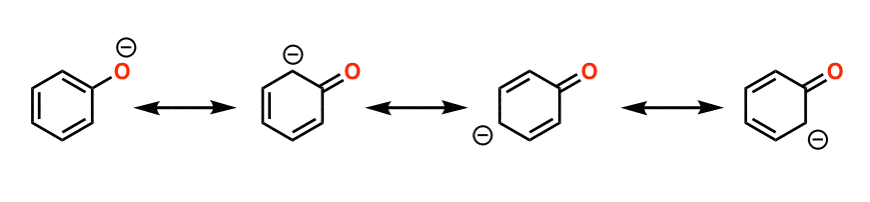
Why? Phenols are much more acidic than alcohols because the negative charge in the phenoxide ion is not localized on the oxygen atom, as it is in an alkoxide ion, but is delocalized-it is shared by a number of carbon atoms in the benzene ring.
Which is more acidic alcohol or phenol?
Phenol is having more acidic character than alcohol.
Why phenols are weak acidic compounds and are high acidity than alcohol?
Acidity of Phenols
Phenol is more acidic than cyclohexanol and acyclic alcohols because the phenoxide ion is more stable than the alkoxide ion.
In an alkoxide ion, such as the one derived from cyclohexanol, the negative charge is localized at the oxygen atom.
Which groups increase acidity of alcohols and phenols?
−NO2, −CN and −X (halogen) groups will increase the acidity of phenol.
These are electron withdrawing groups and stabilize the negative charge of phenoxide ion.
Due to this, the removal of proton becomes easy.
|
15. Benzene and Aromaticity
expected because of hydrogen-bonding. Page 9. 9. Properties of Alcohols and Phenols: Acidity and Basicity. |
|
Hydrogen Bonding Studies. II.1 The Acidity and Basicity of Silanols
acid phenolrespec- tively. The silanols studied are much more strongly acidic |
|
Chapter 7: Alcohols Phenols and Thiols
Phenoxide can be produced by treating with NaOH since it is a stronger acid than an alcohol. (which will not react with NaOH). Page 9. Basicity of Alcohols and |
|
Chapter 17: Alcohols and Phenols
17.3: Properties of alcohols and phenols: acidity and basicity: Like water alcohols are weak Brønsted bases and weak. Brønsted acids. The nature of the R |
|
Degree course in Medicine and Surgery Course: General Chemistry
ALCOHOLS PHENOLS |
|
Degree course in Medicine and Surgery Course: General Chemistry
ALCOHOLS PHENOLS |
|
Hydrogen Bonding between Hydroxylic Donors and MLCT- Excited
< 1 are termed HB acidity and basicity respectively.16. H. 2(B). Accessing KHB (A) Stern-Volmer plots for quenching of 1(T) by alcohols and phenols in ... |
|
Gas-phase basicities of polyfunctional molecules. Part 2: saturated
2018年10月5日 Unsaturated alcohols and phenols. B. Saturated acyclic and cyclic ... Gas-phase basicity and acidity trends in alphabeta-unsaturated amines ... |
|
PHENOLS
16.4 Preparation of Phenols. 16.5 Chemical Properties. Acidity and Basicity of Alcohols and Phenols. Reactions of Phenols. Reactions due to Phenolic. Hydroxyl |
|
Acidity of surface hydroxyl groups
to the basicity of the solvent and the acidity of the phenol. Using this -0.4. Introduction. The nature of the hydrogen bonding interaction between alcohols ... |
|
Chapter 17: Alcohols and Phenols
Chapter 17: Alcohols and Phenols phenol. (aromatic alcohol). pKa~ 10 alcohol. pKa~ 16-18 17.3: Properties of alcohols and phenols: acidity and basicity:. |
|
Hydrogen Bonding Studies. II.1 The Acidity and Basicity of Silanols
Dec. 5 1959. Acidity and. Basicity of .Silanols Compared to. Alcohols admixture with the bases ether and mesitvlene |
|
Alcohols Phenols and Ethers
From the above data you will note that phenol is million times more acidic than ethanol. Arrange the following compounds in increasing order of their acid |
|
120 Chapter 24: Phenols. Alcohols contain an OH group bonded to
24.5: Substituent Effects on the Acidity of Phenols. Electron-donating substituents make a phenol less acidic by destabilizing the phenoxide ion (resonance |
|
23.5 BASICITY AND ACIDITY OF AMINES
In aromatic amines this resonance is at greater chemical shift |
|
6-Alcohols-and-Phenols.pdf
A primary alcohol is oxidized to an aldehyde and then oxidized further to a carboxylic acid. Secondary alcohols are oxidized to ketones. Tertiary alcohols |
|
Chapter 2: Alcohols and Phenols
14 |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
You already know why phenols are more acidic than alcohols. The higher acidity of carboxylic acids as compared to phenols can be understood similarly. The |
