Can thermodynamic models predict hydrate stability?
Since that time, researchers have been developing models, such as CSMGem , to predict the necessary thermodynamic conditions for hydrate stability. Prediction of the hydrate stability conditions using such models is now possible within experimentally measurable accuracy.
What is hydrate research?
The Hydrates in Science community at the Center for Hydrate Research is continuing its strong tradition of investigating the fundamentals of hydrate phenomena with the goal of applying the results to practical aims such as hydrogen storage, kinetic inhibition, and gas recovery.
Can hydrate inhibitors be incorporated into a multi-phase Gibbs energy minimization program?
In part II of this series, we proposed an aqueous phase model tailored specifically for the presence of hydrate inhibitors such as salts and methanol in the aqueous phase. Part III provides a prescription for the incorporation of the new hydrate and aqueous phase models into a multi-phase Gibbs energy minimization program (CSMGem).
Is hydrate standard state a good model?
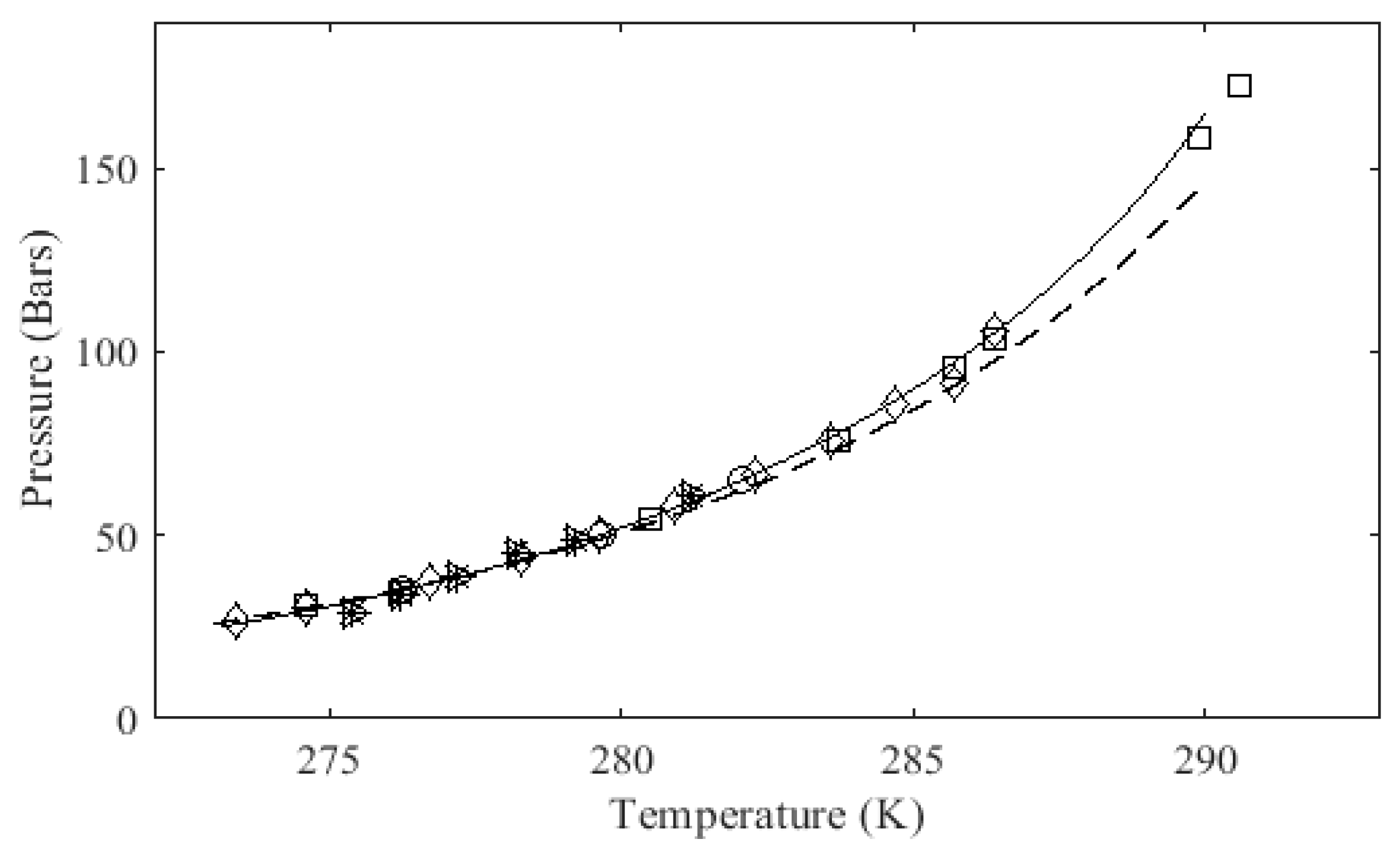
Predictions from this model are relatively good at moderate pressures and temperatures. However, at high pressures ( P >200 bar) and for natural gas hydrates the model predictions deviate from experimental data. This suggests that the hydrate standard state (empty lattice) properties are not well defined.
