|
Acylium Ion Formation in the Reactions of Carboxylic Acid
tions of benzoic acid and mesitoic acid derivatives indicates that hindered benzoic acid derivatives may react 2.—log k/kts for the rates of hydrolysis of ... |
|
Solvent Isotope Effects in Catalyzed Hydrolysis of Carboxylic Acid
The latter mechanism is discarded for carboxylic acid derivatives with good leaving groups. (phenyl esters and anhydrides). Solvent isotopeeffects obtained from |
|
Chapter 21 The Chemistry of Carboxylic Acid Derivatives
21.11 (a). The hydrolysis products consist of the conjugate base of the carboxylic acid and an amine: rates of acid-catalyzed ester and amide hydrolyses is ... |
|
Acylium Ion Formation in the Reactions of Carboxylic Acid
tions of benzoic acid and mesitoic acid derivatives indicates that hindered benzoic acid derivatives may react 2.—log k/kts for the rates of hydrolysis of ... |
|
Hydrolysis of Amides to Carboxylic Acids Catalyzed by Nb2O5
25 дек. 2020 г. carboxylic acid derivatives while ... 2 (A) Correlation between initial reaction rates of carboxylic acid formation from hydrolysis of. |
|
IV.* Rate Constants for the Hydrolysis of Some Cyclic Anhydrides
trans-conformation 38 analogous to the conformational situation in carboxylic esters and other carboxylic acid derivatives.21 Succinic anhydride must neces-. |
|
HYDROLYSIS 2016.pdf
carbon centre such as with carboxylic acid derivatives including esters |
|
Secondary Deuterium Isotope Effects in the Reactions of Carboxylic
on the rates of hydrolysis of these carboxylic acid derivatives has been determined. In the basic hydrolysis of ethyl acetate in aqueous solution at 25.0 |
|
Intermediates in the Reactions of Carboxylic Acid Derivatives. IV
indicated that amide hydrolysis is in general similar to ester hydrolysis. The rate of basic hydrolysis is proportional to the concentrations of amide and |
|
Estimation of hydrolysis rate constants of carboxylic acid ester and
Carboxylic Acid Esters. Ester functions are among the most common acid derivatives present in natural as well as man-made chemicals (e.g. lipids |
|
21.7 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
All carboxylic acid derivatives have in common the fact that they undergo hydrolysis (a cleav- age reaction with water) to yield carboxylic acids. A. Hydrolysis |
|
HYDROLYSIS 2016.pdf
carbon centre such as with carboxylic acid derivatives including esters |
|
Acylium Ion Formation in the Reactions of Carboxylic Acid
acid derivatives. The applicability of this mechanistic criterion is tested here by investigatingthe effect of substituents on the rates of hydrolysis of |
|
Solvent Isotope Effects in Catalyzed Hydrolysis of Carboxylic Acid
The latter mechanism is discarded for carboxylic acid derivatives with good leaving groups Solvent isotopeeffects obtained from rate of. |
|
Acylium Ion Formation in the Reactions of Carboxylic Acid
Acylium Ion Formation in theReactions of Carboxylic Acid Derivatives. III. The Hydrolysis dioxane a powerful acceleration of the rate of hydrolysis. |
|
Carboxylic acid participation in amide hydrolysis. External general
carboxylic acid promoted amide hydrolysis is presented which involves intermediates of rate constant on the basicity of the aniline derivative. From the. |
|
The Effect of Charge-Transfer Complexation on the Hydrolysis of
in the bimolecular rate constants for the reaction be- the hydrolysis of a carboxylic acid derivative by proper complex formation between a donor ... |
|
Intermediates in the Reactions of Carboxylic Acid Derivatives. IV
Intermediates in theReactions of Carboxylic Acid Derivatives. IV. boxylic acid derivatives has led to the investiga- ... The rate of basic hydrolysis is. |
