|
Chapter 5 Carboxylic Acids and Esters
Esters may be broken apart under acidic conditions by water (a hydrolysis reaction) to form a carboxylic acid and an alcohol • This is essentially the |
How does the acidity of a carboxylic acid compare to that of an alcohol?
The acidity of a carboxylic acid is higher than alcohol and even phenols.
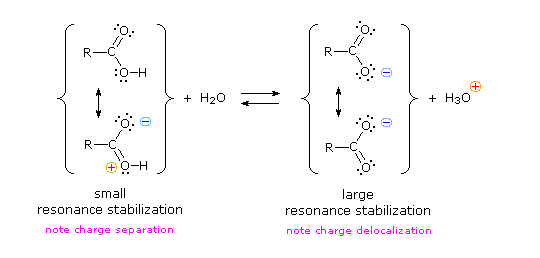
As discussed above, the carboxylate ion, the conjugate base of a carboxylic acid is stabilized by two equivalent resonance structures in which the negative charge is effectively delocalized between two more electronegative oxygen atoms.4 déc. 2019Why are the OH groups of carboxylic acids more acidic than alcohols?
The common explanation for why carboxylic acids are more acidic than other molecules (such as alcohols) is that resonance delocalization of charge stabilizes the conjugate base anion relative to the reactant acid.
Which is more acidic alcohol or carboxylic acid?
Resonance stabilization of their conjugate base.
The resonance effect described here is undoubtedly the major contributor to the exceptional acidity of carboxylic acids.
However, inductive effects also play a role.
For example, alcohols have pKa's of 16 or greater but their acidity is increased by electron withdrawing substituents on the alkyl group.30 mai 2020
|
Chapter 5 Carboxylic Acids and Esters
Esters may be broken apart under acidic conditions by water (a hydrolysis reaction) to form a carboxylic acid and an alcohol. • This is essentially the |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
explain the mechanism of a few selected reactions of aldehydes and ketones;. • understand various factors affecting the acidity of carboxylic acids and their |
|
The role of resonance and inductive effects in the acidity of
Vinyl alcohol and vinyl alkoxide are included in the population analysis for comparison. The greater acidity of carboxylic acids compared to alcohols. |
|
Acidity of Carboxylic Acids: Resonance Delocalization or Induction?
On the basis of calculated acidities of the vinylogues of formic acid and vinyl alcohol Dewar and. Krull have concluded that the higher acidity of |
|
Role of .pi.-Electron Delocalization in the Enhanced Acidity of
acid vinyl alcohol |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
explain the mechanism of a few selected reactions of aldehydes and ketones;. • understand various factors affecting the acidity of carboxylic acids and their |
|
Carboxylic Acid Structure and Chemistry: Part 2
As a result of their relatively acidic nature carboxylic acids will ionize reaction and the role of the acid and alcohol nucleophile are illustrated in ... |
|
Information Sheet for Teachers: St. Elmo Brady (1884 - 1966) This
Derick and the Harvard chemist Arthur Michael disagreed on how the acidity of carboxylic acids was affected by replacing hydrogen atoms on the carbon chain with |
|
Origin of the Acidity of Enols and Carboxylic Acids
Abstract: The origin of the acidity of carboxylic acids and enols has been examined vinyl alcohol to its alkoxide ion the ? electrons of the proton are ... |
|
Carboxylic Acid Structure and Chemistry
Carboxylic acids are referred to as "weak acids" because they partially dissociate in water conjugate base formed from carboxylic acids (where the charge is delocalized by resonance), it is less likely to form Thus alcohols are less acidic than carboxylic acids |
|
Chapter 18: Carboxylic Acids 181: Carboxylic Acid Nomenclature
They are significantly more acidic than water or alcohols Bronsted Acidity (Ch 1 13): Carboxylic acids transfer a proton to water to give H3O+ and carboxylate |
|
Carboxylic Acids