|
PHENOLS
16.4 Preparation of Phenols. 16.5 Chemical Properties. Acidity and Basicity of Alcohols and Phenols. Reactions of Phenols. Reactions due to Phenolic. |
|
Chapter 17: Alcohols and Phenols
76 Chapter 17: Alcohols and Phenols phenol (aromatic alcohol) pKa~ 10 alcohol 17 3: Properties of alcohols and phenols: acidity and basicity: Like water |
|
Alcohol and Phenol Tutorial
Alcohols are considered to be relatively non-acidic and non-basic compounds, while most phenols are classified as very weak acids and non-bases Alcohols and phenols are significantly less basic and nucleophilic than amines, in spite of the presence of two pairs of NBEs |
|
Alcohols, Phenols and Ethers - NCERT
Alcohols, phenols and ethers are the basic compounds for the formation of Phenol, also known as carbolic acid, was first isolated in the early nineteenth |
|
Chapter 2: Alcohols and Phenols
PROPERTIES OF ALCOHOLS AND PHENOLS: ACIDITY AND BASICITY Weakly basic and weakly acidic Alcohols are weak Brønsted bases |
|
131 Alcohols and Phenols 131 Alcohols and Phenols 131
Given the relatively low pKa of phenols, will NaOH be a strong enough base to deprotonate a phenol? 13 2 Acidity of Alcohols and Phenols Copyright 2012 John |
|
Alcohols and Phenols Chem 145
Both alcohol and phenol show acidic property to a certain degree since the hydrogen in the hydroxy group (-OH) can be removed by a base as a proton ▫ |
|
Properties of alcohols and phenols lab report - Squarespace
alcohols or phenols in order to increase or decrease acidity The chart below shows the boiling point of a simple basic alcohol with up to four carbon atoms: |
|
Alcohols and Phenols (pp 571-574)
13 2 Acidity of Alcohols and Phenols 571 13 2 Acidity of The conjugate base of an alcohol is called an alkoxide ion, and it exhibits a negative charge on an |
|
Acidity of phenols, effect of substituents on ac - Yengage
Phenols are less acidic than Carboxylic acids (pKa ≈ 5) COMPARISON OF ACIDITY OF PHENOLS AND ALCOHOLS • Phenol exists as resonance hybrid of |
|
[PDF] Chapter 17: Alcohols and Phenols
76 Chapter 17 Alcohols and Phenols phenol (aromatic alcohol) pKa~ 10 alcohol 173 Properties of alcohols and phenols acidity and basicity Like water |
|
[PDF] Chapter 2: Alcohols and Phenols
PROPERTIES OF ALCOHOLS AND PHENOLS ACIDITY AND BASICITY Weakly basic and weakly acidic Alcohols are weak Brønsted bases Protonated |
|
[PDF] Chapter 12: Alcohols and Phenols
Properties of Alcohols and Phenols Acidity and Basicity ▫ Alcohols and phenols are weakly basic and weakly acidic ▫ Reversibly Protonated by strong acids |
|
[PDF] Chapter 7: Alcohols, Phenols and Thiols
Phenols are much stronger acids than alcohols mainly because the corresponding phenoxide ions are stabilized by resonance |
|
[PDF] Alcohols, Phenols and Ethers Alcohols, Phenols and Ethers - ncert
The reaction of phenol with aqueous sodium hydroxide indicates that phenols are stronger acids than alcohols and water Let us examine how a compound in which hydroxyl group attached to an aromatic ring is more acidic than the one in which hydroxyl group is attached to an alkyl group |
|
[PDF] Ch17 Alcohols and Phenols
173 Properties of Alcohols and Phenols Acidty and Basicity weak acid, weak base The effects of alkyl substituents on alcohol acidity ; primarily solvation |
|
Hydrogen bonding by alcohols and phenols---I - ScienceDirectcom
Abstract Evidence is presented that the dimers of alcohols and phenols have an open as the acidity of the hydrogens and the basicity of the oxygens are both |
|
[PDF] physicochemical properties of organic medicinal agents
The acidity of phenols is discussed further in the following section III Alcohol and Phenol Acid Base Properties Alcohols are considered to be relatively non acidic |
- alcohol phenol and ether questions pdf
- benzyl alcohol vs phenol acidity