|
Chapter 21 The Chemistry of Carboxylic Acid Derivatives
1014 and the discussion on text pp. 1012–1014.) Leaving-group basicities: In the acid-catalyzed hydrolysis of an ester |
|
Hydrolysis of Amides to Carboxylic Acids Catalyzed by Nb2O5
25 ????. 2020 ?. KEYWORDS: Nb2O5 Catalyst Amide hydrolysis |
|
Loudon Chapter 21 Review: Carboxylic Acid Derivatives
Nov 3 2021 · Hydrolysis of derivatives Any of the derivatives of carboxylic acids can be converted back into the acid by using water If it’s a more stable derivative you also need either acid or base as a catalyst |
|
Solved: Carboxylic Acid Derivatives Undergo Hydrolysis To
Carboxylic Acid Derivatives Acid Halide Anhydride Ester Amide Acyl Transfer Reactions Background a Transformation b Relative Reactivity c Mechanism Acid Chlorides/Anhydrides Esters a Hydrolysis 5 Amides i Acid catalyzed ii Base promoted b Transesterification Acid hydrolysis Base hydrolysis i Acid catalyzed ii Base catalyzed 6 |
|
171 Carboxylic Acids and Their Derivatives: Properties and Names
Acid catalyzedester hydrolysis is simply the reverse of esterification reaction In this reaction an ester is treated with water in the presence of a strong acid catalyst such as sulfuric acid Base catalyzedester hydrolysis with a base such as NaOH or KOH is known as saponofication The product of saponification |
|
CARBOXY - authorslibrarycaltechedu
18 Carboxylic Acids and Their Derivatives and phenylmethanol (CHCHOH) The chemical shift of the carboxylic acid proton is here about 9 ppm toward lower magnetic fields than that of the hydroxyl proton of the alcohol This behavior parallels that of the en01 hydrogens of 13-dicarbonyl compounds and is similarly related to hydrogen- |
|
Notes - 19 Carboxylic Acids and Derivatives - CIE Chemistry A
Hydrolysis of acyl chlorides Hydrolysis occurs when an acyl chloride reacts with water The products of hydrolysis are hydrochloric acid and a carboxylic acid The reaction between ethanoyl chloride and water is shown below: CH 3 COCl + H 2 O ? CH 3 COOH + HCl Reactions of acyl chlorides www pmt education |
|
Searches related to rates of hydrolysis of carboxylic acid derivatives filetype:pdf
Hydrolysis of an esters is aqueous base is called saponification Each mol of ester hydrolyzed requires 1 mol of base; for this reason ester hydrolysis in aqueous base is said to be “base-promoted” (not catalyzed) Hydrolysis of an ester in aqueous base involves Nucleophilic acyl substitution |
What determines the rate of hydrolysis of carboxylic acid derivatives?
- The rate of hydrolysis depends on the leaving group, L. Rank the carboxylic acid derivatives according to their reactivity, from the most reactive to the least reactive. Question: Carboxylic acid derivatives undergo hydrolysis to make carboxylic acids.
What are the derivatives of carboxylic acid?
- Chapter 20: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution 20.1: Nomenclature of Carboxylic Acid Derivatives (please read) O C ROH O C ROR' carboxylic acid -oic acid ester -oate O C RO R' lactone cyclic ester O C RCl acid chloride -oyl chloride O C RO O C R acid anhydride -oic anhydride O C RN R' R'' amide -amide O C RN R'
How are nitriles hydrolyzed into carboxylic acids?
- Nitriles are hydrolyzed in either aqueous acid or aqueous base to give carboxylic acids. The corresponding primary amide is an intermediate in the reaction. Base-promoted mechanism (Fig. 20.8, p. 865) Acid-promoted hydrolysis: 184 20.17: Addition of Grignard Reagents to Nitriles.
How do carboxylic acid derivatives decrease electrophilicity?