alcohols (pKa 16 18) because resonance stabilizes the carboxylate anion by tivity than carbon increases the acidity of carboxylic acids, often by several orders |
|
CARBOXYLIC ACIDS
Carboxylic acids are organic compounds containing the carboxyl group (-COOH), wherein the alcohols with acidic chromic acid solutions usually forms esters |
|
Carboxylic acids and their derivatives - Caltech Authors
Thus the K, of ethanoic acid, CH,C02H, is 1011 times larger than that of ethanol, CH3CH20H The acidity of the carboxyl group arises, at least in part, from the |
|
Nomenclature of Carboxylic Acids - Angelo State University
Learn the IUPAC system for naming carboxylic acids and esters Alkanes Water Solubility: Carboxylic acid Alcohols Aldehydes/Ketones Ethers Alkanes (low pH) carboxylic acid carboxylate ion 33 Carboxylate Salts Carboxylate Salts |
|
Carboxylic Acids A carbonyl with one OH attached is called a
Acidity As observed previously, carboxylic acids are far more acidic than alcohols This is due to the stability of the anion formed after deprotonation |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
In fact, carboxylic acids are amongst the most acidic organic compounds you have studied so far You already know why phenols are more acidic than alcohols |
|
Carboxylic Acids - De Boeck Supérieur
Esters, which are derived from the reaction of carboxylic acids and alcohols, are responsible for the Ka = 1 8 × 10–5 M The acidic nature of carboxylic acids |
| Carboxylic Acid Structure and Chemistry |
|
[PDF] Chapter 18: Carboxylic Acids 181: Carboxylic Acid Nomenclature
They are significantly more acidic than water or alcohols Bronsted Acidity (Ch 113) Carboxylic acids transfer a proton to water to give H3O+ and carboxylate |
|
[PDF] Carboxylic Acid Structure and Chemistry
As a result of their relatively acidic nature, carboxylic acids will ionize if reaction and the role of the acid and alcohol nucleophile are illustrated in the following |
|
[PDF] Chapter 5 Carboxylic Acids and Esters - Angelo State University
Learn the major chemical reaction of carboxylic acids and esters, and learn how to predict the products of ester Boiling Point Carboxylic acid Alcohols Aldehydes Ketones Ethers Alkanes The Acidity of Carboxylic Acids • Carboxylic |
|
[PDF] Carboxylic Acids
alcohols (pKa 16 18) because resonance stabilizes the carboxylate anion by tivity than carbon increases the acidity of carboxylic acids, often by several orders |
|
[PDF] 204 acid–base properties of carboxylic acids - Sapling
This pKa is low enough that an aqueous solution of acetic acid gives an acid reaction with litmus or pH paper Carboxylic acids are more acidic than alcohols or |
|
[PDF] Acidity of carboxylic acids
Carboxylic acids are similar in some respects to both aldehydes ketones and alcohols Page 6 SCH 206 Dr Solomon Derese Carboxylic acid dimer |
|
[PDF] Carboxylic Acids
Eg ethanoic acid has pKa = 474, (alcohols have pKa ~ 16, so carboxylic acids are about 1011 times more acidic than alcohols) The reason why carboxylic |
- acidity of carboxylic acids
- acidity of carboxylic acids ppt
- why carboxylic acid is more acidic than alcohol
- aliphatic carboxylic acid
- functional group of carboxylic acid
- acidity of carboxylic acids pdf
- carboxylic acid reactions
- effect of substituents on acidity of carboxylic acid
- acidity of carboxylic acid derivatives
- acidity of carboxylic acid pdf
- acidity of carboxylic acid ppt
- acidity of carboxylic acid order
- acidity of carboxylic acid depends on
- acidity of carboxylic acid explain on the basis of
- acidity of carboxylic acid wikipedia
- acidity of carboxylic acid is increased by

pKa Values - The Acid Strength Indicator - Chemistry Steps
Source:https://image.slidesharecdn.com/carboxylicacid-190311055051/95/carboxylic-acid-9-638.jpg?cb\u003d1552616244

