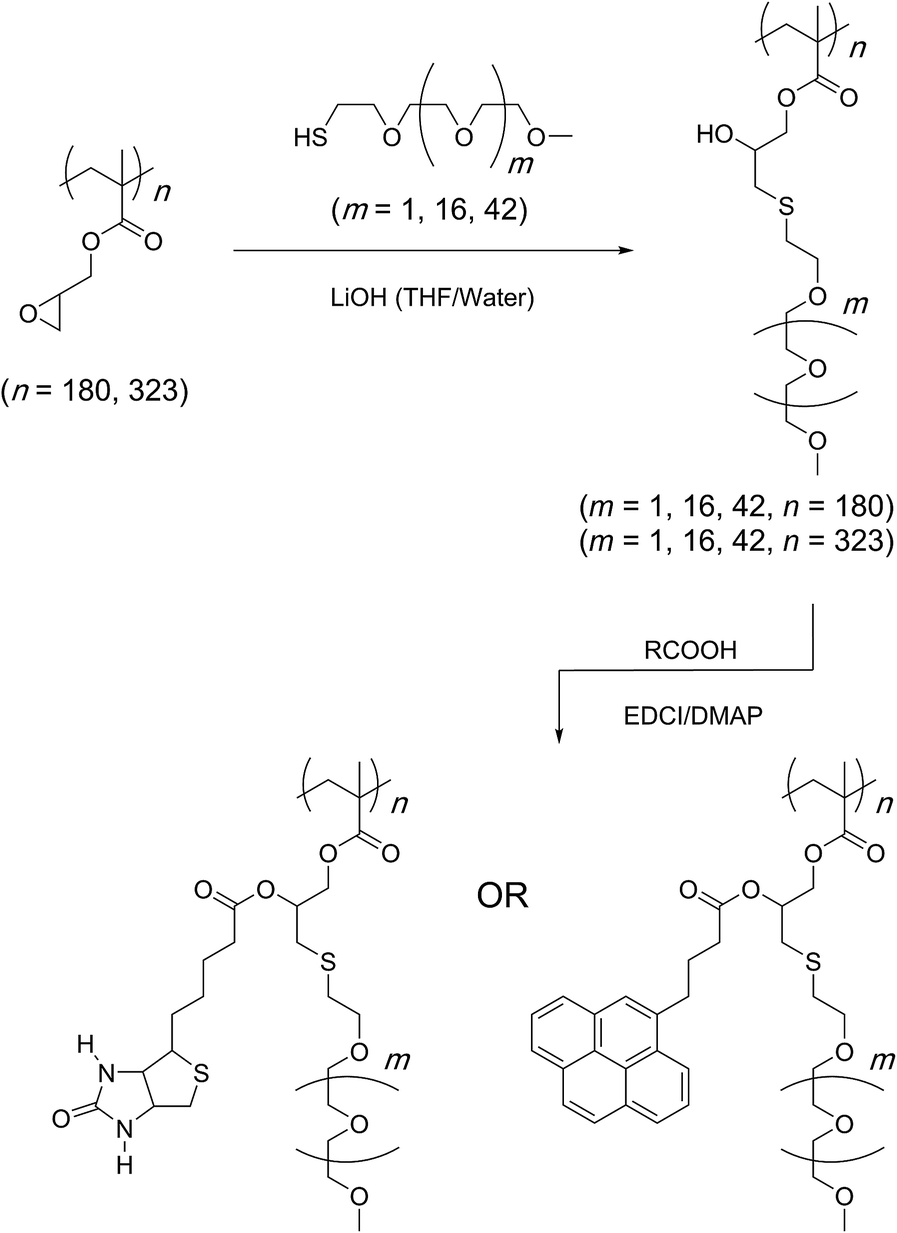
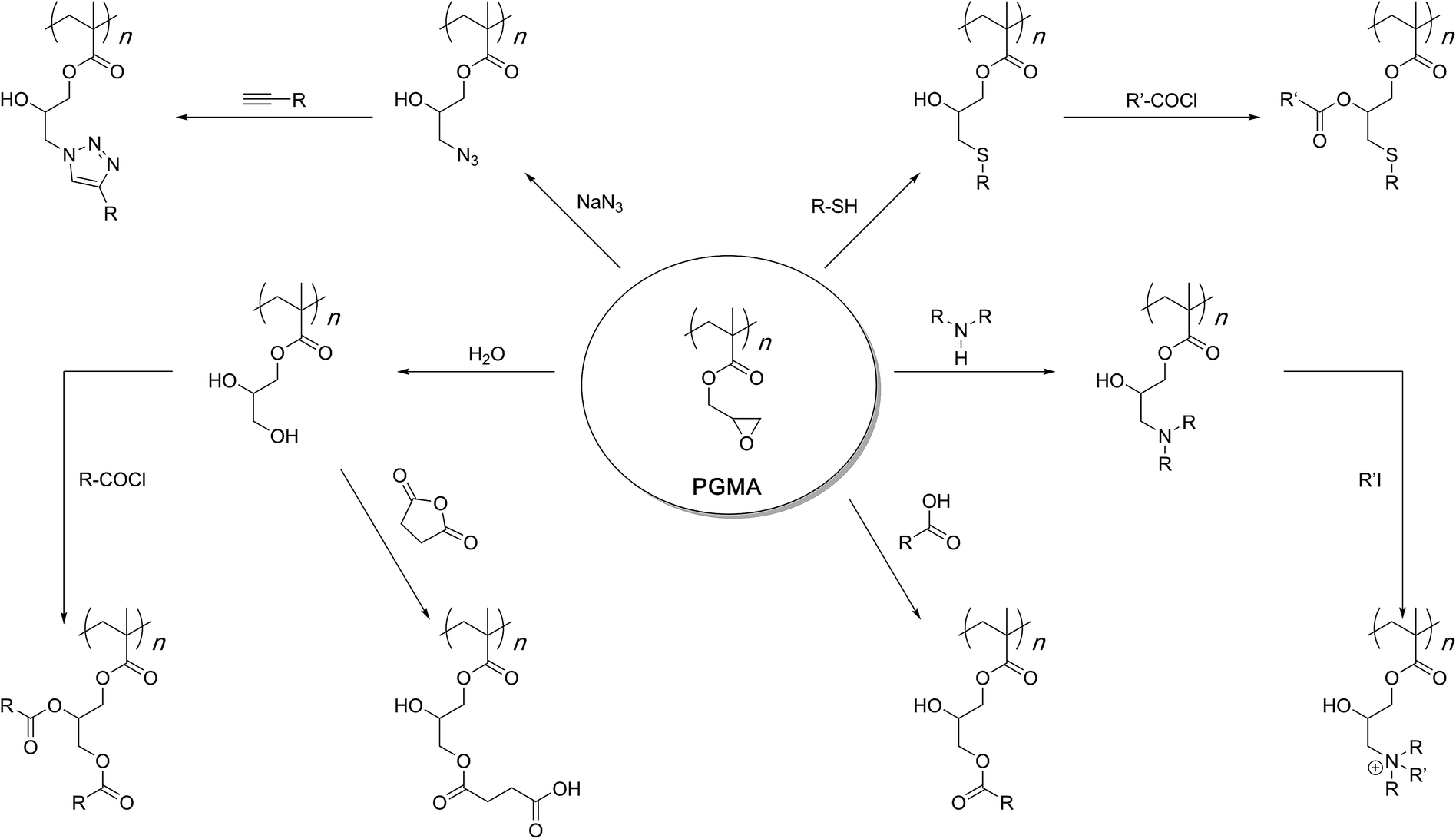
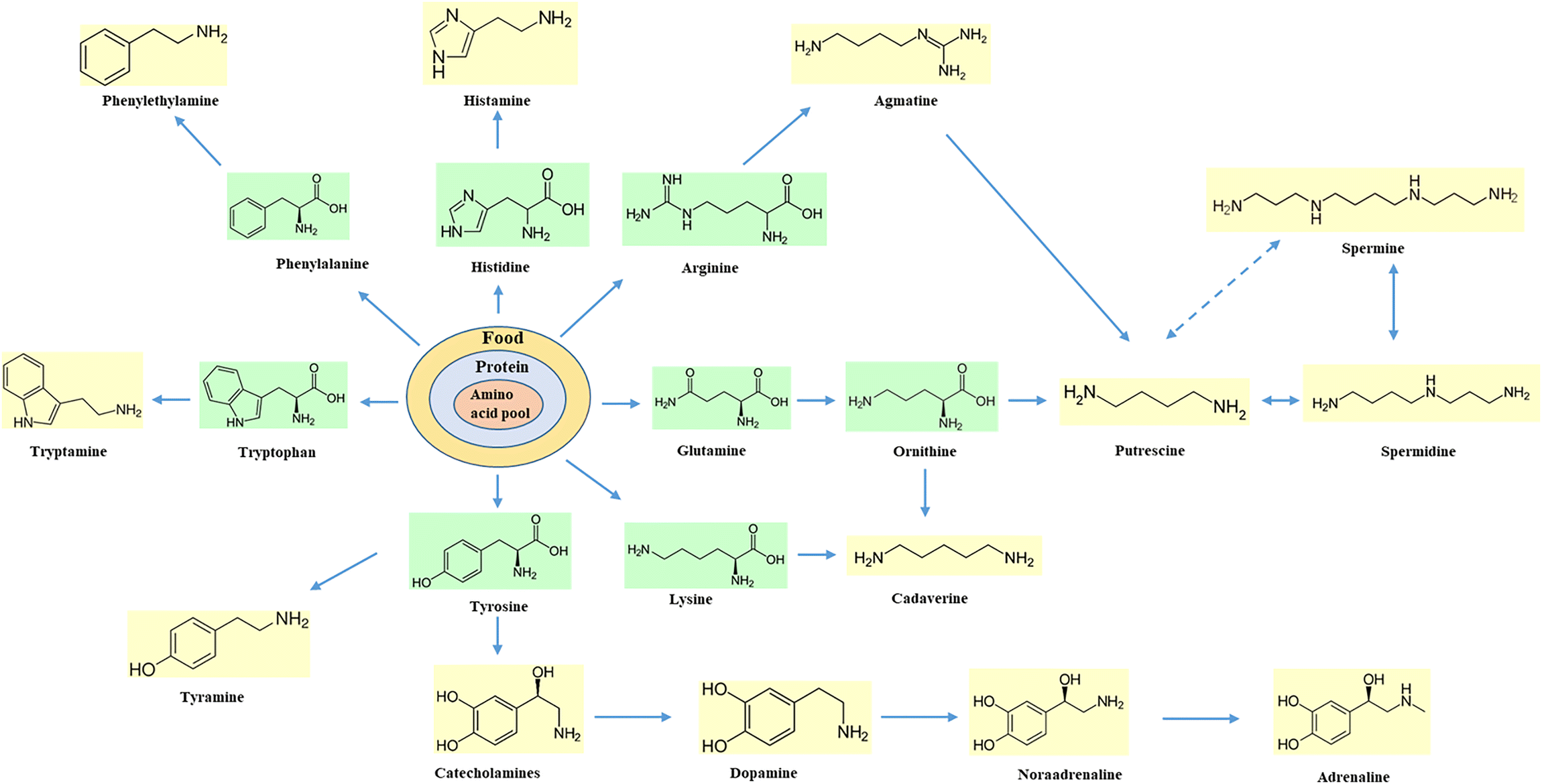
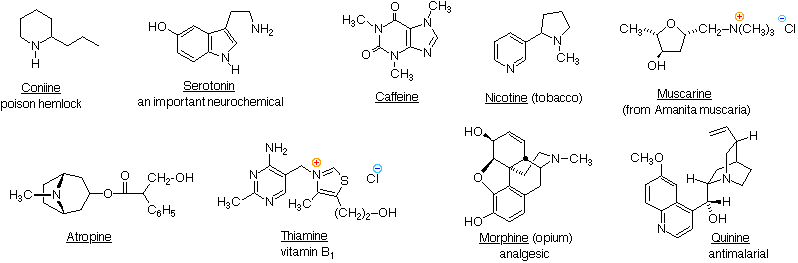
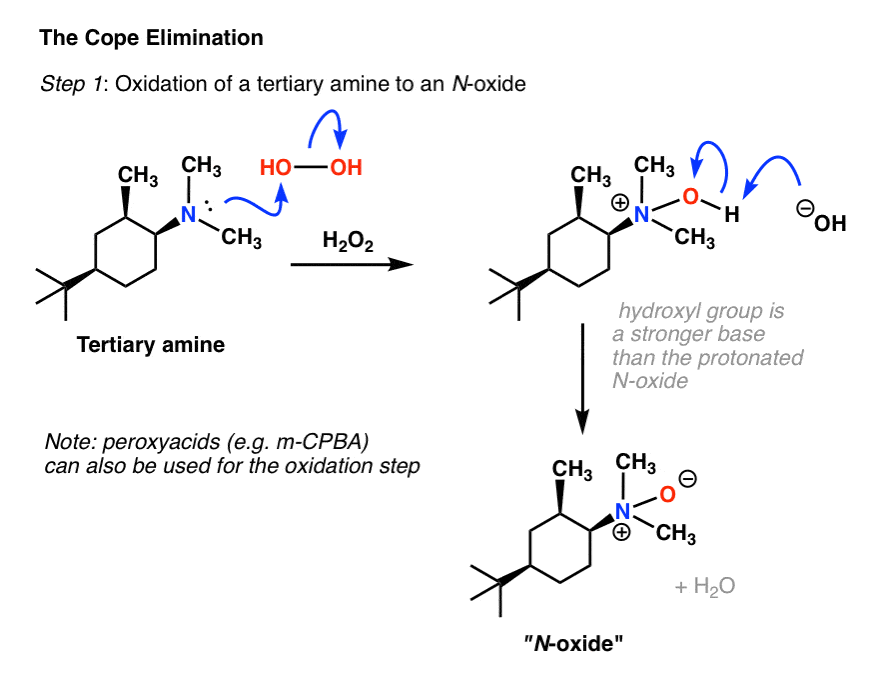
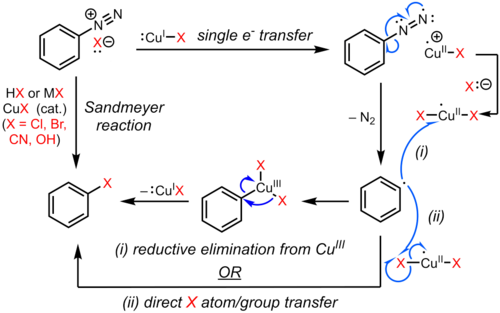
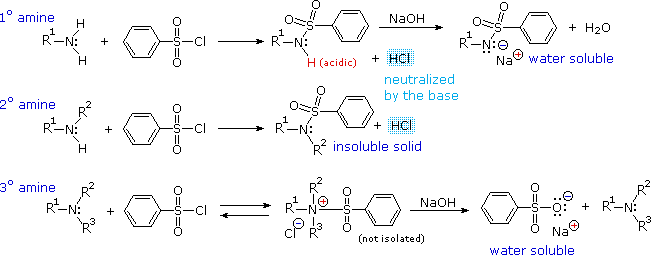
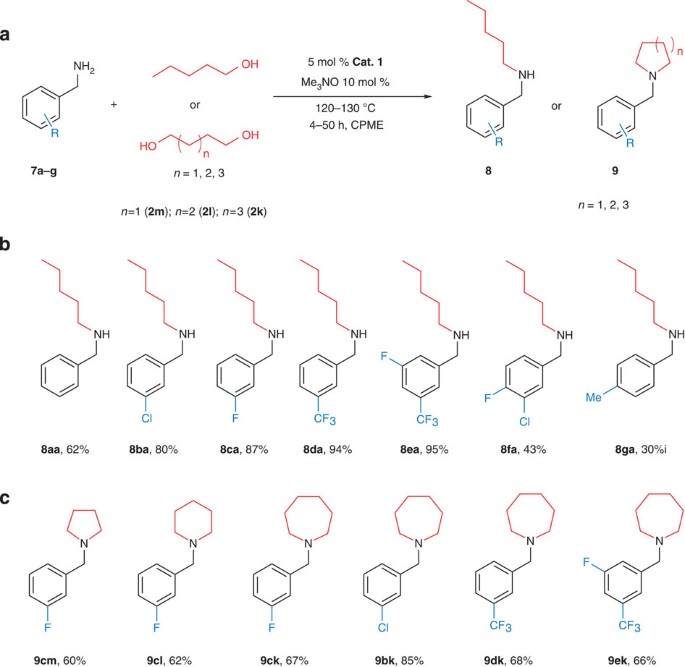
reaction of amine group with water
|
Chapter 6 Amines and Amides
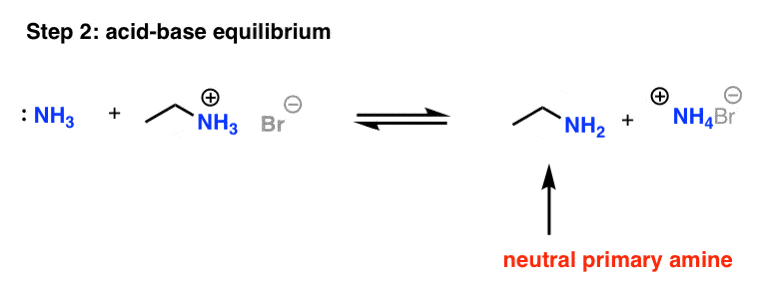
The amine group is located by the position number. Groups that are attached to the Reaction of an amine with water to produce an alkylammonium ion. amine. |
|
Kinetics reaction of primary and secondary amine group in aqueous
Both DETA and water contribute to the carbamate formation in the primary and secondary amine groups. The rate of CO2 absorbed in the primary amine group is |
|
Testsforfunctionalgroups - inorganiccompounds
Organic compounds containing amino group are basic in nature. Thus they easily react with acids to form salts which are soluble in water. Both |
