reaction without metal ligand bond cleavage pdf
|
BSc Chemistry
Reaction without metal ligand bond cleavage. The substitution reactions take place without metal-ligand bond cleavage is the reactions where metal-ligand |
|
Metal-Ligand Bonding and Inorganic Reaction Mechanisms Year 2
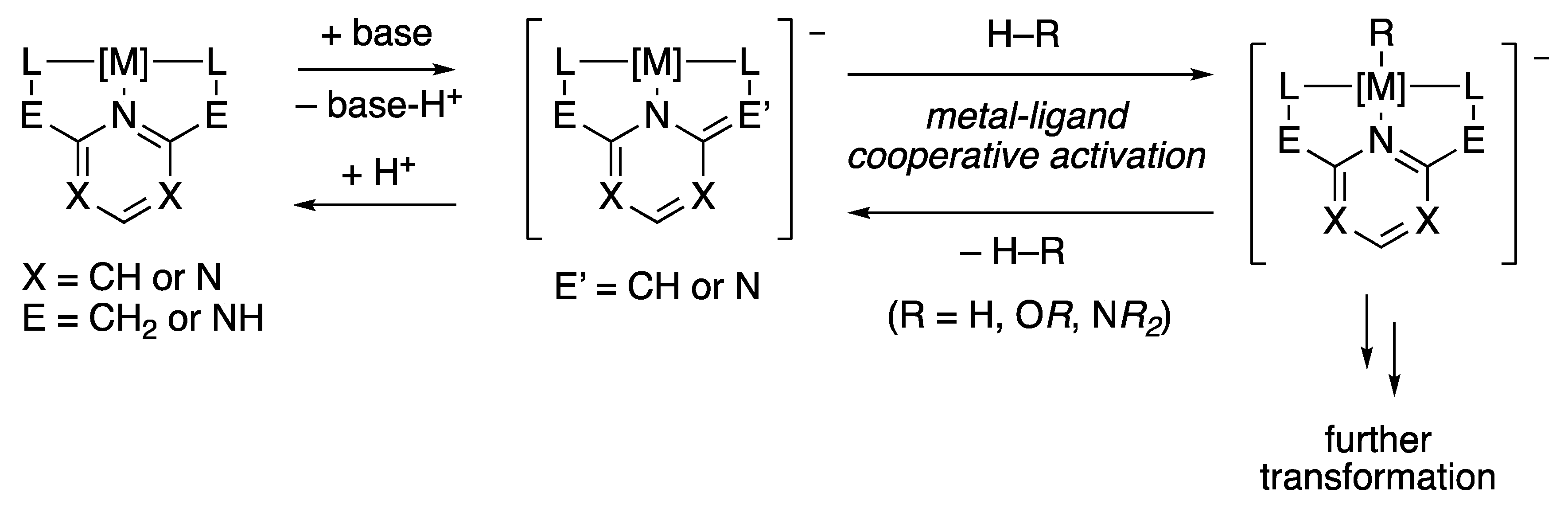
v) Reactions of coordinated ligands. Reactions of a ligand that take place without breaking the M-L bond. + Lots of organometallic and organic chemistry using |
|
Reaction Mechanism of Transition Metal Complexes – I
Hence in both of the reactions given above |
|
Metal-Ligand and Metal-Metal Bonding Core Module 4 RED
orbitals are non-bonding and completely metal based. The 2eg orbitals are σ of O2 occupying 1πg and 3σu of O2 causing cleavage of the O2 bond. Why is η1 ... |
|
Know More
reactions without metal ligand bond cleavage. ▫ Huheey J.E.; Keiter |
|
CSJM UNIVERSITY KANPUR - M.Sc. (Previous) - CHEMISTRY
mechanism anation reactions |
|
Photochemical Heterolysis of the Metal-Metal Bond in (Me3P)(OC
lished in reactions not involving metal-metal bonds. For example metal-ligand bond cleavage reactions from dd excited states are typically heterolytic.8 |
|
Substitution Reaction in Metal Complexes Dr. Jiya Lal Maurya M.Sc
Cleavage of the M-X bond. (fast). ML5XY. ML5Y + X. (fast). The rate law for The rate of reaction correlates with the metal-ligand bond strength of the leaving. |
|
Nickel: The “Spirited Horse” of Transition Metal Catalysis
17-Feb-2015 The cleavage of the metal−ligand bond is an important mechanistic ... Without a suitable ligand many Ni-mediated transformation are likely ... |
|
Reaction mechanisms and thermochemistry of vanadium ions with
Symmetric C-C bond cleavage process10 |
|
BSc Chemistry
Transition Metal Complexes). Module No. 25: Acid and Base hydrolysis Reactions without metal ligand bond cleavage. Subject. Chemistry. Paper No and Title. |
|
Metal-Ligand Bonding and Inorganic Reaction Mechanisms Year 2
Metal-ligand and metal-metal bonding of the transition metal elements. Synopsis Reactions of a ligand that take place without breaking the M-L bond. |
|
SCHEME OF EXAMINATION FOR M
Reaction Mechanism of Transition Metal Complexes: Energy profile of a reaction without metal ligand bond cleavage. Substitution reactions in square ... |
|
Mechanisms of Substitution Reactions of Metal Complexes
A. Classification of Reaction Mechanisms. B. Octahedral Complexes. C. Square Planar Complexes. D. Reactions without Metal-Ligand Atom Bond Cleavage. |
|
Reaction Mechanism of Transition Metal Complexes – I
Octahedral complexes react either by SN1 or SN2 mechanism in which the intermediates are five and complexes occurs without metal-ligand bond breaking. |
|
Know More
