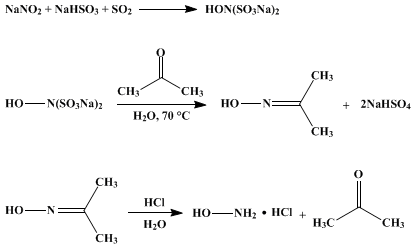
reaction of hydroxylamine with water
|
A CHEMICAL REACTION OF HYDROXYLAMINE WITH
Also for the above reason |
|
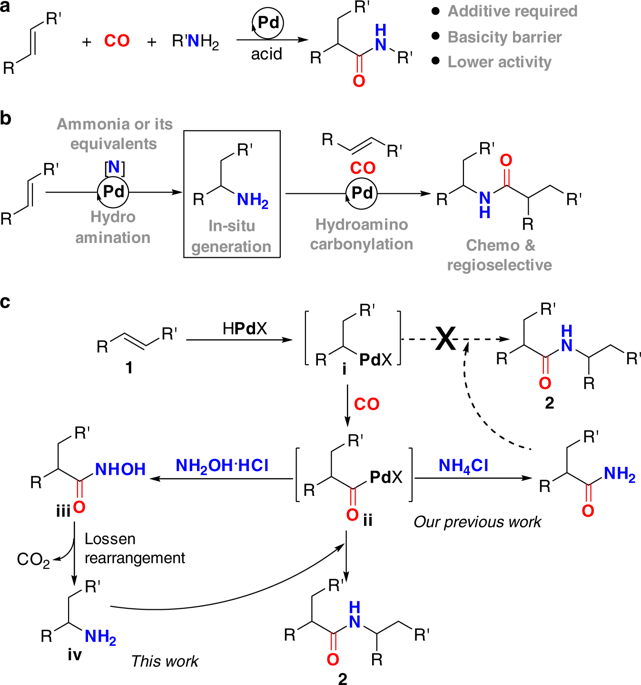
THE REACTION OF ACETYLCHOLINE AND OTHER CARBOXYLIC
The present method which is designed for use with short chain 0-acyl derivatives |
|
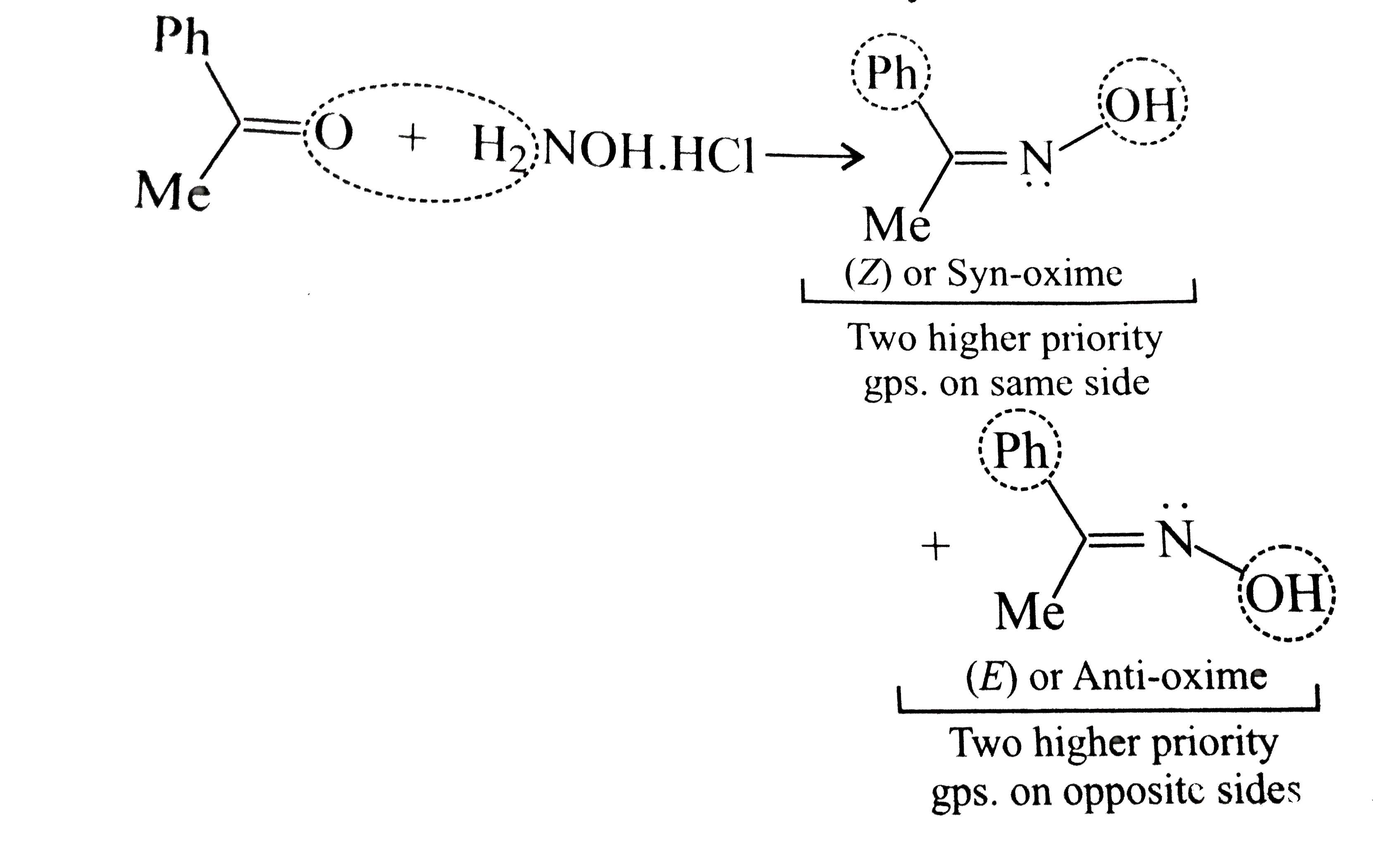
The Reactions of Hydroxylamine-O-sulfonic Acids with Water and
iodide to form iodine and ammonia. Alkyl groups on the nitrogen greatly retard the reaction. Hydrolysis to hydroxylamine and sulfate is first order |
|
Evidence for a chemical reaction of hydroxylamine with the
This indicates a chemical reaction of hydroxylamine with the water-splitting enzyme in the dark. The consequences for the possible states of manganese (as |
|
Kinetics of nitrous oxide production from hydroxylamine oxidation by
such reaction is hydroxylamine (NH2OH) oxidation coupled to the re- interlayer region of birnessite and replace water molecules leading to. |
|
DECOMPOSITION PRODUCTS OF 50 MASS% HYDROXYLAMINE
hydroxylamine=water solution were measured for samples subsequent to a thermally induced runaway reaction. The noncondensable gas phase composition was |
|
Hydroxylamine Nitrate Decomposition under Non-radiological
Refill the apparatus with water for storage. The chemistry of the reactions involving hydroxylamine nitrate relevant to plutonium oxidation have been. |
|
INSTRUCTIONS - NHS and Sulfo-NHS
on water-content and pH of the reaction solution. (Optional): Quench reaction by adding hydroxylamine to a final concentration of 10mM. |
|
Concerning the Mechanism of the Hydrolysis and Aminolysis of
