preparation of a solution from solid
|
Laboratory Solution Preparation
The following exam ples illustrate the calculations for preparing solutions. If starting with a solid use the following procedure: • Determine the mass in |
|
Laboratory Solution Preparation
The following exam- ples illustrate the calculations for preparing solutions. If starting with a solid use the following procedure:. |
|
1 Preparation of Liquid and Solid Samples - Wiley-VCH
Sampling and sample preparation of liquids and solids often present significant require the use of an abrasive or even a strong HCl solution. |
|
Pharmaceutical Compounding and Dispensing Sample.pdf
When solid dosage forms are taken orally the drug When preparing a solution |
|
Preparation of Li2S-P2S5 Solid Electrolyte from N-methylformamide
Preparation of Li2S-P2S5 Solid Electrolyte from N-methylformamide solution and. Application for All-Solid-State Lithium Battery. Shingo Teragawa Keigo Aso |
|
Solid-State Preparation of Metal and Metal Oxides Nanostructures
20 janv. 2022 A number of solution fabrication methods have been developed for the synthesis of metal and metal oxides nanoparticles but few solid-state ... |
|
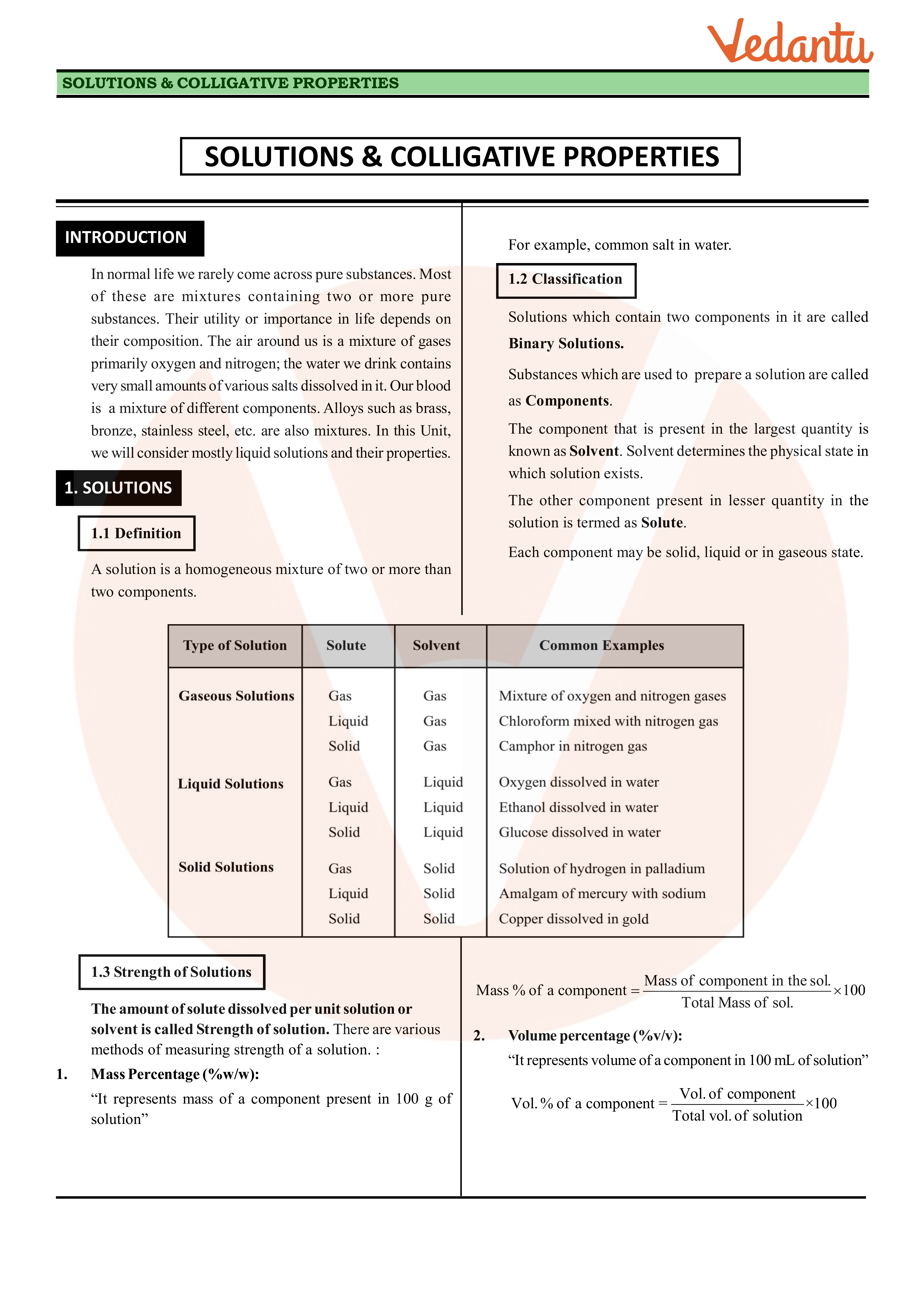
(2) Preparation and Dilution of Solutions
Understanding how to prepare solutions and make dilutions is an essential Once the solid is dissolved the volume is transferred to 100 ml volumetric. |
| Preparation and characterization of zirconia-alumina system via |
|
Preparation & Characterization of Solid Solutions of Phosphate
Phosphate and vanadate apatites of calcium and a series of their solid solutions over the entire composition range have been prepared. |
|
Laboratory Math II: Solutions and Dilutions
To make a solution from a solid solute (that which is being When preparing to perform serial dilutions there are three main components to consider. |
|
Preparation • Over 300 recipes of common - St Norbert College
of the solution and its method of preparation must be as accurate as possible The Flinn Laboratory Solution Preparation reference section is designed for both the novice and experienced solution maker It provides valuable information on the basic concepts of preparing solu tions and instructions for preparing most solutions required in the high |
|
Introductory chemistry : an atoms first approach [First
