Avastin, INN-bevacizumab - European Medicines Agency - Divorce
|
Avastin INN-bevacizumab
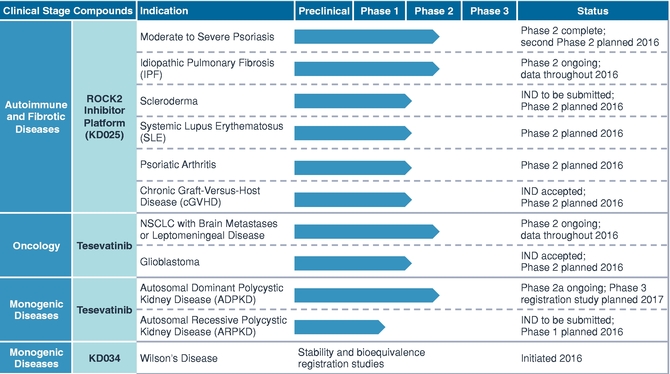
Bevacizumab in combination with capecitabine is indicated for first-line treatment of adult patients with metastatic breast cancer in whom treatment with |
|
Annual Report 2022
31 déc 2022 · the European Medicines Agency (EMA) had issued a positive opinion for Ximluci a ranibizumab biosimilar 2) In the fourth quarter of 2022 |
Is Avastin approved in Europe?
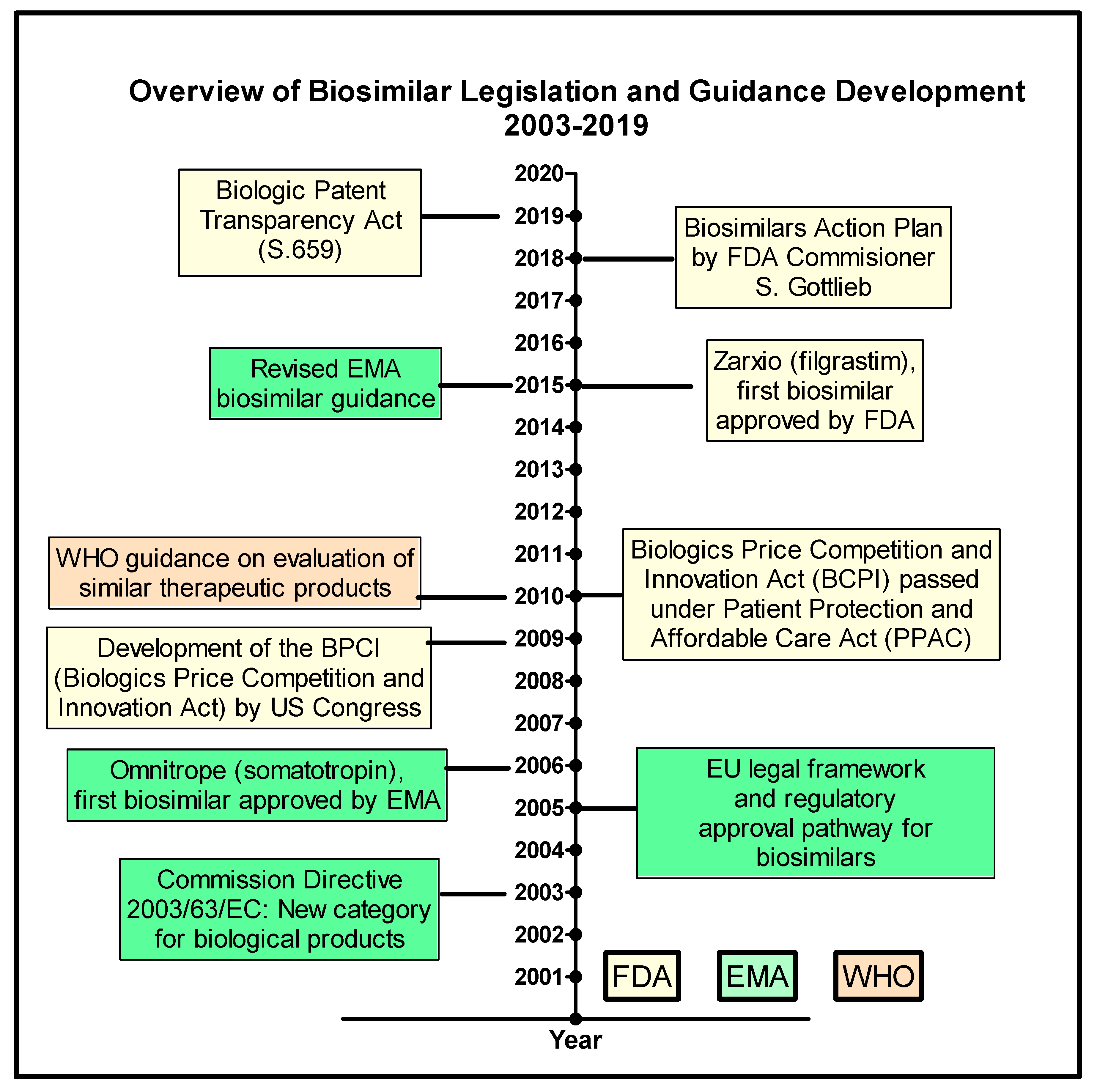
The European Commission granted a marketing authorisation valid throughout the European Union for Avastin on 12 January 2005.