|
Reactions of Amines
Why: Amine D is protonated in acid to give a water soluble ammonium ion. The NaNO2 HCl (converts amine to diazo). 5. CuCN (replaces N2 group with CN). 52 ... |
|
Continuous CO2 Capture in Dual Fluidized Beds Using Silica
water vapour on reaction mechanism between CO2 and various amine groups was discussed. secondary and hindered (containing an amine functional group ... |
|
Distribution and Transport of CO2 in Hydrated Hyperbranched Poly
21 Jan 2021 out in the amine−amine distance because each amine group could be solvated by water molecules. ... the amine groups for reaction. Likewise ... |
|
Control of Nitrosamine Impurities in Human Drugs
nitrosating reaction between amines (secondary tertiary |
|
Assessing the Potential for the Reactions of Epoxides with Amines
18 Sept 2015 for a secondary amine addition product an alcohol group would be formed at the ... the amine−epoxide nucleophilic addition reactions are water-. |
|
N-nitrosamine Impurities in Biological Medicinal Products
11 Nov 2020 ... water) though only the molecule's outer |
|
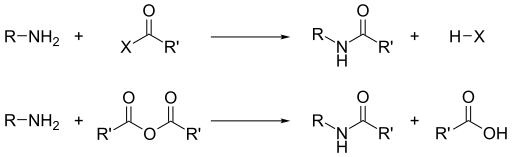
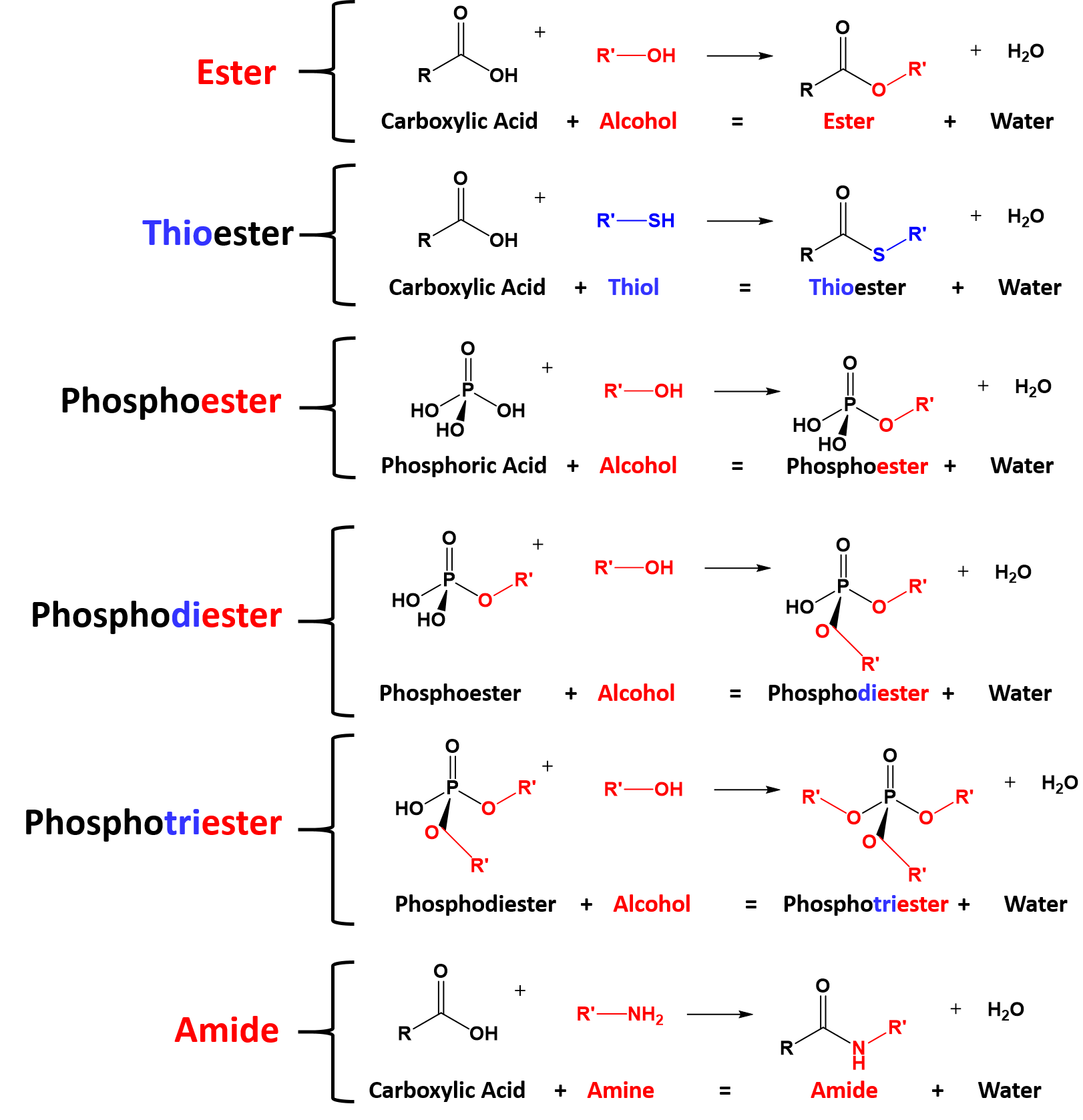
Chapter 5 Carboxylic Acids and Esters
monomers contain two of the same functional group so the reaction can take place at both ends. Esters may be broken apart under acidic conditions by water (a ... |
|
Chapter 6 Amines and Amides
Learn to recognize the amine and amide functional groups. Learn the major chemical reactions of amines and amides and learn how to predict the. |
|
Reactions of Amines
Application in synthesis: The amine (an o/p director) is often derived from a nitro (a meta director). Using the nitro group to direct meta then reducing and |
|
Kinetics reaction of primary and secondary amine group in aqueous
Both DETA and water contribute to the carbamate formation in the primary and secondary amine groups. The rate of CO2 absorbed in the primary amine group is |
|