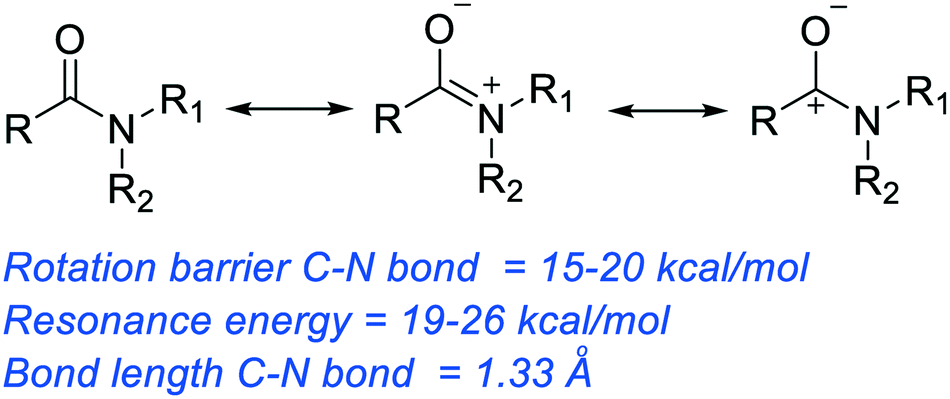
- In carboxylic acid derivatives, the partial positive charge on the carbonyl carbon is stabilized by electron donation from nonbonding electrons on the adjacent heteroatom, which has the effect of decreasing electrophilicity.
|
217 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
Acyl substitution reactions of carboxylic acid derivatives are the major focus of this Ester hydrolysis in aqueous hydroxide is called saponification because it is Figure 21 6 How the structure of a carbonyl compound affects the rates of its |
|
Carboxylic acid Derivatives
Carboxylic acid derivatives are described as compounds that can be converted as carboxylic acid derivatives since they are hydrolyzed to carboxylic A poor leaving group (like alkoxide) gives rise to slow rates (i e reaction does not work) |
|
Chapter 20 Carboxylic Acid Derivatives Nucleophilic Acyl
The rates for hydrolysis are about the same as for esters But thioesters are much more reactive toward amine nucleophiles than esters This helps to account for |
|
HYDROLYSIS
carbon centre, such as with carboxylic acid derivatives including esters, (where the pH is generally buffered) with an overall observed hydrolysis rate constant |
|
100 Chapter 21 Carboxylic Acid Derivatives and Nucleophilic Acyl
201 Relative reactivity of carboxylic acid derivatives: The mechanism of nucleophilic acyl substitution involves two critical steps that can influence the rate of the |
|
85 Chapter 20: Carboxylic Acid Derivatives: Nucleophilic Acyl
Aminolysis: Reaction of acid chlorides with ammonia, 1° or 2° amines to afford amides 4 Hydrolysis: Acid chlorides react with water to afford carboxylic acids 2 |
|
Functional Derivatives of Carboxylic Acids
14 1 What Are Some Derivatives of Carboxylic Acids, and How Are They Named? the rates and experimental conditions under which they undergo hydrolysis |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
We have seen that carboxylic acid derivatives usually react through nucleophilic However, the rate of ester hydrolysis can be substantially increased by |
|
Carboxylic acids and their derivatives - Caltech Authors
acids with the same K, values can differ greatly in the rates at which they give up a proton The nomenclature of carboxylic acids and their derivatives was dis- esters of long-chain acids, can be hydrolyzed with alkali to give the correspond- |
|
[PDF] 217 hydrolysis of carboxylic acid derivatives - Sapling
Acyl substitution reactions of carboxylic acid derivatives are the major focus of this Ester hydrolysis in aqueous hydroxide is called saponification because it is Figure 216 How the structure of a carbonyl compound affects the rates of its |
|
[PDF] Carboxylic acid Derivatives
Carboxylic acid derivatives are described as compounds that can be converted as carboxylic acid derivatives since they are hydrolyzed to carboxylic A poor leaving group (like alkoxide) gives rise to slow rates (ie reaction does not work) |
|
[PDF] PDF (Chapter 18) - Caltech Authors
acids with the same K, values can differ greatly in the rates at which they give up a proton The nomenclature of carboxylic acids and their derivatives was dis esters of long chain acids, can be hydrolyzed with alkali to give the correspond |
|
[PDF] Carboxylic Acid Derivatives
They can be hydrolyzed to carboxylic The typical reaction type of carboxylic acid derivatives is and Y, the rate of reaction is usually in the order 1o > 2o > |
|
[PDF] HYDROLYSIS
carbon centre, such as with carboxylic acid derivatives including esters, (where the pH is generally buffered) with an overall observed hydrolysis rate constant |
|
[PDF] 100 Chapter 21 Carboxylic Acid Derivatives and Nucleophilic Acyl
Relative reactivity of carboxylic acid derivatives The mechanism of nucleophilic acyl substitution involves two critical steps that can influence the rate of the |
