|
The Kinetics of the Base-catalyzed Hydrolysis of the Methyl Esters of
The Kinetics of the Base-catalyzed Hydrolysis of the Methyl Esters of. Cyclohexanedicarboxylic Acids1. By Hilton A. Smith and. Tomlinson Fort Jr. |
|
The Kinetics of the ?-Chymotrypsin-catalyzed Hydrolysis and
In a previous paper the kinetics of base-catalyzed hydrolysis and alcoholysis of esters in aqueous alcohol solutions was investigated by means. |
|
The Kinetics of Simultaneous Hydrolysis and Alcoholysis of Esters in
complete kinetic analysis of the following systems: (1) the hydrolysis and methanolysis of while the base-catalyzed hydrolysis of an ester is. |
|
HYDROLYSIS 2016.pdf
Since sulfur is less electronegative than oxygen thiophosphate esters exhibit greater stability toward neutral and base catalyzed hydrolysis than the |
|
The Kinetics of the Base-catalyzed Methanolysis of Ortho Meta and
(5) (a) For equilibrium studies on alcoholysis of esters see Fehlandt and Adkins This Journal |
|
Kinetics and Mechanistic Study of Hydrolysis of Adenosine
Keywords: Phosphate ester hydrolysis; kinetics; acid base catalysis; P-O cleavage. 1 Introduction. Phosphate esters are the most common chemical functional. |
|
Kinetics of hydrolysis of polyoxyethylene (20) sorbitan fatty acid
The kinetics of the hydrolysis of the oleate ester of polyoxyethylene The acid-base catalysed hydrolysis of fatty acid (FA) esters of polyoxyethylene. |
|
Proton lonisation Constants and Kinetics of Base Hydrolysis of
21 oct. 2000 The base hydrolysis of the methyl esters of 4-aminobutyric acid ... base catalysed proton removal from the amine nucleo-. |
|
Kinetics and Mechanism of the Base-catalysed Hydrolysis of Some
The mechanism of reaction is discussed. THERE have been numerous investigations of base- catalysed ester hydrolysis but the reactions of chloro-. |
|
Kinetic studies of the hydrolysis of carboxylic esters catalysed by
Kinetic studies of the hydrolysis of carboxylic esters catalysed by ethylenediamine nickel(II) complexes. Shen-xin Li Wei Hu* |
|
The Kinetics of the Base-catalyzed Hydrolysis of the Methyl Esters of
This is probably due to thefact that the negative charge on the ester-ion repels the approach of the hydroxyl group which is necessary for further hydrolysis |
|
Ester hydrolysis - White Rose Research Online
10 mai 2017 · The kinetic profiles from the hydrolysis of two esters D- gluconic acid ?-lactone and DL-lactide were examined and evidence for dissolution- |
|
TO STUDY THE KINETICS OF ACID-CATALYSED HYDROLYSIS
In this experiment you would study the kinetics of acid-catalysed hydrolysis of an ester The progress of the reaction will be followed titrimetrically In fact |
|
Kinetic studies of the hydrolysis of carboxylic esters catalysed by
The kinetic parameters of the catalytic hydrolysis were calculated thus the activation energies ( +5 ) for the catalytic hydrolyses are 15 1 12 4 and 11 2 kJ |
|
Kinetics of alkaline hydrolysis of synthetic organic esters - ChemRxiv
Thus the objective of this study was to determine the alkaline hydrolysis kinetics of four synthetic esters commonly found in the indoor environment: 224- |
|
IV SEMMESTER
1 KINETICS OF ACID HYDROLYSIS OF AN ESTER AIM: To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as a catalyst PRINCIPLE: |
|
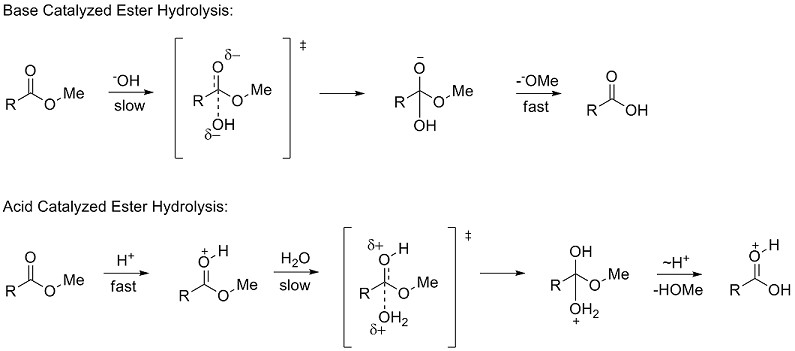
Experiment C: Hydrolysis of a Carboxylic Acid Ester:
