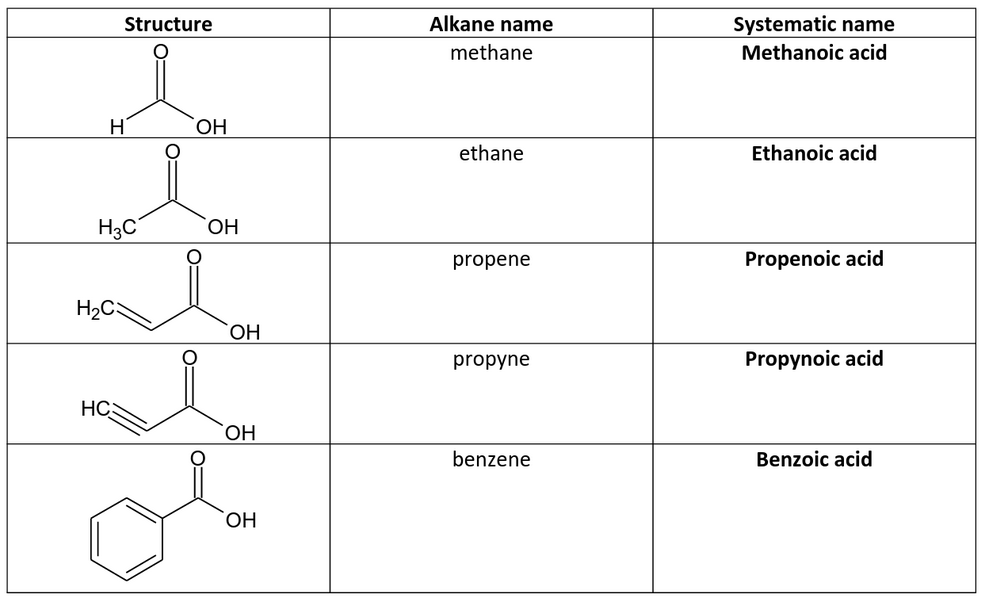
Alkanoic acids are alkane-derived organic compounds that contain the carboxylate (-COOH) functional group.
What functional group is nh2?
The substituent −NH 2 is called an amino group.
Compounds with a nitrogen atom attached to a carbonyl group, thus having the structure R−CO−NR′R″, are called amides and have different chemical properties from amines.
Is COOH a carboxylic acid group?
The carboxyl (COOH) group is so-named because of the carbonyl group (C=O) and a hydroxyl group.
They include carboxylic acids and amino acids.
Is carboxylic acid a carbonyl group?
Both carboxylic acids and esters contain a carbonyl group with a second oxygen atom bonded to the carbon atom in the carbonyl group by a single bond.
In a carboxylic acid, the second oxygen atom also bonds to a hydrogen atom.
In an ester, the second oxygen atom bonds to another carbon atom.
|
Examples Of Carboxylic Acid Functional Group
textbook pilot project functional group of carboxylic acid can help provide Carboxylate ester A molecule or functional group bearing a carbonyl group. |
|
Infrared Spectroscopy
15 de mai. de 2013 Infrared absorption data for some functional groups not listed in the preceding table are ... IR Spectroscopy Tutorial: Carboxylic Acids. |
|
Chapter 5 Carboxylic Acids and Esters
Learn to recognize the carboxylic acid ester |
|
End Functionalization by Ring Opening Polymerization: Influence of
26 de jun. de 2017 or carboxylic acid functionalization independent on the lactide/catalyst molar ... a wide variety of functional groups into the polymer. |
|
Knowledge check
a) A carboxylic acid contains the -OH functional group. Answer: should be -COOH instead of -OH functional group. b) The names of carboxylic acids all end in |
|
Article
10 de dez. de 2013 carboxylic acids with different alcohols and thiols using ... manipulation of the carboxylic acid functional group as. |
|
INFRARED SPECTROSCOPY (IR)
A carboxylic acid functional group combines the features of alcohols and ketones because it has both the O-H bond and the C=O bond. Therefore carboxylic acids. |
|
Surface modification of activated carbon by corona treatment
in functional groups increasing carboxyl acid and decreasing lactone and phenol groups. ... by surface modifications on their chemical structure. |
|
A direct potentiometric titration study of the dissociation of humic
of humic acid with selectively blocked functional groups order to determine their carboxyl and phenol group concentrations and apparent and intrinsic pK ... |