Preparing a Standard Solution from a Solid A solution of known concentration can be prepared from solids by two similar methods Although inherent errors exist with each of the methods with careful technique either will suffice for making solutions in General Chemistry Laboratory |
|
1 Preparation of Liquid and Solid Samples - Wiley-VCH
Preparation of Liquid and Solid Samples Brian M Cullum and Tuan Vo-Dinh 1 1 Introduction Sampling and sample preparation of liquids and solids often present signi?cant challengesforreal-worldquantitativeanalysesusingspectrometrictechniques(e g UV–vis and infrared absorption luminescence and Raman spectroscopies) Very |
|
Preparation and Dilution of Solutions
To prepare standard solution 1 1 ml of the stock 2 0M solution is needed and volume made up to 2 ml with distilled water (never forget to mix properly) To prepare standard solutions 2-4 1 ml of the previously diluted solution is taken and volume is made up to a final volume of 2 ml by the addition of distilled water |
|
PREPARATION AND STANDARDIZATION OF SOLUTIONS
4 Preparation of 0 1 N sodium thiosulphate solution (Na 2 S 2 O 3 5H 2 O) 1 Dissolve approximately 24 8 g of sodium thiosulphate crystals in previously boiled and cooled distilled water 2 Make the volume to 1000 ml 3 Store the solution in a cool place in a dark colored bottle 4 After storing the solution for about two weeks filter if |
|
Searches related to preparation of a solution from solid filetype:pdf
Factors Involved in the Preparation of Solutions from solids Obtain 2 small (about the volume of a pea) crystals of rock salt Grind one portion to a powder Place this ground portion in a medium test tube and place the unground portion in another Fill each test tube about 1/3 full with water |
How to prepare a solution from a solid?
- Preparation of a Solution from a Solid The procedure for preparing a solution of known molar concentration from a solid is as follows: 1. The solute is weighed accurately and transferred to a volumetric flask of the desired volume. 2. Water is added to the flask, which is then swirled to dissolve the solute.
How is a basis solution prepared?
- A basis solution is prepared by adding a solid base to water. A result of the action: water on some solid bases and the production of hydroxide ions. For example, when NaOH is added to water (H2O), the result of the reaction is Na+ and OH-. OH- is called hydroxide ions. This reaction includes solvent and solute.
How do you prepare a molal solution?
