|
GLUCOSE HEMI-ACETAL & ACETAL (GLYOSIDE) FORMATION
GLUCOSE HEMI-ACETAL ACETAL (GLYOSIDE) FORMATION: SOME COMMON CONCEPTS IN CARBOHYDRATE (“SUGAR”) CHEMISTRY Carbohydrates • (carbon + hydrate) are molecules |
|
21 Introduction 22 00-Acetals
cyclic acetal formation: 1 Acetals are more easily prepared from aldehydes In aromatic aldehydes and ketones electron donating substituents on the arene |
|
Aldehydes and Ketones 2
Acetal formation is catalyzed by acids commonly p-toluenesulfonic acid (TsOH) Page 17 A Dean–Stark trap is an apparatus used for removing water from a |
|
Addition of Alcohols—Acetal Formation
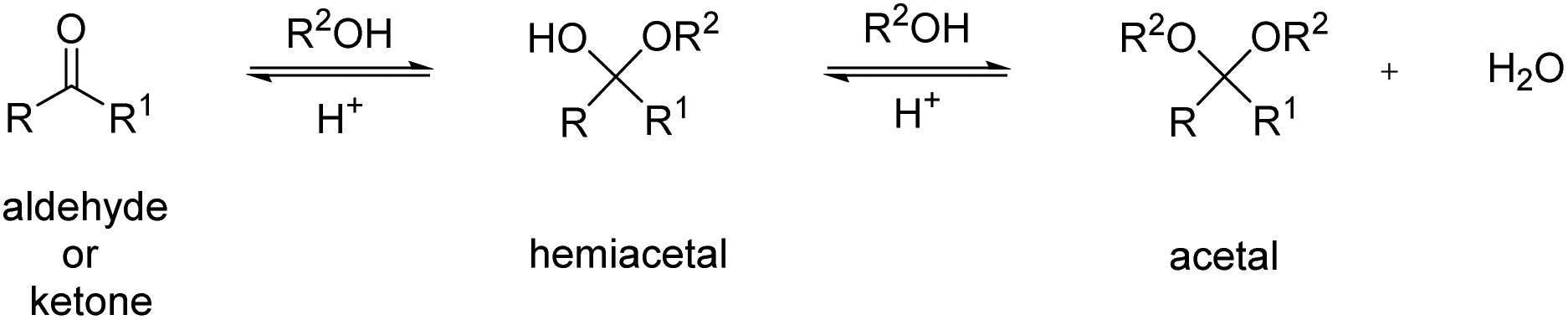
The mechanism for acetal formation can be divided into two parts the first of which is addition of one equivalent of alcohol to form the hemiacetal • The |
|
RÉACTIVITÉ DU GROUPEMENT CARBONYLE
12 jan 2016 · – La formation de l'acétal s'effectue via la formation d'un hémiacétal – Un hémiacétal peut être formé en conditions basiques ou acides |
|
Lecture 9: Acetals
Acetal formation is a reversible process; hence the reverse reaction (acetal hydrolysis) is also acid-catalysed and proceeds through a mechanism that is the |
Les acétals sont formés par traitement d'un aldéhyde ou d'une cétone avec un alcool en présence d'acide (anhydre).
Voir aussi un acétaldéhyde.
Un carbone qui est lié à deux groupes OR est généralement appelé acétal.