|
Dansylation of hydroxyl and carboxylic acid functional groups
26 de fev. de 2001 Fluorescent detection of compounds by dansylation of chemical groups such as alcoholic hydroxyls and carboxylic acids has applications in ... |
|
Nomenclature of Carboxylic Acids - Angelo State University
Chapter Objectives: • Learn to recognize the carboxylic acid, ester, and related functional groups • Learn the IUPAC system for naming carboxylic acids and |
|
Chapter 18: Carboxylic Acids 181: Carboxylic Acid Nomenclature
The carboxylic acid functional group contains both a hydrogen bond donor (-OH) and a hydrogen bond acceptor (C=O) Carboxylic acids exist as hydrogen |
|
Functional Groups
–CH=O or as –CHO I Carboxylic Acids • contain a “carboxylic acid” group – a carbonyl (C=O) group bonded to a hydroxyl group at the carbonyl carbon atom |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
In the previous Unit, you have studied organic compounds with functional groups containing carbon- oxygen single bond In this Unit, we will study about the |
|
10 Functional Groups of Organic Compounds - UZH Chemistry
Organic acids with pKa values of ca < 5 get fully deprotonated by relatively weak bases Carboxylic acids usually dissolve as carboxylate salts in 0 5 M aqueous |
|
An Introduction to Functional Groups in Organic Chemistry What are
the case of formaldehyde A carboxyl group looks similar to a carbonyl group, but at least one of its sigma bonds is to a second oxygen atom The carboxylic acid |
|
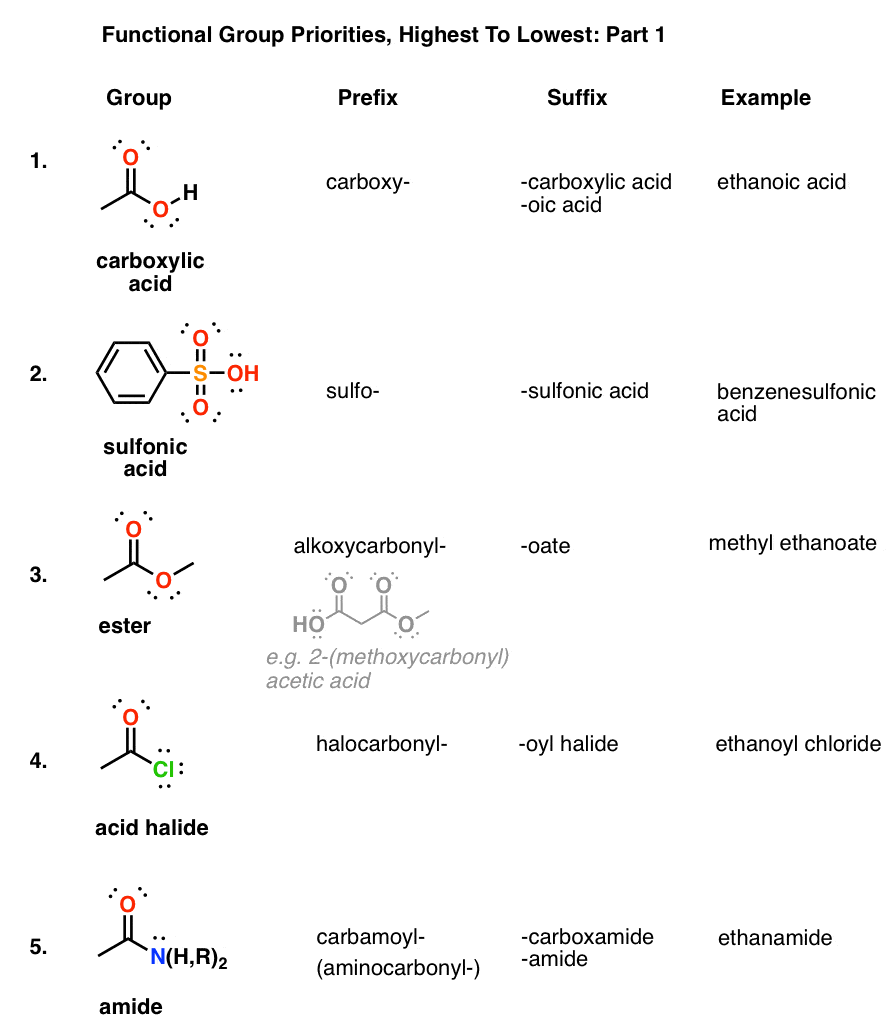
Unit One Part 2: naming and functional groups
Systematic (IUPAC) naming: functional groups gjr-–- 3 Functional group Structure Suffix Prefix General form acid R OH O –oic acid –carboxylic acid |
|
Chapter 7 slides
4 oct 2017 · CHAPTER 7: Organic Functional Groups Functional groups define a molecule' s chemical Carboxylic acids combine their carbonyl group |
| Carboxylic Acid Structure and Chemistry |
|
[PDF] Nomenclature of Carboxylic Acids - Angelo State University
Learn to recognize the carboxylic acid, ester, and related functional groups • Learn the IUPAC system for naming carboxylic acids and esters • Learn the |
|
[PDF] Chapter 18: Carboxylic Acids 181: Carboxylic Acid Nomenclature
The carboxylic acid functional group contains both a hydrogen bond donor ( OH) and a hydrogen bond acceptor (C=O) Carboxylic acids exist as hydrogen |
|
[PDF] Functional groups in Organic Chemistry - RIT