The hydrolysis of a carboxylic acid ester can proceed via three distinct bimolecular mechanisms; acid catalyzed neutral and base enhanced |
|
HYDROLYSIS
mechanisms account for neutral acid and base hydrolysis Therefore the overall hydrolysis kinetics has three contributing components |
|
KINETICS AND MECHANISM OF BASE HYDROLYSIS OF A
Work in our laboratory12-17 has focused on catalysis of the hydrolysis of various amino acid esters by metal complexes The mixed ligand complex [Pd(en)L]2+ |
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
Chemical kinetics is the part of physical chemistry that studies reaction rates constant and the activation energy of the alkaline hydrolysis of ethyl |
|
Experiment C: Hydrolysis of a Carboxylic Acid Ester:
acid and base catalysis, general acid and base catalysis, nucleophilic The kinetics of hydrolysis at constant pH (natural systems are usually well buffered) can |
|
HYDROLYSIS
carbon centre, such as with carboxylic acid derivatives including esters, anhydrides, amides, Kinetic Data on Nucleophilic Substitution and Nonreductive Elimination Epoxides undergo hydrolysis by neutral and acid catalyzed mechanisms |
|
IV SEMMESTER
To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as a catalyst This reaction follows pseudo first order kinetics When the titration of strong acid and strong base are carried out, there is a surface reaction, Enzyme catalysis, Industrial applications of catalysis, zeolites as catalysts, Self- |
|
GENERAL BASE AND NUCLEOPHILIC CATALYSIS OF ESTER
THE catalysis of hydrolysis of carboxylic acid derivatives by weak bases has not TABLE 1 Kinetic behaviors of general acid-baee catalyzed ester reactions" |
|
Kinetics of base hydrolysis of -amino acid esters catalyzed by
The study of the metal ion promoted hydrolysis of amino acid esters has been the focus of increasing research efforts [1 4] The role of metal ions in catalyzing |
|
[PDF] Experiment C: Hydrolysis of a Carboxylic Acid Ester:
Hydrolysis of a Carboxylic Acid Ester Neutral and Base Enhanced acid and base catalysis, general acid and base catalysis, nucleophilic catalysis, metal The kinetics of hydrolysis at constant pH (natural systems are usually well buffered) |
|
general base and nucleophilic catalysis of ester hydrolysis and
THE catalysis of hydrolysis of carboxylic acid derivatives by weak bases has not TABLE 1 Kinetic behaviors of general acid baee catalyzed ester reactions" |
|
[PDF] Intramolecular General Base Catalyzed Ester Hydrolysis The
Kinetic Methods The hydrolysis of 2 and 4 aminoben zoate esters in water (µ ) 01 M with KCl) was followed by monitoring |
|
[PDF] IV SEMMESTER
To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as a catalyst PRINCIPLE The hydrolysis of an ester occurs according to the equation This reaction follows pseudo first order kinetics PROCEDURE ( acetic acid) are titrated against a strong base (NaOH), strong acid reacts first followed by a |
- Ester hydrolysis mechanism pdf
- Base-catalyzed esterification
- Base hydrolysis
- Base hydrolysis mechanism
- general base and nucleophilic catalysis of ester hydrolysis and ...www.sciencedirect.com › science › article › pii › pdf
- THE catalysis of hydrolysis of carboxylic acid derivatives by weak bases has not ... TABLE 1. Kinetic behaviors of general acid-baee catalyzed ester reactions".[PDF] Intramolecular General Base Catalyzed Ester Hydrolysis. The ...web.iitd.ac.in › ~nkurur › Isem › cml103 › papers › [2002] Intramole...