|
GLUCOSE HEMI-ACETAL & ACETAL (GLYOSIDE) FORMATION
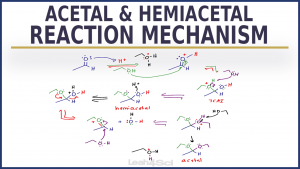
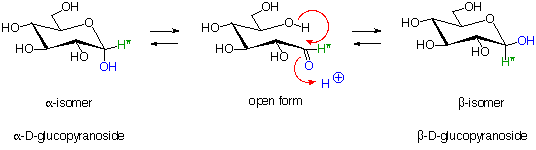
the suffix (ending) for sugar names is: -ose. • the prefix defines the number of carbons: o triose (3 carbons) o tetrose (4 carbons) o pentose (5 carbons). |
|
Highly Efficient Acetalization and Ketalization Catalyzed by
???/???/???? The most general method for the synthesis of acetals or ketals is the reaction of carbonyl compounds with an alcohol or diol. Traditionally ... |
|
Addition of Alcohols—Acetal Formation
Aldehydes and ketones react with two equivalents of alcohol to form acetals. • Acetal formation is catalyzed by acids such as TsOH. |
|
Acetal Formation for Ketones and Aromatic Aldehydes with Methanol1
with acetal determination since when hemiacetal formation occurs We find that rates parallelequilibria for acetal hydrolysis but bear an inverse. |
|
Stereoelectronic control in acetal formation
Stereoelectronic control in acetal formation. NORMAND BEAULIEU' ROBERT A. DICK INS ON |
|
Acetalization Catalysts for Synthesis of Valuable Oxygenated Fuel
???/???/???? Keywords: acetalization; glycerol; acetal; ketal; fuel additives ... articles containing information about the formation of acetals or ... |
|
19.10 ACETALS AND THEIR USE AS PROTECTING GROUPS
When an aldehyde or ketone reacts with a large excess of an alcohol in the presence of a trace of strong acid an acetal is formed. An acetal is a compound in |
|
Multiple-Division of Self-Propelled Oil Droplets through Acetal
Synthesis of 4-(heptyloxy)-1-didecyloxybenzene (diC10A). To evaluate the effect of the primary product by the reaction of C7BA and C10OH on the dynamics of |
|
Reaction of the Acetals with TESOTf? Base Combination
application for an efficient mixed acetal formation (Scheme 1). Deprotection of the Acetals by TESOTf-26-Lutidine or. TESOTf-2 |
|
RÉACTIVITÉ DU GROUPEMENT CARBONYLE - Retour à la page d
12 jan 2016 · Thierry Ollevier CHM-2000 Chimie organique II Mécanisme de formation de l' acétal (en milieu acide ) O H R O R1 H ROH R1 H RO OR |
|
Addition of Alcohols—Acetal Formation
Aldehydes and ketones react with two equivalents of alcohol to form acetals • Acetal formation is catalyzed by acids, such as TsOH • Acetals are NOT formed |
|
Acetal formation in carbohydrates - Squarespace
and maltose Jmol: D Glucose Jmol: Acetal Formation Prof downloads since November 2008 (both PDF and HTML) across all institutions and individuals |
|
Lecture 9: Acetals
draw an arrow-pushing mechanism for acetal hydrolysis; • use acetals as protecting groups for aldehydes and ketones; • use dithioacetals as protecting groups |
|
Acetals and Ketals
ketal hemiacetal acetal Substitution of the Carbonyl Oxygen in Ketones and Reactions such as ester, imine, enamine, and acetal formation are reversible |
|
Aldehydes can react with alcohols to form hemiacetals
out the whole mechanism of acetal formation from aldehyde or ketone plus alcohol through the hemiacetal to the acetal, preferably without looking at the |
|
1910 ACETALS AND THEIR USE AS PROTECTING GROUPS
The first step in the mechanism of acetal formation is acid-catalyzed addition of the alco- hol to the carbonyl group to give a hemiacetal—a compound with an LOR |
|
[PDF] Addition of Alcohols—Acetal Formation
Hydration Level vs Carbonyl Stability • Aldehydes and ketones react with two equivalents of alcohol to form acetals • Acetal formation is catalyzed by acids, |
|
[PDF] GLUCOSE HEMI-ACETAL & ACETAL (GLYOSIDE) FORMATION
the suffix (ending) for sugar names is ose • the prefix defines the number of carbons o triose (3 carbons) o tetrose (4 carbons) o pentose (5 carbons) o hexose |
|