Functional Groups in Organic Chemistry, Teacher's Guide 6 Unknowns Unknown Found in Functional Group Buy from Quantity Price Acetic Acid Distilled |
|
[PDF] Functional Groups
–CH=O or as –CHO I Carboxylic Acids • contain a “carboxylic acid” group – a carbonyl (C=O) group bonded to a hydroxyl group at the carbonyl carbon atom |
|
[PDF] Carboxylic Acids
The carboxyl functional group that characterizes the carboxylic acids is unusual in that it is composed of two functional groups described earlier in this text |
|
[PDF] An Introduction to Functional Groups in Organic Chemistry What are
The carboxylic acid functional group has a hydrogen atom bonded to this oxygen atom It is generally acidic, for reasons we discussed in Chapter One ▫ The |
|
[PDF] Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine molecule, be Carboxylic acids contain a functional group attached to a |
- Acetic acid functional...
- Functional groups of...
- Functional groups in...
- More results
- List of functional groups in organic chemistryCarboxyl group
- Carboxylic acid uses
- Hydroxy carboxylic acid
- Carboxylic acid naming
- Aromatic carboxylic acid
- Carboxylic acids and their derivatives
- Naming carboxylic acids worksheet
- Functional groups
- carboxylic acid functional group structure
- carboxylic acid functional group examples
- carboxylic acid functional group ir
- carboxylic acid functional group formula
- carboxylic acid functional group properties
- carboxylic acid functional group name
- carboxylic acid functional group test
- carboxylic acid functional group isomer
Carboxylic Acid
Source: Carboxylic Acid
Carboxylic Acids The functional group of carboxylic acids
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/2-table-of-functional-group-seniority-priority-for-iupac-nomenclature.gif
Table of Functional Group Priorities for Nomenclature – Master
Source:https://upload.wikimedia.org/wikipedia/commons/b/b5/Carboxylic-acid.svg
Carboxylic acid - Simple English Wikipedia the free encyclopedia
Source:https://d3uigcfkiiww0g.cloudfront.net/wordpress/blog/pics-en/uploads/carboxylic-acids-and-their-systematic-names.png
Carboxylic Acids and their Derivatives
Source: Medical Library
asam karboksilat
Source: Ester
carboxylic acid mcqs pdf
[PDF] GRE ® Chemistry Test Practice Book (PDF) - ETS
- Carboxylic acid Questions and Answers pdf
- Multiple Choice Questions on carboxylic acids
- Organic Chemistry MCQ PDF
- Organic Chemistry multiple choice questions with answers doc
- [PDF] Chapter 5 Carboxylic Acids and Esters - Angelo State Universityhttps://www.angelo.edu › faculty › kboudrea › index_2353