- Preparation of a Molal Solution First, determine the molar mass of the solute. Next, decide on what the molality of the solution will be, and what mass of solvent will be used. Weigh out the needed mass of solute. Weight out the needed mass of solvent. Add the solute to a beaker before adding the solvent.
How do you prepare a laboratory solution?
- Here are some general steps for preparing a laboratory solution: Gather materials: Assemble all the necessary materials, including a clean container for the solution, a calibrated balance or scale, a calibrated measuring cylinder or pipette, and the necessary chemicals.
|
Laboratory Solution Preparation
If starting with a solid, use the following procedure: • Determine the mass in grams of one mole of solute, the molar mass, MMs • Decide volume of solution |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
A 10 NaCl solution is equal to 10 g dissolved in 100 ml of solvent Because the solid is measured based upon its weight (w) while the solvent is measured based |
|
Solution Preparation (From Solid Reagent)
Solution Preparation (From Solid Reagent) • You need to know the amount of solution required and the desired molarity – Determine the number of grams that |
|
Solutions Preparation ‐ A solution that has a known, accurate
Preparing a standard solution from a solid Step 1: Calculate what mass of solid is needed to make the required volume and concentration of solution n = cv |
|
(2) Preparation and Dilution of Solutions
1 Molar solution is a solution in which 1 mole of solute is dissolved in a total You are provided with solid NaOH, Prepare 50ml with 0 08M NaOH solution |
|
Solutions and Dilutions - NIH Office of Intramural Training and
In this case, we will be looking at how to make a solution from a solid substance or “solute” being dissolved in a liquid, or “ solvent” To do this, you need to know: |
|
1 Preparation of Liquid and Solid Samples - Wiley-VCH
Sampling and sample preparation of liquids and solids often present significant In order to ensure that the entire analyte has been dissolved in the solution |
|
Andreas Help Sheet on Preparing Solutions - Duke Wiki
Mix 20ml EtOH with 80ml H2O for a final volume of 100ml Next, let's look at Molar (M) solutions First, know the definition of a Molar solution: 1M = 1MW |






/assortment-of-laboratory-glassware-flasks-680805969-59db9d15845b340012f6d3e5.jpg)















/volumetric-flask-containing-solution-of-potassium-dichromate--vi---k2cr2o7--and-other-flasks-of-transition-metal-salts--dry-chemicals-and-solutions--compounds-with-transition-metals-are-often-colored-702545755-5a579379eb4d520037b7a397.jpg)


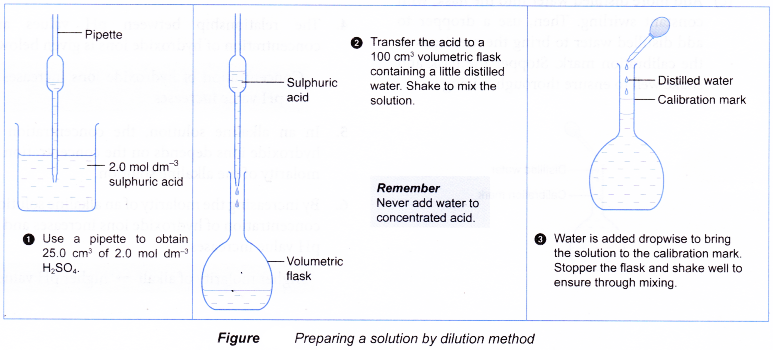
![Solid solutions [SubsTech] Solid solutions [SubsTech]](https://www.cbsetuts.com/wp-content/uploads/2018/08/NCERT-Class-9-Science-Lab-Manual-Solution-Colloids-Suspension-1.png)







![1 [5p] Temperature of a large solid particle 2 [6p] Temperature of 1 [5p] Temperature of a large solid particle 2 [6p] Temperature of](https://www.substech.com/dokuwiki/lib/exe/fetch.php?w\u003d\u0026h\u003d\u0026cache\u003dcache\u0026media\u003dinterstitial_solution.png)
![Solid solutions [SubsTech] Solid solutions [SubsTech]](https://static.kopykitab.com/blog/wp-content/uploads/2015/05/CBSE-Syllabus-For-Class-12-Chemistry.png)