Carboxylic acid
Source:https://i1.rgstatic.net/publication/326534132_PCI_Syllabus_BPharm_UNIT-V_Carboxylic_Acids/links/5b52d1260f7e9b240ff526de/largepreview.png
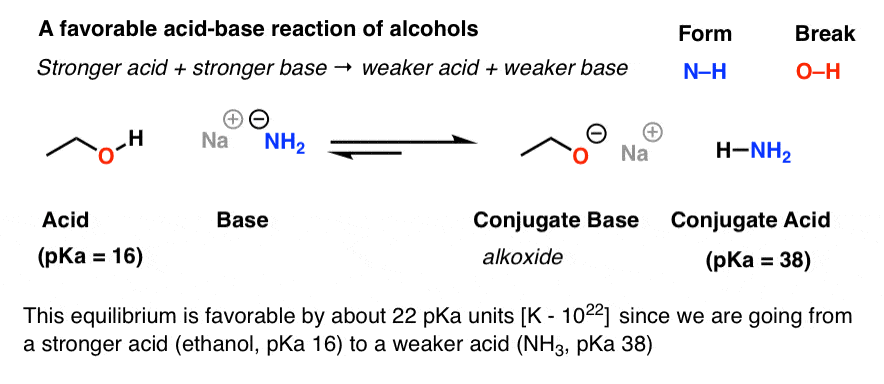
PDF) PCI Syllabus: BPharm UNIT-V: Carboxylic Acids
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/1-acid-base-reaction-of-alcohols-favorable-with-nanh2-stronger-acid-stronger-base-gives-weaker-acid-weaker-base.gif

Acidity and Basicity of Alcohols – Master Organic Chemistry
Source:https://image.slidesharecdn.com/carboxylicacid-190311055051/95/carboxylic-acid-11-638.jpg?cb\u003d1552616244
Carboxylic acid
Source:https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/Images2/carbres1.gif

Carboxylic Acid Reactivity
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/11/1-pka-table-long-going-from-strongest-to-weakest-acid-introductory-organic-chemistry-showing-conjugate-bases.gif
acidity of carboxylic acid and phenol
[PDF] Chapter 5 Carboxylic Acids and Esters - Angelo State University
- Acidity of carboxylic acids
- Monocarboxylic acid
- Aliphatic carboxylic acid
- Why carboxylic acid is more acidic than alcohol
- Aromatic carboxylic acid
- Carboxyl group
- Increasing order of acidity of carboxylic acids
- Properties of carboxylic acid
- acidity of carboxylic acid derivatives
- acidity of carboxylic acid pdf
- acidity of carboxylic acid ppt
- acidity of carboxylic acid order
- acidity of carboxylic acid depends on
- acidity of carboxylic acid explain on the basis of
- acidity of carboxylic acid wikipedia
- acidity of carboxylic acid is increased by
acidity of carboxylic acid slideshare
[PDF] Oxalic acid
- carboxylic acid pdf notes
- acidity of carboxylic acids
- carboxylic acid questions and answers pdf
- carboxylic acid reactions summary pdf
- acidity of carboxylic acids pdf
- preparation of carboxylic acid pdf
- naming carboxylic acids and esters worksheet
- pka of carboxylic acid derivatives
- acidity of carboxylic acid derivatives
- acidity of carboxylic acid pdf
- acidity of carboxylic acid ppt
- acidity of carboxylic acid order
- acidity of carboxylic acid depends on
- acidity of carboxylic acid explain on the basis of
- acidity of carboxylic acid wikipedia
- acidity of carboxylic acid is increased by
acidity of carboxylic acids and derivatives
[PDF] Chapter 16: Carboxylic Acids, Esters, and Other Acid Derivatives
- carboxylic acid and its derivatives pdf
- acidity of carboxylic acids ppt
- acidity of carboxylic acids pdf
- carboxylic acid pdf notes
- effect of substituents on acidity of carboxylic acids
- carboxylic acid questions and answers pdf
- carboxylic acids and esters
- aliphatic carboxylic acid
- acidity of carboxylic acids pdf
- acidity of carboxylic acids with halogens
- acidity of carboxylic acids ppt
- acidity of carboxylic acids depends on
- acidity of carboxylic acids order
- acidity of carboxylic acids wikipedia
- acidity of carboxylic acids and alcohols
- acidity of carboxylic acids and effect of substituents on acidity
acidity of carboxylic acids with halogens
[PDF] 14: Substituent Effects
- acidity of carboxylic acids ppt
- increasing order of acidity of carboxylic acids
- carboxylic acid pdf
- carboxylic acid reactions summary pdf
- acidity of carboxylic acids pdf
- pka of carboxylic acid
- pka of aliphatic carboxylic acid
- pka of carboxylic acid derivatives
- acidity of carboxylic acids pdf
- acidity of carboxylic acids with halogens
- acidity of carboxylic acids ppt
- acidity of carboxylic acids depends on
- acidity of carboxylic acids order
- acidity of carboxylic acids wikipedia
- acidity of carboxylic acids and alcohols
- acidity of carboxylic acids and effect of substituents on acidity