- Chapter Objectives: • Learn to recognize the carboxylic acid
- ester
- and related functional groups. • Learn the IUPAC system for naming carboxylic acids ...[PDF] MCQs - Intermediate Fc Collegehttps://intermediate.fccollege.edu.pk › 2019/07 › 2nd-Year-MCQS
- Of the following four reactions
- formic acid and acetic acid differ in which respect? (a) Replacement of hydrogen by sodium. (b) Formation of ester with alcohol. (c) ...[PDF] CHEMISTRY IN MULTIPLE CHOICE QUESTIONSrep.bsmu.by › bitstream › handle › BSMU
- c) the dissolving of calcium carbide in water; d) the dissolving of sodium bicarbonate in acetic acid. 3. Choose pure chemical elements: a) chlorine gas; b) iron;.[PDF] GRE ® Chemistry Test Practice Book (PDF) - ETShttps://www.ets.org › gre › pdf › practice_book_chemistry
- alcohols
- ethers
- epoxides
- sulfides
- thiols
- aromatic compounds
- aldehydes
- ketones
- carboxylic acids and their derivatives
- amines. C. Reaction Mechanisms — ...Related searchesMCQ on IUPAC Nomenclature PDF
- Multiple Choice questions on carboxylic acid derivatives
- Organic MCQs
- Organic Chemistry multiple choice test bank pdf
- Inorganic Chemistry MCQs PDF
- Questions on carboxylic acids and their derivatives
- multiple choice questions on alkanes
- alkenes and alkynes pdf
- Chemistry MCQs book PDF
carboxylic acid notes pdf
[PDF] Synthesis of Carboxylic Acids
- Carboxylic acid reactions Summary pdf
- Carboxylic acid ppt
- Carboxylic acid Questions and Answers pdf
- Preparation of carboxylic acid pdf
- [PDF] Organic Chemistry II / CHEM 252 Chapter 18 – Carboxylic Acids and ...alpha.chem.umb.edu › files › Overheads › Lecture_Chapter_18
- Introduction. 2. • The carboxyl group (-CO. 2. H) is the parent group of a family of compounds called acyl compounds or carboxylic acid derivatives ...[PDF] Chapter 5 Carboxylic Acids and Esters - Angelo State Universityhttps://www.angelo.edu › faculty › kboudrea › index_2353
- Chapter Objectives: • Learn to recognize the carboxylic acid
- ester
- and related functional groups. • Learn the IUPAC system for naming carboxylic acids ...[PDF] Chapter 12. Aldehydes
- Ketones and Carboxylic Acidshttps://ncerthelp.com › cbse notes › chemistry › Chemistry Notes for c...
- Chemistry Notes for class 12 Chapter 12. Aldehydes
- Ketones and Carboxylic Acids. In aldehydes
- the carbonyl group ( )C=O) is bonded to carbon and ...[PDF] CARBOXYLIC ACIDS AND THEIR ... - Semantic Scholarhttps://pdfs.semanticscholar.org › ...