[PDF] Acetals and Ketals
Aldehydes with Two C–O Bonds Acetals and Ketals Addition of Reactions such as ester, imine, enamine, and acetal formation are reversible equilibria that |
|
[PDF] 1910 acetals and their use as protecting groups - Sapling
reactions that begin as additions but involve other types of mechanistic steps A Preparation and Hydrolysis of Acetals When an aldehyde or ketone reacts with |
|
[PDF] Lecture 9: Acetals
The mechanism of acetal hydrolysis is the reverse of the mechanism of acetal formation • Acetals are not stable to aqueous acid and are very readily hydrolysed |
|
[PDF] Aldehydes can react with alcohols to form hemiacetals
out the whole mechanism of acetal formation from aldehyde or ketone plus alcohol through the hemiacetal to the acetal, preferably without looking at the |
|
[PDF] 17_08_09htmlppt [Read-Only]
First stage is analogous to hydration and leads to hemiacetal acid catalyzed nucleophilic addition of alcohol to C=O Mechanism of Acetal Formation |
- acetal formation mechanism
- trimethyl orthoformate acetal formation mechanism
- cyclic acetal formation
- hemiacetal formation
- acetal and ketal formation mechanism
- intramolecular acetal formation
- cyclic hemiacetal formation
- acetal formation mechanism from diol
- acetal formation pdf

On Acetals and Hemiacetals – Master Organic Chemistry
Source:https://www.chemistrysteps.com/wp-content/uploads/2020/01/Acid-Catalyzed-Hydrolysis-of-Acetals.png
Ketal Vs Acetal
Source:http://leah4sci.com/wp-content/uploads/2018/03/Acetal-Hemiacetal-Reaction-Mechanism-Video-Leah4sci-300x169.png
Acetal and Hemiacetal Formation Reaction Mechanism From Aldehydes
Source:http://www.chem.ualberta.ca/~vederas/Chem_164/images/Resource%20Folder/glc_chair.gif
Vederas - Intro Organic
Source:https://pubs.rsc.org/image/article/2019/PY/c8py01219a/c8py01219a-s1_hi-res.gif

Thermoplastic polyacetals: chemistry from the past for a
Source:https://pubs.rsc.org/image/article/2018/OB/c8ob00017d/c8ob00017d-u1_hi-res.gif

Preparation of acetals from aldehydes and alcohols under basic
Source:https://ars.els-cdn.com/content/image/3-s2.0-B9780815520276100078-f07-34-9780815520276.jpg
acetals and hemiacetals
[PDF] Base Catalyzed Formation of a Hemiacetal Reaction - Chemwiscedu
- cyclic acetals and hemiacetals are important biologically as
- cyclic hemiacetal
- acetal formation
- acetal formation mechanism
- acetal and hemiacetal difference
- hemiacetal formation
- cyclic hemiacetal formation
- hemiacetal of ethanal
- acetals and hemiacetals khan academy
- cyclic acetals and hemiacetals are important biologically as
- identifying acetals and hemiacetals
- can acetals and hemiacetals
- formation of acetals and hemiacetals
- difference between acetals and hemiacetals
- stability of acetals and hemiacetals
- hydrolysis of acetals and hemiacetals
acetamide mea wikipedia
Epidemiology of Ocular Chemical Burn Injuries
- cocamide mea
- cocamide dea
- cocamide mea in shampoo
acetamide reacts with br2 and naoh
[PDF] psc2pdf
- reactions of amines pdf
- amine + naoh
- amine reactions
- reaction of amines with nitrous acid mechanism
- amine reaction with water
- amine reaction with hcl
- amine to alcohol
- amine reactions summary
- acetamide reacts with br2 and naoh
- acetamide reacts with br2/naoh to give
acetamide reacts with br2/naoh to give
[PDF] Lab 14: Qualitative Organic Analysis - California State University
- reactions of amines pdf
- reaction of amines with nitrous acid mechanism
- amine reaction with water
- amine + naoh
- amine reactions
- amine reaction with hcl
- amine reactions summary
- alcohol and amine reaction
- acetamide reacts with br2/naoh to give
- acetamide reacts with br2 and naoh