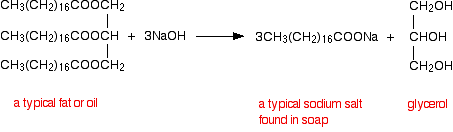
products of alkaline hydrolysis of ethyl ethanoate
|
A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate Using
Industrial importance of the reaction product sodium acetate |
|
Determination of the Expression Rate of Ethyl Acetate Hydrolysis
where subscripts A B refer to alkali (sodium hydroxide) |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide) HCl was reactant or product in a particular reaction is. |
|
Chapter 5 Carboxylic Acids and Esters
predict the products of ester synthesis and hydrolysis reactions. Ethanoic acid/acetic acid is the main ingredient in vinegar. |
|
Reaction Rate of the Alkaline Hydrolysis of Ethyl Acetate
sodium acetate or ethanol. The addition of the products actually retarded the overall reaction rate but it is too small to illustrate the deviation from the |
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
rate or rate of reaction for a reactant or product in a particular reaction rate constant and the activation energy of the alkaline hydrolysis of ethyl. |
|
Demulsification of a Mixture of Di-Chloro-Floro-Acetophenone Di
1 févr. 2017 Industrial significance of the reaction product sodium acetate and ethanol ... Kinetics of Alkaline Hydrolysis of Ethyl Acetate. |
|
DIETHYL PHTHALATE
Diethyl phthalate is used in a wide range of consumer goods. It has been the subject Major metabolism is by partial hydrolysis to ethanol and the mono-. |
|
HKDS EE Chemistr yy && Combine dd Scienc ee (Chemistry
12 déc. 2012 No. of items ... between ethenyl ethanoate and ethyl ethanoate. (2) ... One of the products in the alkaline hydrolysis of Y has a. |
|
11177.03 NEW SPEC ONLINE MARKING GCE Chemistry A21
5 juin 2018 (b) (i) Show the organic products for the reaction of ... 13 Ethyl ethanoate is hydrolysed in alkaline conditions as follows:. |
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
Determine the rate constant and the activation energy of the alkaline hydrolysis of ethyl acetate using sodium hydroxide This experiment illustrates a bimolecular reaction (reacting species are ethyl acetate and sodium hydroxide): CH 3 –COO–CH 2–CH 3 + NaOH ? CH 3–COONa + CH 3–CH 2–OH E |
|
Reaction rate and rate constant of the hydrolysis of ethyl
Determine the rate constant and the activation energy of the alkaline hydrolysis of ethyl acetate using sodium hydroxide This experiment illustrates a bimolecular reaction (reacting species are ethyl acetate and sodium hydroxide): CH 3 –COO–CH 2–CH 3 + NaOH ? CH 3–COONa + CH 3–CH 2–OH E |
|
Kinetics of alkaline hydrolysis of synthetic organic esters
Products of SE hydrolysis include alcohols organic acids (or carboxylate salt under alkaline conditions) or in the case of di- and tri- esters smaller ester compounds Elevated gas- phase concentrations of volatile organic compounds (VOCs) from various indoor sources |
|
KINETICS OF HYDROLYSIS OF ETHYL ACETATE - Simon Fraser University
hydrolysis of ethyl acetate which can be represented by the chemical equation: CH 3COOC 2H 5 + OH ? CH 3COO ? + C 2H 5OH The kinetics data will be obtained • at 25 C under three different initial mixing conditions (Ethyl acetate in excess hydroxide in excess second order conditions – ie 1:1 mixing) • at ~35 C under second order |
|
Determination of rate of reaction and rate constant of the
In this hydrolysis of ethyl acetate with sodium hydroxide hydrochloric acid was used as catalyst to accelerate the reaction mixture 1ml and 2ml of ethyl acetate was injected separately into the 500 ml conical flask which contains 0 05N of sodium hydroxide and thoroughly mixed |
|
Searches related to products of alkaline hydrolysis of ethyl ethanoate filetype:pdf
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide) HCl was used as catalyst to accelerate it 1ml and 2ml of ethyl acetate was injected separately into the 500ml reactor vessels which contains 0 05N of NaOH and thoroughly mixed |