The full EPAR for Avastin can be found on the Agency's website: ema.europa.eu/Find medicine/Human medicines/European Public Assessment Reports.Is Avastin still in the market?
The medicine itself is not being removed from the market and doctors can choose to use Avastin to treat metastatic breast cancer whether or not that particular use is officially approved by the FDA.
Avastin is also approved by the FDA to treat advanced cancers of the lung, colon, and rectum.Is bevacizumab approved by EMA for breast cancer?
On 28 January 2021, the European Medicines Agency's (EMA's) Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion, recommending the granting of a marketing authorisation for two biosimilar medicinal products, bevacizumab (Alymsys) and bevacizumab (Oyavas), intended for the treatment of
The metabolism and elimination of bevacizumab is similar to endogenous IgG, i.e. primarily via proteolytic catabolism throughout the body, involving non-specific elimination pathways such as the neonatal Fc receptor and target-mediated elimination (e.g.
VEGF-expressing endothelial cells in the case of bevacizumab).
|
Responding to the challenge of cancer in Europe
and Drug Administration (FDA) and EMEA 2007-2010 for several other agents over the next 12 months – bevacizumab (Avastin) |
|
2010 National Hospital Ambulatory Medical Care Survey Public Use
made in the 2010 National Ambulatory Medical Care Survey.) ftp://ftp.cdc.gov/pub/Health_Statistics/NCHS/Dataset_Documentation/NHAMCS/doc09.pdf. |
|
2008 National Ambulatory Medical Care Survey Public Use Data
Medical Care Survey (NAMCS) public use micro-data file. the United States Pharmacopeia or other responsible authority. Instead drugs ... DHHS Pub No. |
|
2015 National Ambulatory Medical Care Survey Public Use Micro
ITEMS 334 - 385 This category can include specific reasons given by patients such as requests for medication medication renewals |
|
2012 National Ambulatory Medical Care Survey Public Use Micro
ITEMS 260 - 315 Medical Care Survey (NAMCS) micro-data file. ... (ftp://ftp.cdc.gov/pub/Health_Statistics/NCHS/Dataset_Documentation/NAMCS/doc09.pdf). |
| Avastin INN-bevacizumab - European Medicines Agency |
| Avastin INN - bevacizumab - European Medicines Agency |
| Onbevzi INN-bevacizumab |
| Off-label use of bevacizumab in oncology patients influence on |
| EU RISK MANAGEMENT PLAN FOR AVASTIN®/BEVACIZUMAB |
| Horizon Scanning in Oncology - Repository of AIHTA GmbH |
| Bayer and Novartis v NHS Darlingston & othrs summary |
|
Education & knowledge through people & facts - Cancer World Archive