|
[PDF] Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution
07_13_46_PM.pdf |
- Hydrolysis of carboxylic acid mechanism
- Preparation of carboxylic acid derivatives
- Stability of carboxylic acid derivatives
- Carboxylic acid derivatives pdf
- [PDF] 100 Chapter 21. Carboxylic Acid Derivatives and Nucleophilic Acyl ...as.vanderbilt.edu › chemistry › Rizzo › Chapter_21
- Relative reactivity of carboxylic acid derivatives: The mechanism of nucleophilic acyl substitution involves two critical steps that can influence the rate of the ...[PDF] Chapter 21 The Chemistry of Carboxylic Acid Derivativeswww.colorado.edu › lab › zhanggroup › chapter-21
- The hydrolysis products consist of the conjugate base of the carboxylic acid and ... of the resonance effect for predicting the relative rates of acid-catalyzed ester.[PDF] Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution ...uomustansiriyah.edu.iq › media › lectures
- In addition
- carboxylic acid derivatives called thioesters and acyl ... nucleophilic acyl substitution reaction
- but the addition is generally the rate-limiting step. Thus
- any factor that makes ... Hydrolysis Reaction with water to yield a carboxylic acid.Mechanisms of Catalysis of Nucleophilic Reactions of Carboxylic ...pubs.acs.org › doi › pdfplus
- reactions of carboxylic acid derivatives through the for- mation of complexes. The rate of hydrolysis of benzo- caine in aqueous solution can be substantially ...[PDF] Estimation of Hydrolysis Rate Constants of Carboxylic Acid ... - EPAcfpub.epa.gov › si_public_file_download
- base-catalyzed carboxylic acid ester hydrolysis rate constants in water and in mixed ... Ester functions are among the most common acid derivatives present in ...Related searchesFunctional derivatives of carboxylic acid
- Carboxylic acid derivatives reactions
- Boiling points of carboxylic acid derivatives
- Carboxylic acid Derivatives practice Problems pdf
- Carboxylic acid derivatives mcat
- Hydrolysis of ester
- In general the order of hydrolysis among acid derivatives is
- Rate of ester hydrolysis
Molecules
Source: Free Full-Text
Carboxylic Acid Derivatives - Organic Chemistry - Lecture Notes
Source:https://pubs.rsc.org/image/article/2021/cy/d0cy02230f/d0cy02230f-f1_hi-res.gif

Hydrolysis of amides to carboxylic acids catalyzed by Nb2O5
Source:https://pubs.rsc.org/image/article/2021/cy/d0cy02230f/d0cy02230f-s1_hi-res.gif
Hydrolysis of amides to carboxylic acids catalyzed by Nb2O5
Source:https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/Images2/acylprb1.gif

Carboxyl Derivative Reactivity
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/10/1-amides-are-the-product-of-a-net-condensation-reaction-between-a-carboxylic-acid-and-an-amine-showing-carboxylic-acid-plus-amine-gives-amide-and-water.gif

Amide Hydrolysis Using Acid Or Base – Master Organic Chemistry
Source:https://upload.wikimedia.org/wikipedia/commons/thumb/e/ed/Fischer_esterification-hydrolysis_equilibrium.svg/824px-Fischer_esterification-hydrolysis_equilibrium.svg.png
rational and intuitive decision making styles
[PDF] Decision-Making Styles and Personality Traits - global business
- Rational decision making
- Rational vs intuitive decision making
- Dependent decision-making style
- Intuitive decision making steps
- Rational decision making model PDF
- How is the rational model of decision making different from bounded rationality and intuition
- Importance of rational decision making
- General decision-making style questionnaire scoring
rational approach to decision making pdf
[PDF] The factors that effect on rational decision making - International
- Rational decision making model PDF
- Rational decision making model example
- Rational decision making model six steps
- Rational approach PDF
- [PDF] Models of Decision-Makingwww.uky.edu › PoliSci › Peffley › pdf › Lau. Models of Decision-Ma...