|
??????? ???????: ??????? ?????????????? ?????
A non-ideal hydrate solid solution model for a multi-phase equilibria program: Ph.D. Thesis Colorado: Colorado School of Mines |
|
CET Volume 86
Phase Equilibria Analysis in the Presence of Solid Carbon Ballard A.L. 2002 |
|
Methane Hydrate Formation in Ulleung Basin Under Conditions of
Ballard A.L.: A non-ideal hydrate solid solution model for a multi-phase equilibria program. Technical report |
|
CSH Solubility Modeling At Different Temperatures JA Gisby1 RH
Equilibria Software from the NPL) and measured solubility data such as pH and silicate hydrate gel |
|
THERMODYNAMICS OF GAS HYDRATE EQUILIBRIA
the ideal solid solution theory to the prediction of heat capacities of appropriate for the solution of the specified multiphase equilibrium problem is. |
|
Modelling Chemical Equilibrium Partitioning with the GEMS-PSI Code
The equilibrium partitioning in a multiphase system including for example an aqueous solution |
|
Phase Equilibria Analysis in the Presence of Solid Carbon Dioxide
Ballard A.L. 2002 |
|
Thermodynamic modeling of compressible hydrates and
2. Natural. Gas Hydrates. 3. Phase Equilibrium Algorithm. 4. Thermodynamic Modeling. 5. Solid Solution. Compressibility. I. Tavares Frederico W.(orient.). |
|
TITLE OF DISSERTATION GOES HERE IN ALL CAPS
DEVELOPMENT OF THERMODYNAMIC MODELS FOR PHASE EQUILIBRIA OF. WATER-ICE-GAS-HYDRATE IN AQUEOUS SOLUTIONS OF INHIBITORS AND. IN POROUS MEDIA. Doctoral Thesis. |
|
Simultaneous Multiphase Flash and Stability Analysis Calculations
Sep 1 2021 other types of phase equilibria involving solid CO2 in natural ... (37) Ballard |
|
Chapter 2 - Mountain Scholar
A NON-IDEAL HYDRATE SOLID SOLUTION EQUILIBRIA PROGRAM by 1 6 3 Implementation of Hydrate Model into a Multi-Phase Flash Program 14 |
|
PHASE EQUILIBRIA OF GAS HYDRATES - Mountain Scholar
hydrate phase equilibria for systems containing CO2 and H2S incorporated into the non-ideal hydrate solid solution Gibbs energy minimization multi-phase equilibria program, CSMGem, to better predict hydrate formation for hydrates CHAPTER 3 PHASE EQUILIBRIA DATA AND MODEL COMPARISONS FOR H2S |
|
Phase Equilibria Modelling of Petroleum Reservoir Fluids - CORE
state, has been employed to model the phase equilibria are modelled by the solid solution theory of van der Waals and Platteeuw Predictions of 5 5 Prediction of Hydrate Inhibitor Distribution in Multiphase results show that n- propanol and ethanol do not display the same level of hydrate highly non-ideal systems |
|
TREATMENT OF NON-IDEALITY IN THE MULTIPHASE MODEL
5 1 Multiphase chemistry approach considering non-ideal solutions 45 140 Appendix 152 A Thermodynamics of phase equilibria in multicomponent systems 152 However, gas phase mixtures don't show strong formation of a solid salt (or hydrate), when the solution becomes supersaturated as well as |
|
3 Review of Gas Hydrate Phase Equilibrium Models
Figure 5 6: Phase equilibrium of clathrate/semi-clathrate hydrates for the carbon dioxide vdW-P = van der Waals and Platteeuw solid solution theory WP = high when dealing with complex systems including multi-phases, which are not known in advance In other word, the Ideal model (Raoult‟s law) or ideal model + |
|
Thermodynamic modeling of compressible hydrates and - EPQB
The algorithm for multiphase equilibrium calculations proposed is based the hydrate phase is a crystalline solid composed of a water lattice and cages which the dynamic model for a clathrate solution and the assumptions made in the 4 species in each cage, R is the ideal gas constant and ν is the ratio between num- |
|
[PDF] Development of a multiphase flash in presence of hydrates
