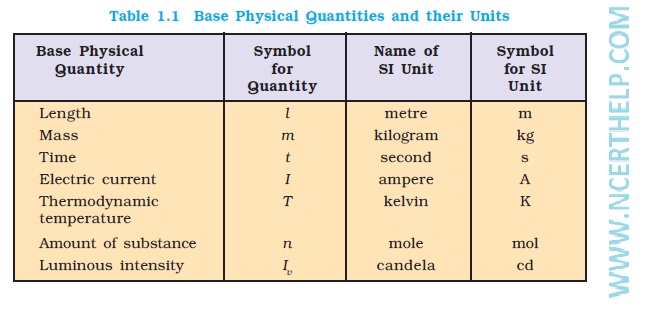
Source:https://ncerthelp.com/ncertimages/class%2011/chemistry/CH01/Physical%20Quantities%20and%20their%20Units.jpg

chemistry notes for class 11 Chapter 1 SOME BASIC CONCEPTS
Source:https://1.bp.blogspot.com/-sJ6F34MI1gE/XWlOP4bITGI/AAAAAAAANTo/0h-Nskv8BLUD_dTEaqSrYhpl3qABvKElwCLcBGAs/s16000/9th%2Bclass%2Bchemistry%2Bmcqs%2Bpdf.jpg

9th class chemistry MCQs PDF English Medium - Zahid Notes
Source:https://3.bp.blogspot.com/-kWPeYPDcgyw/WdmEjFscELI/AAAAAAAAG6U/KvlvnIwDk0ML_szFGtwWDYEq2DDjKQM2wCLcBGAs/s1600/Chemisrty%2BCh%2B01-2.jpg

10th Class Chemistry Chapter 01: Chemical Equilibrium Notes and
Source:http://latestcontents.com/wp-content/uploads/2014/06/130.jpg
class 12 chemistry unit 3 electrochemistry
[PDF] TOPIC 6: ELECTROCHEMISTRY - Manitoba Education
- electrochemistry pdf class 12
- introduction to electrochemistry pdf
- electrochemistry problems and solutions pdf
- advanced electrochemistry pdf
- electrochemistry ppt
- electrochemistry pdf for engineering
- electrochemistry questions and answers pdf
- electrochemistry mcq pdf download
- class 12 chemistry unit 2
- class 12 chemistry unit 1 notes
- class 12 chemistry unit 6
- class 12 chemistry unit 2 notes
- class 12 chemistry unit 16
- class 12 chemistry unit 10
- class 12 chemistry unit 3 notes
- class 12 chemistry unit 1
class 12 english book chapter 1
[PDF] Bbc English Class 12 Solutions - Neat Neat Neat Records
- successcds english class 12
- the last lesson characters
- short story the last lesson
- the last lesson lesson plan
- the last lesson reference to context
- the last lesson long answer questions
- the last lesson about the author
- the last lesson summary study rankers
- class 12 english book pdf
- class 12 english book name
- class 12 english book ncert
- class 12 english book chapters
- class 12 english book up board
- class 12 english book flamingo
- class 12 english book chapter 1
- class 12 english book price
class 12 english book chapters
[PDF] Class: XII Subject: English NO OF PERIODS TOPICS LEARNING
- class 12 english book pdf
- class 12 english book name
- class 12 english book ncert
- class 12 english book flamingo
- class 12 english book up board
- class 12 english book chapters
- class 12 english book chapter 1
- class 12 english book price
class 12 english book flamingo pdf download
[PDF] [PDF] Ncert Solutions For Class 12 Chemistry - Browser Quest
- class 12 english book vistas pdf download
- ncert solutions for class 12 english flamingo pdf download
- class 12 english book pdf download
- class 12 english book pdf 2020
- flamingo - textbook in english (core course) for class - 12 - 12074
- class 12 english flamingo chapter 1 pdf
- english core book class 12 pdf
- flamingo book class 12 in. hindi
- class 12 english book pdf
- class 12 english book name
- class 12 english book ncert
- class 12 english book flamingo
- class 12 english book up board
- class 12 english book chapters
- class 12 english book chapter 1
- class 12 english book price