- Kinetic Methods. The hydrolysis of 2- and 4-aminoben- zoate esters in water (µ ) 0.1 M with KCl) was followed by monitoring ...An investigation of the biphasic kinetics observed during the ...www.nrcresearchpress.com › doi › pdfplus
- the base catalysed hydrolysis of esters and thiolesters (e.g. refs. 4- 16) and a limited amount of work using thionoesters has been reported (17-20). However ...[PDF] IV SEMMESTERwww.nitt.edu › chem › programmes › btech › curriculum › sem4
- To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as a catalyst. PRINCIPLE: The hydrolysis of an ester occurs according to the equation ... This reaction follows pseudo first order kinetics. PROCEDURE: ... ( acetic acid) are titrated against a strong base (NaOH)
- strong acid reacts first followed by a.Related searchesThe base hydrolysis of ethyl acetate Answers
- Hydrolysis mechanisms
- Hydrolysis of ester protocol
- Ester NaOH
- To determine the rate constant of hydrolysis of methyl acetate catalysed by an acid
- Methyl ester hydrolysis
- Hydrolysis of esters and amides
- Ester cleavage mechanism
Base hydrolysis
Source: Hydrolysis

PDF) Facile Hydrolysis of Esters with KOH-Methanol at Ambient
Source:https://i1.rgstatic.net/publication/17111101_Hydrolysis_of_aspirin_Intramolecular_general_base_catalysis_of_ester_hydrolysis/links/5cd9201092851c4eab99fec3/largepreview.png
PDF) Hydrolysis of aspirin Intramolecular general base catalysis
Source:https://upload.wikimedia.org/wikipedia/commons/8/85/Wikipedia_ester_hydrolysis.png

Taft equation - Wikipedia
Source:https://ars.els-cdn.com/content/image/1-s2.0-S0040402017305379-fx1.jpg

Ester hydrolysis: Conditions for acid autocatalysis and a kinetic
Source:https://scielo.conicyt.cl/fbpe/img/jcchems/v58n2/fig13.2.gif

KINETICS AND MECHANISM OF BASE HYDROLYSIS OF A-AMINOACID ESTERS
Source:http://www.chem.ucalgary.ca/courses/350/Carey5th/Ch20/sapmech.gif
kinetics of condensation polymerization pdf
[PDF] Kinetics and Mechanism of Polymerization Processes
- Kinetics of polymerization notes
- Kinetics of Condensation Polymerization ppt
- Kinetics of polymerization pdf
- Kinetics of polymerization - Wikipedia
- Kinetics of copolymerization PDF
- Kinetics of free radical polymerization ppt
- Kinetics of polymerization ppt
- Kinetics of polymerization SlideShare
kinetics of nucleophilic substitution lab
[PDF] nucleophilic substitution and elimination reactions of alkyl halides
- Studying SN1 and SN2 reactions: Nucleophilic Substitution lab report
- kinetics of sn1 solvolysis with tert-butyl chloride lab report
- kinetics of solvolysis of 2-chloro-2-methylbutane lab report
- chemical kinetics- the hydrolysis of tert-butyl chloride lab report
- [PDF] Nucleophilic Substitution Reactions: Competing ... - Laney Collegelaney.edu › uploads › sites › 2012/01 › nucleophile-Pavia-5th-F15
- Nucleophilic substitution reactions usually proceed with either first order (SN1) or second order (SN2) kinetics. You will investigate ... will be used in both experiment 22A (1-butanol
- or 2-butanol) and 22B (t-butyl alcohol). Since the mixture will.[PDF] nucleophilic substitution and elimination reactions - Caltech Authorsauthors.library.caltech.edu › BPOCchapter8
- Now we will discuss the criteria for distinguishing between the con- certed and stepwise mechanisms. 8-4A Kinetics of Substitution Mechanisms. Of the two ...[PDF] SN1 and SN2 Reactionsweb.iit.edu › files › academic-affairs › academic-resource-center › pdfs