Classification and Nomenclature of Amines
Learn to recognize the amine and amide functional groups. Learn the major chemical reactions of amines and amides and learn how to predict the. |
|
Chemical Modification of Poly(Vinyl Alcohol) in Water
20-Oct-2015 Amine functional PVA was also prepared by grafting propargylamine using click chemistry reaction. Through this approach a tosyl group (a ... |
|
Functional Group Characteristics and Roles
of acidic and basic functional groups the roles of water and lipid soluble let us focus on the size of the alkyl group on the secondary amine. |
|
Mechanism and kinetics of CO2 adsorption for TEPA- impregnated
The reason is that some amine groups reacted with CO2 at molar ratio of 1:1 in the presence of water vapor according to the reaction stoichiometry |
|
Continuous CO2 Capture in Dual Fluidized Beds Using Silica
water vapour on reaction mechanism between CO2 and various amine groups was Keyworks: CO2 capture ; dual fluidized beds; supported amine; water vapour; ... |
|
Synthesis and Characterization of Lactose-Amines with Respect to
groups (water-fearing group as their "tails") and hydrophilic groups non-polymerized (non brown Maillard reacted) lactose-amines and b) their influence. |
|
Advances in Amine-Surface Functionalization of Inorganic
19-Jan-2022 Adsorbents for Water Treatment and Antimicrobial ... amine group facilitates the attachment of many hazardous pollutants such as negatively. |
|
Ch 06 Amines and Amides - Angelo State University
1° 2° and 3° amines can all form hydrogen bonds with water Low-molecular weight amines are generally water-soluble CH3 O H N H H O H CH3 O H NH CH3 O H CH3 O H CH3 CH3 Physical Properties of Amines: Odor Low molecular-weight amines tend to have sharp penetrating odors similar to ammonia |
|
CHAPTER 7 AMINES - Oregon Institute of Technology