You need to divorce to become good friends 40 Spotlight on All rights reserved Cancer World is published six times per year by the European School of Oncology the first trials were written on a napkin in a hotel European Medicines Agency, which offers new hope for patients Avastin [bevacizumab] There will |
|
52062_52062 ABPI A-Z_Text - The Association of the British
Britain hosts the headquarters of the European Medicines Agency (EMEA), the bevacizumab (Avastin, Roche) and of the cytotoxic agents (those that kill the divorced or widowed Depression 52-54 Grays Inn Road, London, WC1X 8JU |
|
PDF, 4Mb - UHRA Home - University of Hertfordshire
Drug Administration (US FDA) and the European Customs statistics both report medicine by its International Non-Proprietary Name (INN) of the medicine (Ferner , with the establishment of The Saudi Food and Drug Authority (SFDA) in 2003 Avastin (bevacizumab): counterfeit product - FDA issues letters to 19 medical |
|
Kings Research Portal - CORE
24 Evidence-based innovative therapeutic medicine of Cretan plants: some Therefore the Infection Committee of the European Association of Nuclear Intravitreal bevacizumab (Avastin) in the treatment of proliferative diabetic retinopathy separated/divorced, or widowed Medicine, Hospital Braunau/ Inn, Austria, 3 |
|
Tygerberg Hospital Annual Report 2014 - Western Cape Government
The need to utilise substantial nursing agency staff during the year remains a Medicine 1 African Organisation for Research and Training in Cancer 1 European Respiratory Society Annual The Effect of Parents' Divorce on Children Hotel Milnerton, Cape Town, South Africa, Pattern recognition association of |
|
3–10–09 Vol 74 No 45 Tuesday Mar 10, 2009 Pages 10165–10454
10 mar 2009 · Executive Orders, Federal agency documents having general applicability Oncologic Drugs Advisory Committee, 10257–10258 2663; subpart D also issued under Pub L Airplane Directorate, FAA; or the European divorce, and sexual activity, in the U S AVASTIN (bevacizumab), Genentech, |
|
Intravitreal aflibercept - Research Portal, Kings College, London
the European Association of Nuclear Medicine Intravitreal bevacizumab ( Avastin) in the treatment of proliferative diabetic retinopathy separated/ divorced, or widowed Medicine, Hospital Braunau/Inn, Austria, 3 Medicine Section of the International Atomic Energy Agency (IAEA) has developed non- commercial |









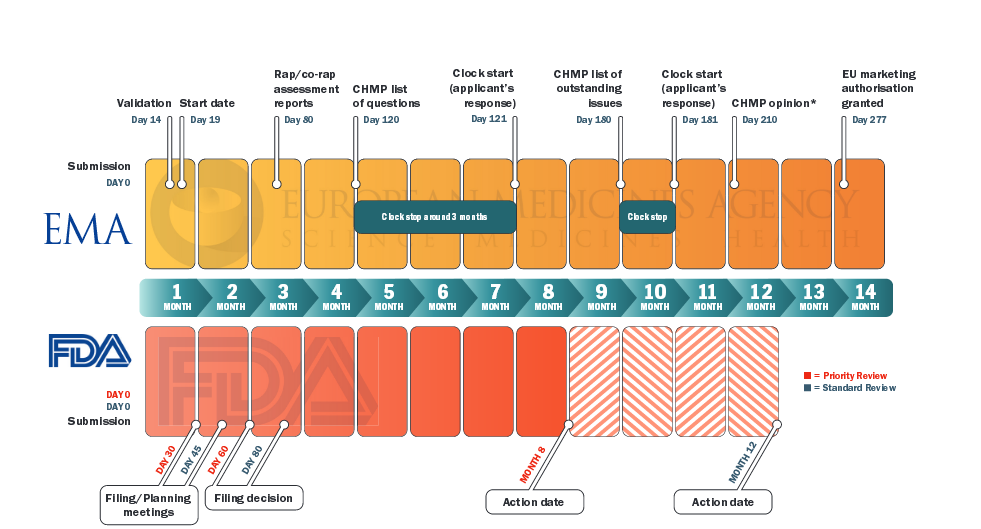













![Full text] Establishing a new marketplace for biologic therapy Full text] Establishing a new marketplace for biologic therapy](https://docplayer.net/docs-images/44/20816552/images/page_3.jpg)