How is ethyl acetate hydrolyzed with sodium hydroxide?
- In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide), HCl was used as catalyst to accelerate it. 1ml and 2ml of ethyl acetate was injected separately into the 500ml reactor vessels which contains 0.05N of NaOH and thoroughly mixed.
What is the activation energy of ethyl acetate in hydrolysis reaction?
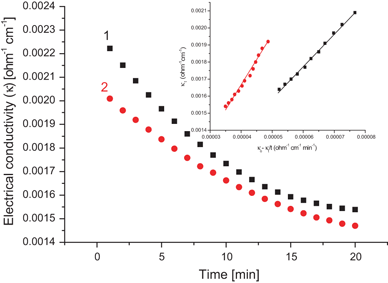
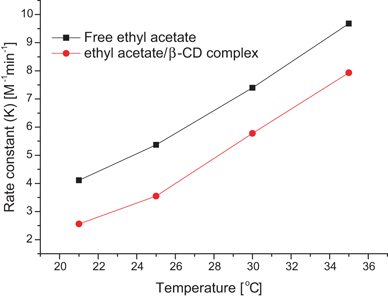
- The results show that hydrolysis of the ethyl acetate is a forward one-way second order reaction. The analysis of the experimental data showed that the activation energy is 29.775 kJ mol-1and frequency factor 27038. Keywords:Activation energy, arrhenius equation, ethyl acetate, hydrolysis reaction, batch reactor
How does ethyl acetate react with ethanol?
- OH Ethyl acetate sodium hydroxide sodium acetate ethanol As the reaction proceeds, each hydroxide ion removed in the formation of ethanol removes one molecule of ethyl acetate, being the number of moles of either OH- or ethyl acetate so removed, the concentration of the reactants decreases.
What are the products of SE hydrolysis?
- Products of SE hydrolysis include alcohols, organic acids (or carboxylate salt, under alkaline conditions), or in the case of di- and tri- esters, smaller ester compounds.
|
A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate Using
Industrial importance of the reaction product, sodium acetate, necessitate for process improvement in terms of maximum conversion and economical usage of raw |
|
Reaction rate and rate constant of the hydrolysis of ethyl - Sci Hub
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide), HCl was used as catalyst to reactant or product in a particular reaction is intuitively |
|
Questions ESTERS: HYDROLYSIS - Chemguide
The equation below shows the acid hydrolysis of the ester ethyl ethanoate using an c) Write the formulae for the products if you hydrolysed methyl propanoate in the Esters are more usually hydrolysed by alkaline hydrolysis using sodium |
|
Chemistry - Pearson qualifications
5 jui 2018 · (a) A pale yellow precipitate is formed when iodine in alkali reacts with (1) A W and X (i) Write the equation for the hydrolysis of ethyl ethanoate using sodium hydroxide and name the organic products State symbols are not |
|
Exercise 8 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
rate or rate of reaction for a reactant or product in a particular reaction is intuitively the rate constant and the activation energy of the alkaline hydrolysis of ethyl |
|
39 Carboxylic acids and esters
d Methyl propanoate 2) Write balanced chemical reactions for the alkaline hydrolysis of the following Name the products for each reaction: a Ethyl ethanoate b |
|
Download - Bradford Scholars - University of Bradford
Alkaline Hydrolysis of Mlethyl and Ethyl Acetate in Water 47 2 1 4 Solvent Effects so definite decompositicn rate to yeild the products of the reaction The |
|
Estimated carboxylic acid ester hydrolysis rate constants for - CORE
28 sept 2011 · For six representative volatile esters (ethyl butanoate, ethyl hexanoate, ethyl octanoate, isobutyl acetate, isoamyl acetate, and hexyl acetate) |
























![PDF] A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate PDF] A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate](http://www.scielo.br/img/revistas/jbchs/v18n1/18t2.gif)

![PDF] A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate PDF] A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate](https://demo.vdocuments.mx/img/378x509/reader024/reader/2021010504/5750057e1a28ab1148a4b142/r-1.jpg)


![PDF] A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate PDF] A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate](https://data01.123doks.com/thumb/qo/5l/jkky/Pki4ZiPT5VIqTD1so/cover.webp)