One polar amine group can pull about 4 C atoms of hydrophobic alkyl chain into water CH3CH2CH2NH2 NH2 1 aminopropane 1-aminocyclohexane (cyclohexylamine) very soluble in water not very soluble in water When an amine is converted into an alkylammonium ion (by the addition of acid) the |
|
7: Reactions of Haloalkanes Alcohols and Amines
This chapter describes nucleophilic substitution reactionsof haloalkanes alcohols amines and compounds related to them These are ionic reactions in which one group on the molecule (a leaving group) is replaced by another group (a nucleophile) |
|
Reactions of Amines
Reactions of Amines Reaction as a proton base (Section 19-5 and 19-6) H-X(proton acid)HRN HamineNaOHbase RNHX Hammonium salt(acidic) Mechanism: Required (protonation) Reverse Mechanism: Required (deprotonation) Amines are completely converted to ammonium salts by acids Ammonium salts are completely neutralized back to amines by bases |
|
Chemical Reactions of Amines
names of the alkyl group when two or more identical groups are present If more than one amino group is present in the amine then the parent chain and the position of amino groups is identified by numbering the carbon atoms in the parent chain The numbering is done in such a way that the carbon atom bearing the |
|
Searches related to reaction of amine group with water filetype:pdf
Mar 9 2015 · Amine-reactive reagents react with non-protonated aliphatic amine groups including the amine terminus of proteins and the ?-amino group of lysines The ?-amino group has a pK of around 10 5; in order to maintain this amine group in the non-protonated form the conjugation must take place in a buffer with slightly basic pH |
What happens when an amine is converted into an alkylammoniumion?
- not very soluble in water When an amine is converted into an alkylammoniumion (by the addition of acid) the ionic ammonium ion is much more effective at pulling non-polar groups(up to ~15-18 C atoms) into aqueous solution, just as was the case for carboxylate ions. They do this by forming micelles, just like the carboxylate ions.