- Mechanism of Decarboxylation. Dicarboxylic Acids. Methods of preparation of unsaturated monocarboxylic acids. Derivatives of Carboxylic Acids. Acyl Chloride .[PDF] CHAPTER 20 – Carboxylic Acids and Their Derivativeswww2.onu.edu › organic › Chapter20_notes_handout_S20-complete
- Mar 22
- 2020 · Nucleophilic Acyl Substitution. Nearly all the chemistry of carboxylic acid derivatives involves the addition of a nucleophile to the carbonyl.[PDF] Synthesis of Carboxylic Acidsweb.mnstate.edu › jasperse › Chem360 › Handouts
- Chem 360 Jasperse Ch. 20
- 21 Notes. Carboxylic Acids
- Esters
- Amides
- Acid- Chlorides. 1. Synthesis of Carboxylic Acids. 1. From 1º Alcohols and Aldehydes: ...Related searchesCarboxylic acid pdf download
- Naming carboxylic acids worksheet pdf
- Qualitative test for carboxylic acid PDF
- Naming carboxylic acids and esters worksheet
- Carboxylic acids and their Derivatives Notes pdf
- Acidity of carboxylic acids pdf
- Carboxylic acid and its derivatives notes
- Chemical properties of carboxylic acids pdf
- aldehyde ketone and carboxylic acid notes pdf
carboxylic acid pka
[PDF] Carboxylic Acids
- Amide pKaEster pKaCarboxylic acid pKa table
- HSO4 pKa
- [PDF] pKa Table.1www.mch.estranky.sk › file › pka_tables
- see also page 9a for more carboxylic acids. Benzoic acid o m p. Ortho-substituted benzoic acids. H-. 4.20*.[PDF] 20.4 acid–base properties of carboxylic acids - Saplingwww.saplinglearning.com › media › loudon
- This pKa is low enough that an aqueous solution of acetic acid gives an acid reaction with litmus or pH paper. Carboxylic acids are more acidic than alcohols or ...Correlations and Predictions of Carboxylic Acid pKa Values Using ...pubs.acs.org › doi › pdf
- Jan 8
- 2008 · The linear correlations resulting from the fit to the available pKa values can be used to predict the pKa values of similar carboxylic acids. The pKa.THE pKa OF PROTONATED α
- β-UNSATURATED CARBOXYLIC ...https://www.nrcresearchpress.com › doi › pdf
- The pKa of protonated o- (1) and $-substituted (2) benzoic acids and of propionic (3) and acetic (4) acid have been reported. These values range from -6.2* to -8.6 ...[PDF] Carboxylic Acidshttps://pa01000125.schoolwires.net › cms › lib › Centricity › Domain
- The functional group of a carboxylic acid is a carboxyl group
- so named ... As we discussed in Section 2.5B
- carboxylic acids (pKa 4 5) are stronger acids than.[PDF] Chapter 20 The Chemistry of Carboxylic Acidshttps://www.colorado.edu › lab › zhanggroup › chapter-20
- By Le Châtelier's principle
- this drives the carboxylic acid–ester equilibrium ... Consequently
- the HBr–acetic acid system has an effective pKa of about –6.Related searchesAmine pKa
- pKa of acetic acid
- Amino pka
- pKa formula
- More results
- H3O+ pKapKa of acids
- Amine pKa
- Alcohol pKa
- NH3 pKa
- Cyclohexane pKa
- pKa of carboxylic acid derivatives
- carboxylic acid pka table
- carboxylic acid pka value
- carboxylic acid pka amino acids
- carboxylic acid pka list
- protonated carboxylic acid pka
- carboxylic acid derivatives pka
- aromatic carboxylic acid pka
- phenyl carboxylic acid pka
carboxylic acid produced from the acid hydrolysis of butyl acetate
[PDF] Exp't 81
- Hydrolysis rates of esters lab report
- Ester hydrolysis
- Hydrolysis rate definition
- [PDF] Chapter 9 Lecture Notes: Carboxylic Acids
- Amines
- and Amides ...https://www.saddleback.edu › faculty › jzoval › a.version2.0 › lecture...
- Given the structure of a carboxylic acid
- carboxylate ion
- ester
- amide
- or amine ... Predict the products for the acid-catalyzed hydrolysis of an amide. ... The ester formed in the reaction above
- pentyl ethanoate
- has the distinct aroma of bananas.CARBOXYLIC ACIDS AND THEIR DERIVATIVES 1. Carboxylic ...https://link.springer.com › content › pdf
- acid is anhydrous (in such a case esters and amides are prepared) or in aqueous solution ... dissociation and hydrolysis
- either in a medium containing stronger acids or in an ... Procedure: A mixture of 0.1 ml of butyl acetate and 0.5 ml of 50%.[PDF] Exp't 81courses.chem.psu.edu › chem36 › Experiments › Exp81
- Synthesis of n-Butyl acetate via Esterification ... Prelab Exercise: Give the detailed mechanism for the acid-catalyzed hydrolysis of n-butyl acetate. ... Esters can be prepared by the reaction of a carboxylic acid with an alcohol in the presence of ...[PDF] Kinetic Study of Esterification of Acetic Acid with n- butanol and ...https://ejournal.undip.ac.id › index.php › bcrec › article › download
- Esterification of carboxylic acids with alcohols in presence of acid catalysts has been the subject of investigation by many research workers [5-12]. Salts like ... carboxylic acids formed from