24: Base hydrolysis-conjugate base mechanism direct and indirect evidences in favour of conjugate mechanism |
|
Untitled
reactions without metal ligand bond cleavage. Substitution reaction in square planer complexes the trans effect |
|
Substitution Reaction in Metal Complexes Dr. Jiya Lal Maurya M.Sc
Cleavage of the M-X bond. Mechanisms of ligand exchange reactions ... The rate of reaction correlates with the metal-ligand bond strength of the leaving. |
|
M.Sc.Chemistry (CBCS) Program M1CHE01-CT01 Inorganic
Energy profile of a reaction reactivity of metal complexes Anation reactions |
|
Subject Chemistry Ligand Equilibria and Reaction Mechanism of
The reactions without metal ligand bond cleavage are a type of decarboxylation reactions The base hydrolysis of complexes follow millions times faster than acid hydrolysis Due to dependency of base hydrolysis on HO- ion the mechanism follows SN1(CB) mechanism |
|
Know More - epgpinflibnetacin
reactions without metal ligand bond cleavage Glossary A Associative Nucleophilic Substitution reaction: Nucleophilic substitution reaction in which ligand first associate with the metal ion before breaking of metal- leaving ligand bond B Base Hydrolysis Hydrolysis of octahedral complexes in the presence of hydroxyl (HO-) ions ions is |
|
MASTER OF SCIENCE (MSc) CHEMISTRY SEMESTER I - RKDF University
Stepwise and overall formation constants and their interaction trends in stepwise constant factors affecting the stability of metal complexes with reference to the nature of metal ion and ligand Chelate effect and its thermodynamic origin determination of binary formation constants by potentiometry and spectrophotometry |
What is the mechanism for cleaving the C–C bond?
- In this regard, the mechanism proposed for cleaving the C–C bond comprises a series of C–H bond cleavage reactions to access a benzyne-type intermediate [ h2 - C2-C6H4(NCCN)]W (PMe3)4H2, from which [k2 - C2-C6H4(NC)2]W (PMe3)4 is obtained via a sequence that involves reductive elimination of H2 and C–C bond cleavage.
What is the key to the carbon–nitrogen triple-bond cleavage?
- The DFT calculations also revealed the detailed reaction mechanism and indicated that the key for the carbon–nitrogen triple-bond cleavage could be attributed to the presence of nucleophilic nitrogen atom in one of the intermediates. A triple bond having three shared electron pairs between two atoms is known as one of the strongest chemical bonds.
Is cleavage of a triple bond possible without transition metals?
- Recently, chemistry of single-bond cleavage by using main-group element compounds is rapidly being developed in the absence of transition metals. However, the cleavage of a triple bond using non-transition-metal compounds is less explored.