What are amines and alkaloids?
- Many amines are found in natural products and many of them are biologically active when ingested or injected. There has been a long-standing interest in these compounds and they are collectively referred to as alkaloids,because of the basic (alkaline) properties resulting from the amine group.
How do you know if an amine is secondary or tertiary?
- If no gas was evolved but an insoluble yellow or orange liquid separated from the solution, then this is indicative of a secondary aliphatic or aromatic amine. If no gas was evolved and no yellow liquid separated then this is indicative that no reaction occurred and the amine tested is a tertiary amine.
Do amines have lower boiling points than alcohols?
- 1° and 2° amines have lower boiling points than alcohols of similar molecular weight. 3° amines, since they do not hydrogen bond to each other, have boiling points similar to hydrocarbons of the same molecular weight. Physical Properties of Amines: Boiling Points
|
Reactions of Amines
Amines 1 Reactions of Amines 1 Reaction as a proton base (Section 19-5 and 19-6) R N H H N NZ Notes: • “Z” can be a carbon, nitrogen, oxygen, or hydrogen atom/group Water drives to the carbonyl side; removal of water drives to |
|
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
by designating the organic groups separately in front of the word amine water • Tertiary amines have lower boiling points than primary or secondary amines of |
|
Amines Amines - NCERT
secondary and tertiary amines, when two or more groups are the same, the prefix di or tri is Lower aliphatic amines are soluble in water because they can form |
|
Chapter 17: Amines and Amides
Secondary amines (2o): Nitrogen with two R groups • Tertiary amines Even tertiary amines are water-soluble because of its ability to form hydrogen bonds |
|
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
For this reason, primary and secondary amines have higher boiling points than tertiary amines of similar size hydroxide ion The general form of the reaction of an amine with water is shown below The lone pair on the amine nitrogen forms a bond to the H+ from water |
|
Kinetics reaction of primary and secondary amine group in - CORE
Both the termolecular and the zwitterion mechanisms were applied to interpret the experimental data and gave identical result Both DETA and water contribute to |
|
Amine
Primary, secondary and tertiary amines have 1, 2 or 3 alkyl groups attached to nitrogen In these Amines are soluble in water if they have 1 to 5 carbon atoms; |
|
AMINES
29 mar 2020 · groups in primary and secondary amines decreases from tetrahedral it decreases its ability to form hydrogen bond with water molecules |
|
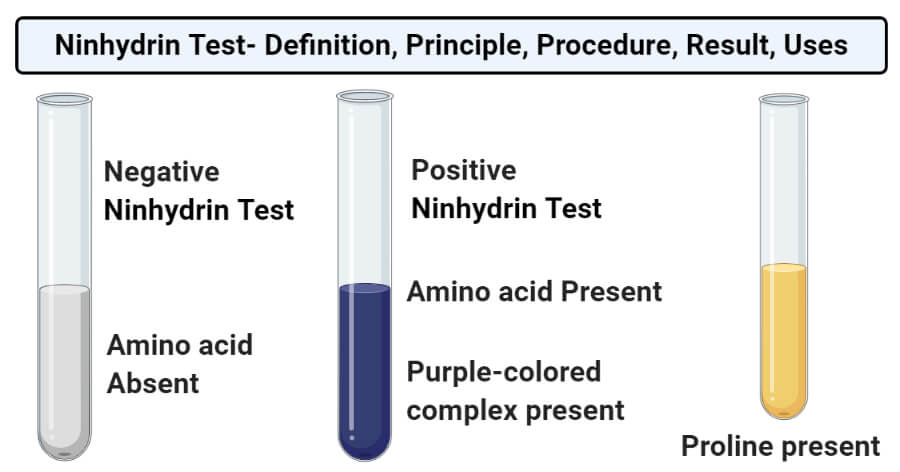
Experiment 16 Qualitative Analysis of Amines and Amine Unknown
Test in order to discriminate between primary, secondary and tertiary amines groups and aryl groups withdraw electrons and decrease the strength of the base product therefore is not soluble in water and furthermore it will not be soluble |