hydrates inside the hydrate phase boundary for a natural gas mented in CSMGEM software [2]) which is an adaptation of the original work by phase equilibrium solution, one may not need to perform stability analysis separate ly sets of initial K values, namely, (1) Composition Independent Ideal K values and (2) |
|
[PDF] Development of Predictive Techniques for Estimating Liquid Water
Nov 11, 2008 · Water Hydrate Equilibrium of Water Hydrocarbon System Amir H Mohammadi and The fugacity of water in the hydrate phase is calculated using the solid solution water being in equilibrium with gas hydrates show less than [15] A L Ballard, A non ideal hydrate solid solution model for a multi phase |
|
[PDF] Brazilian Journal of Chemical Engineering - SciELO
1 Multiphase Flow Research Center (NUEM), Post graduate Programme in Abstract – Gas hydrates are crystalline compounds, solid structures where water assessment of models simulators for hydrate prediction equilibria with and without thermodynamic inhibitors, this If the solution is non ideal, as is the case for |
|
Modelling gas hydrate thermodynamic behaviour - ScienceDirectcom
general phase equilibrium model is presented in detail and is tested against Keywords Review; Modelling; Computational method; Gas hydrates; Multiphase; Petroleum; Equilibrium; inherent complexity, it is not surprising that their program has not proven The ideal solid solution theory for the hydrate phases |
|
[PDF] Hydrate Equilibrium Modelling with the Cubic two-state - SciELO
compounds and from phase equilibrium data of binary gas–water and pressure did not exceed 8 , whereas for multi gas hydrates, these de id solutions, treating them as an ideal liquid solution, and the In this model, the hydrates are solid solutions important to mention that there are evidences which show that |

Non-ideal hydrate solid solution model for a multi-phase
Source:https://i1.rgstatic.net/publication/257209499_Phase_equilibria_with_hydrate_formation_in_H2O_CO2_mixtures_modeled_with_reference_equations_of_state/links/59d8b8290f7e9b12b36857ca/largepreview.png

PDF) Phase equilibria with hydrate formation in H2O + CO2 mixtures
Source:https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs10953-019-00909-8/MediaObjects/10953_2019_909_Fig7_HTML.png
Thermodynamic Modeling Study on Phase Equilibrium of Gas Hydrate
Source:https://www.mdpi.com/energies/energies-13-04135/article_deploy/html/images/energies-13-04135-g001.png
Energies
Source: Free Full-Text

Advancements in hydrate phase equilibria and modeling of gas
Source:http://www.scielo.br/img/revistas/bjce/v35n1//1678-4383-bjce-35-01-265-gf5.jpg

An Examination of the Prediction of Hydrate Formation Conditions
Source:https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs10953-019-00909-8/MediaObjects/10953_2019_909_Fig3_HTML.png
a parallel arithmetic for hardware realization of digital filters
[PDF] Power and Area Efficient Implementation for Parallel FIR Filters
a patient is to receive his daily isoniazid
[PDF] Canadian Tuberculosis Standards 7th Edition Chapter - Canadaca
- anti tuberculosis drugs dosage pdf
- isoniazid and b6 dose
- should isoniazid be taken with vitamin b6
- pyridoxine in tb treatment
- 2(hrze)3 means
- isoniazid dosage
- fixed-dose combination drugs for tuberculosis
- 2hrze/4hr meaning
a pocket style manual apa version
Pocket Style Manual Apa Version - 50000+ Free eBooks in the
a quadratic equation that has two imaginary solutions
[PDF] Mod 11 review answers
- why can't a quadratic equation have one imaginary solution
- quadratic formula
- solving quadratic equations with complex solutions worksheet
- quadratic equation with complex solutions
- solving quadratic equations with complex numbers worksheet answers
- quadratic formula calculator
- quadratic equation calculator
- quadratic equation b^2 4ac