reaction rates for the reaction with water for which attack of the dehyde semicarbazones with hydroxylamine and the reaction of a series of substituted ... |
|
The Reaction of Hydroxylamine with Activated Acyl Groups. I
the reaction of hydroxylamine with acylating agents have been N-substituted hydroxylamines (hydrox- amic acids)5 with the apparent single exception of. |
|
INSTRUCTIONS Hydroxylamine•HCl - Thermo Fisher Scientific
Hydroxylamine converts carbonyl compounds (aldehydes and ketones) to their oxime derivatives in the presence of a weak base Therefore crosslinkers and other compounds that contain a carbonyl within their structure are cleavable with hydroxylamine•HCl |
|
Amine-Reactive Probes - Thermo Fisher Scientific
Abstract—The reaction kinetics of NN-diethyl hydroxylamine (DEHA) and free oxygen in distilled water was studied The kinetic model of chemical reaction was created based on experimental work by measuring dif-ferent pH values reaction temperature DEHA dosage and the removal rate of free oxygen in water The |
|
Amine-Reactive Probes - Thermo Fisher Scientific
Mar 9 2015 · hydroxylamine-containing reaction overnight at 4°C Note: Treatment with hydroxylamine at this stage is required to remove the trifluoroacetyl protecting groups of Rhodamine Green™ (Cat no R6112) succinimidyl ester Hydroxylamine may also remove dye from unstable conjugates with tyrosine serine threonine and histidine |
|
Experiment 10 - University of Idaho
Phenanthroline solution: Dissolve 100 mg 110-phenanthroline monohydrate in 100 mL water Add two drops conc HCl to your solution Stir to dissolve 2 Preparing your iron top standards—these do need to be prepared using volumetric you will do the spectrophotometric analyses |
How do you dissolve hydroxylamine hydrochloride?