- alcohol phenol and ether reactions
- acidity of phenol
- how are phenols and alcohols similar
- will alcohol phenol soluble in water why
- alcohols and phenols lab report answers
- acidity of alcohols
- acidity and basicity of organic compounds
- acidity and basicity of salts
- acidity and basicity of amines
- acidity and basicity of salt solutions
- acidity and basicity of amino acids
- acidity and basicity of alcohols
- acidity and basicity of organic compounds pdf
- acidity and basicity of aromatic compounds

Acidity and Basicity of Alcohols – Master Organic Chemistry
Source:https://online.fliphtml5.com/ebwbc/dmhs/files/large/1.jpg?1594970881
AMINES ACIDS \u0026 PHENOLS -1 Pages 1 - 39 - Flip PDF Download
Source: FlipHTML5
Phenols
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/4-resonance-forms-of-conjugate-base-of-phenol-shows-4-resonance-forms-delocallized-charge-more-stable.gif
Acidity and Basicity of Alcohols – Master Organic Chemistry
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2020/03/571-reverse.gif

Acidity and Basicity of Alcohols – Master Organic Chemistry
Source:https://online.fliphtml5.com/ebwbc/dmhs/files/large/2.jpg?1594970881
AMINES ACIDS \u0026 PHENOLS -1 Pages 1 - 39 - Flip PDF Download
Source: FlipHTML5
acidity in drinking water
[PDF] LEVEL OF pH IN DRINKING WATER OF AN OIL AND GAS
- how to raise ph in drinking water
- causes of acidity in water
- how to lower ph in drinking water
- drinking water ph level chart
- alkaline water
- ph of water
- ph of drinking water
- acidity of water
- acidity limit in drinking water
- reduce acidity in drinking water
- total acidity in drinking water
- high acidity in drinking water
- determination of acidity in drinking water
- effects of acidity in drinking water
- causes of acidity in drinking water
- acidity in stomach after drinking water
acidity of alcohols phenols and carboxylic acids
[PDF] Alcohols and Phenols Chem 145
- alcohols and phenols pdf
- acidity of alcohols and phenols
- benzyl alcohol vs phenol acidity
- how are phenols and alcohols similar
- how alcohol differ from phenol
- acidity of phenol
- will alcohol phenol soluble in water why
- alcohol phenol and ether reactions
- acidity of alcohols phenols and carboxylic acids
- acidity of alcohols and phenols
- acidity of alcohols and phenols class 12
- relative acidity of alcohols phenols and carboxylic acids
- acidity of phenol alcohol and water
- difference in acidity of alcohols phenols and carboxylic acids
- compare acidity of alcohols and phenols
- acidity and basicity of alcohols and phenols
acidity of aromatic carboxylic acids ppt
[PDF] Carbonic acids and heterofunctional compounds
- acidity of carboxylic acids ppt
- acidity of carboxylic acids pdf
- increasing order of acidity of carboxylic acids
- carboxylic acid pdf notes
- carboxylic acid questions and answers pdf
- carboxylic acid reactions summary pdf
- preparation of carboxylic acid pdf
- physical properties of carboxylic acids pdf
- acidity of aromatic carboxylic acids
- acidity of aromatic carboxylic acids ppt
- acidity of aliphatic and aromatic carboxylic acids
- acidic strength of aromatic carboxylic acids
- acidic nature of aromatic carboxylic acids
- effect of substituents on acidity of aromatic carboxylic acids
acidity of benzoic acid and ethanoic acid
[PDF] Prelab 6: Carboxylic Acids
- carboxylic acid
- carboxylic acid pdf notes
- acidity of carboxylic acids
- carboxylic acid questions and answers pdf
- acidity of carboxylic acids ppt
- increasing order of acidity of carboxylic acids
- acidity of carboxylic acids pdf
- effect of substituents on acidity of carboxylic acids
- acidity of benzoic acid derivatives
- acidity of benzoic acid substituents
- acidity of benzoic acid and acetic acid
- acidity of benzoic acid and formic acid
- acidity of benzoic acid and phenol
- acidity of benzoic acid ppt
- acidity of benzoic acid
- acidity of carboxylic acid