- economic rational choice approach to decision-making associated with von. Neuman and Morgenstern (1947). This approach provides a standard against which ...Management Theory and Rational Decision Makingwww.emerald.com › insight › content › doi › full › pdf › title=managemen...
- Common to all these rationally based approaches is a detailed
- ordered process of decision making. Thus
- the decision maker is confronted with a problem
- in the ...[PDF] A Discussion of Rational and Psychological Decision-Making ...ejbo.jyu.fi › pdf › ejbo_vol12_no2_pages_12-17
- This paper examines rational and psychological decision-making models. Descriptive and normative methodologies such as attribution theory
- schema theory ...[PDF] The factors that effect on rational decision making - International ...www.allresearchjournal.com › archives › PartC
- Apr 20
- 2019 · going through the formal steps of the rational decision-making model may make ... wide range of disciplines
- because of its ability as a theory.Related searchesRational decision making model advantages and disadvantages
- What is rational approach
- Decision making models PDF
- Rational decision making example
- Decision-making theories and models Pdf
- Rational decision making model Essay
- Rational comprehensive model of decision making
- Bounded rationality model of decision making
rational art definition
[PDF] A RATIONAL-METAPHORICAL PICTURE OF THE WORLD
- Empirical art definition
- Rational meaning
- [PDF] PHILOSOPHY OF ARTS: A contemporary introduction87.98.129.49 › freebookshouse › art › download
- Objections to the aesthetic definition of art 174. Part II The aesthetic ... structures of Poussin's landscapes are rational; that the film Seven is paranoiac. But
- the ...[PDF] Architecture – a rational or intuitive definition?suw.biblos.pk.edu.pl › ...
- Architects
- poets and philosophers have tried to create a definition of architecture for a number of years. As happens in the world of art
- subsequent epochs ...[PDF] Sherlock Holmes and the Adventure of the Rational Manager ...repository.essex.ac.uk › Kociatkiewicz_and_Kostera_-_Holmes_and_t...
- definition
- rooted in the social perspective. ... rational action would thus mean irrational behaviour ... fact
- and art and science
- have become increasingly.[PDF] A RATIONAL-METAPHORICAL PICTURE OF THE WORLDwww.cia.gov › library › readingroom › docs › DOC_0005517516
- May 1
- 1989 · frequent cases works on art are ''streams of words'' without any strictly defined rational meaning. Yet the universally known names of.
rational decision making model
[PDF] rational decision- making: kepner and tregoe's model - FutureLearn
- Rational decision making model PDF
- Rational decision making model example
- Rational decision making model six steps
- Rational decision making model advantages and disadvantages
- [PDF] Intuitive and rational approaches to decision making in ... - Corecore.ac.uk › download › pdf
- employ a range of strategies very different from the oft espoused rational models of decision making. Instead they habitually use strategies that rely very heavily ...Management Theory and Rational Decision Makingwww.emerald.com › insight › content › doi › full › pdf › title=managemen...
- rational decision maker formulates decisions by choosing between ... a continuum of decision making models varying from classical rationality at one pole ...[PDF] rationalist model in public decision making - Journal of Public ...www.jopafl.com › uploads › issue4 › RATIONALIST_MODEL_IN_P...
- Keywords: Decisional process
- public decision
- rationalist model
- rational choice theory. INTRODUCTION. Based on the distinction between objectives ...[PDF] rational decision- making: kepner and tregoe's model. - FutureLearnugc.futurelearn.com › uploads › files › Rational_decision_making
- The rational decision making model is a specific technique for decision making which aims to reach a conclusion based on the decision maker's goals and an ...Related searchesRational decision making model Essay
- Use the rational decision making model when
- Bounded rationality decision making model
- Rational decision making in business organizations
- Decision making models PDF
- Rational model of decision making international relations
- Organizational decision making models
- Rational decision making example