- You may use 1.5 M hydroxylamine, pH 8.5, to terminate the reaction and to remove weakly bound probes. To prepare this reagen t, dissolve hydroxylamine hydrochloride at 210 mg/mL in distilled water and adjust the pH to 8.5 with 5 M NaOH. Dilute the resul ting 3 M solution with an equal volume of distilled water to get 1.5 M hydroxylamine, pH 8.5.
Why do you add hydroxylamine to a water sample?
- You will add the reducing agent, hydroxylamine, to your water sample 2+and standards in order to reduce Fe3+to Fe . Because dissolved oxygen in water can reoxidize Fe2+3+to Fe , an excess amount of the hydroxylamine reducing agent is needed to 2+keep iron in the +2 state during the course of the experiment.
Is hydroxylamine soluble in water?
- Hydroxylamine•HCl is more stable to oxidation than the free base form of hydroxylamine; nevertheless, always prepare hydroxylamine solutions immediately before use and store the product desiccated. Hydroxylamine•HCl is soluble in polar solvents such as water, ethanol, methanol, glycerol and propylene glycol.
How do you make hydroxylamine reagen t?
- To prepare this reagen t, dissolve hydroxylamine hydrochloride at 210 mg/mL in distilled water and adjust the pH to 8.5 with 5 M NaOH. Dilute the resul ting 3 M solution with an equal volume of distilled water to get 1.5 M hydroxylamine, pH 8.5. Prepare this reagent freshly before use (see step 1.5, page 3).
|
Hydroxylamine·HCl - G-Biosciences
Hydroxylamine·HCl is soluble in polar solvents such as water, ethanol, Immediately before use, prepare a 0 5M solution of hydroxylamine in the Reaction |
|
Evidence for a chemical reaction of hydroxylamine with the
After treatment with low concentrations of hydroxylamine the corresponding flash patterns showed a delay of two S-state transitions of the water-splitting enzyme S |
|
DECOMPOSITION PRODUCTS OF 50 MASS% HYDROXYLAMINE
hydroxylamine=water solution were measured for samples subsequent to a thermally induced runaway reaction The noncondensable gas phase composition |
|
Mechanism of the Redox Reaction of Hydroxylamine and Bromate
Hydroxylammonium chloride, the oxidant and the other solutions were prepared with distilled water The rate of reactions of the oxidant (BrO3 – ) and the |
|
Hydroxylamine•HCl - Thermo Fisher Scientific
group (-SH) for the final conjugation reaction, hydroxylamine is used to remove the solvents such as water, ethanol, methanol, glycerol and propylene glycol |
|
In the presence of hydroxylamine The reagent reacts with iron, after
702 g of reagent grade ferrous ammonium sulfate hexahydrate (Fe(NH4)2(SO4)2 •6H2O) in 50 mL of purified water, add 20 mL of 1:3 sulfuric acid and dilute to 1 L |
|
Product Class 5: Hydroxylamines - thieme-connectde
from the appropriate 9-hydroxythiaxanthenes by reaction with hydroxylamine hydro- dride in water at pH 5 can be used to produce various α-(hydroxyamino) |















![PDF) [Chem 28] Spectrophotometric Determination of Iron in Aqueous PDF) [Chem 28] Spectrophotometric Determination of Iron in Aqueous](https://upload.wikimedia.org/wikipedia/commons/thumb/6/6c/Hydroxylamine-dimensions-2D.png/242px-Hydroxylamine-dimensions-2D.png)