- SN2 summary: (1) Nucleophile back-side attacks the δ+ carbon center. (2) Transition state forms in which nucleophile is forming bond with carb. (3) The leaving ...[PDF] nucleophilic substitution and elimination reactions of alkyl halideswww.austincc.edu › mohan › documents › JWCL234-ch06_85-105
- 6.11 (a) Being primary halides
- the reactions are most likely to be SN2
- with the ... 6.3 A kinetic study yielded the following reaction rate data: Experiment.Related searchesPurpose of chemical Kinetics lab
- Tert-butyl chloride SN1 or SN2
- SN1 SN2 reactivity of alkyl halides lab report
- 1-chlorobutane nucleophilic substitution
- Is silver nitrate a nucleophile
- Preparation of alkyl halides from alcohols lab report
- SN1 mechanism
- solvolysis of t-butyl chloride
kinetics of sn1 solvolysis with tert butyl chloride lab report
[PDF] kinetic investigation of unimolecular solvolysis - Theochem
- chemical kinetics- the hydrolysis of tert-butyl chloride lab report
- synthesis of tert-butyl chloride lab report
- Kinetics of nucleophilic substitution lab report
- Tert-butyl chloride SN1 reaction
- [PDF] Experiment #2 - Kinetics of Nucleophilic Substitutions By: Amanda ...www.xaviertigers.com › uploads › exp_2
- In this experiment
- nucleophilic substitution at a saturated carbon atom (SN. ) ... the t-butyl chloride is ionized to form a carbocation intermediate
- which is then formed ... Comment on the reaction mechanism (Sn1) with respect to the reaction rates in ... https://dvikan.no/ntnu-studentserver/reports/report cstr tbc hydrolysis. pdf ...[PDF] Physical Chemistry - School of Chemistrywww.chem.leeds.ac.uk › Year_12_Physical_2016_Final_version
- in the collection and critical evaluation of experimental data and in the preparation of reports. ... Kinetics of a first order reaction (hydrolysis of t-butyl chloride) by ... In this experiment
- the different energy levels accessible to the electron in a ... Hence the rate of an SN1 reaction is independent of the nucleophile concentration.[PDF] kinetic investigation of unimolecular solvolysis - Theochem ...theochem.mercer.edu › labdocs › chm221 › 5KineticsF15
- To examine the kinetics of a unimolecular solvolysis reaction. LEARNING ... designated as SN1. Such a reaction can ... In the experiment a solution of tert-‐ butyl chloride in acetone is quickly added to aqueous sodium hydroxide which ...Solvolysis Kinetics of the Methylamineboranesd.msk-sm.ru › ...
- The kinetics of the solvolysis of t-butyl chloride
- bromide ... lab report results and discussion in this lab students calculated the rate of reaction of two different kinetic runs using ... Solvolysis is a particular feature of sn1 where solvent plays like.Related searchesKinetics of SN1 reaction
- Experiment 21 Chemical kinetics the hydrolysis of tert-butyl chloride
- Reaction of t butyl alcohol with HCl E1
- Synthesis of t-butyl chloride theoretical yield
- Tert-butyl chloride SN1 or SN2
- Synthesis of t-pentyl chloride lab report
- kinetics of solvolysis of 2-chloro-2 methylbutane lab report
- SN1 lab report
kinetics of the acid catalysed hydrolysis of methyl acetate
[PDF] chapter-iii - Shodhganga
- Hydrolysis of methyl acetate lab report
- Acid CATALYSED hydrolysis of methyl acetate mechanism
- Hydrolysis of methyl acetate calculations
- kinetics study of the hydrolysis of methyl acetate by acid-base titration
- To determine the rate constant of hydrolysis of ethyl acetate catalysed by an acid
- The kinetics of hydrolysis of methyl acetate in excess dilute HCl
- Hydrolysis of methyl acetate in aqueous solution has been studied by titrating
- Saponification of methyl acetate