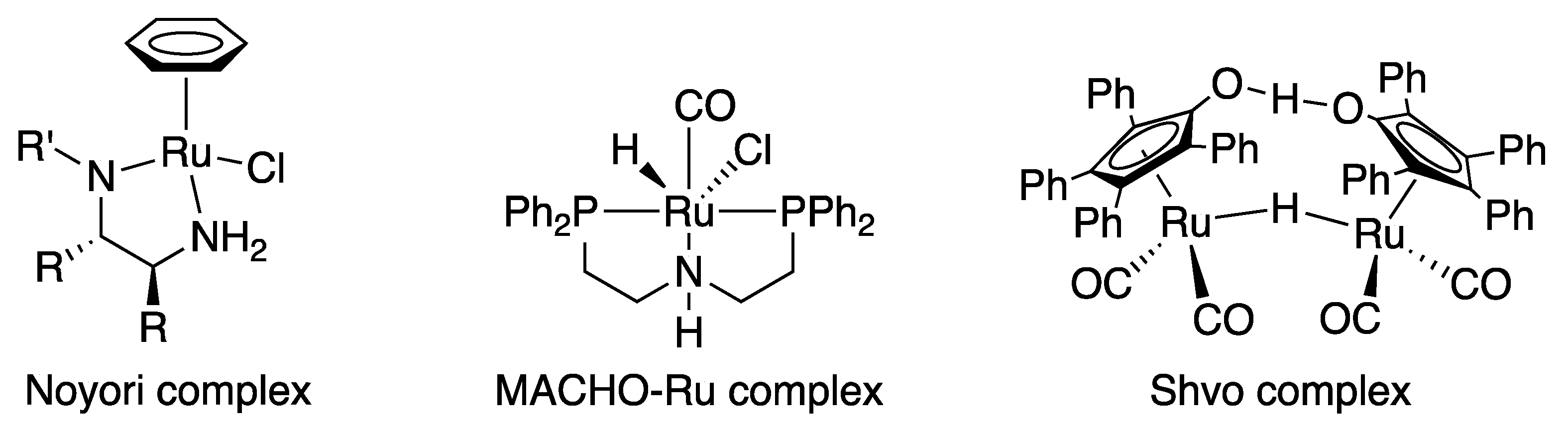
What is the effect of metal to ligand bonding?
- The metal to ligand bonding creates a synergic effect which strengthens the bond between CO and the metal . SOLVED QUESTIONS 1 MARK QUESTIONS 1. What are ambidentate ligands?
|
BSc Chemistry - e-PG Pathshala
Transition Metal Complexes) Module No 25: Acid and Base hydrolysis, Reactions without metal ligand bond cleavage Subject Chemistry Paper No and Title |
|
Metal-Ligand bonding and Inorganic reaction mechanisms
Octahedral ML6 molecular orbital energy diagram Lecture 3: π-acceptor ligands and synergic bonding Binding of CO, CN-, N2, O2 and NO |
|
Reaction Mechanism of Transition Metal Complexes – I - Dalal Institute
The metal complexes in which the rate of ligand displacement reactions is very fast and hence show complexes occurs without metal-ligand bond breaking member benefits like downloading any PDF document for your personal preview |
|
M Sc CHEMISTRY – I SEM EXAM - 2017pdf - Jiwaji University
reactions without metal ligand bond cleavage Substitution reaction in square planer complexes, the trans effect, Mechanism of substitution reactions A Lhawal |
|
MSc (CHEMISTRY) I SEM - EXAM 2014pdf - Jiwaji University
anion reactions, reactions without metal ligand bond cleavage Substitution reactions Reaction Mechanism of Transition Metal Complexes-ll and HSAB theory |
|
Ligand Substitution - CORE
2 mai 2006 · The subject of the mechanistic study of ligand substitution reactions is currently metal complexes should provide sufficient background Thus, we expected, since in this case no Pt-OH2 bond breaking is necessary, just a |
|
CH307 Inorganic Kinetics - NUI Galway
Metal complexes that undergo substitution reactions with t 1/2 is concurrent with bond cleavage between the metal and the leaving process where there is no intermediate When a dissociative mechanism is operative, the rate of ligand |
|
Reaction kinetics and mechanism - MIDNAPORE COLLEGE
sociates from the metal center and the vacancy in the coordination sphere is taken by the new intermediates in the reaction path that leads to ligand substitution, then bond making is more important than bond breaking and no detectable |
|
Mechanisms of Substitution Reactions of Metal - ScienceDirectcom
A Classification of Reaction Mechanisms B Octahedral Complexes C Square Planar Complexes D Reactions without Metal-Ligand Atom Bond Cleavage |
|
Reaction Mechanism (Inorganic Chemistry) - Jaipuria College
entering nucleophile (i e entering ligand) makes a bond with the metal centre in while interchange reaction is a one-step reaction without the formation of a true complex, both the bond breaking by the leaving group (i e slight rate |















![Metal–Ligand Cooperativity of the Calix[4]pyrrolato Aluminate Metal–Ligand Cooperativity of the Calix[4]pyrrolato Aluminate](https://i1.rgstatic.net/publication/331852167_Effect_of_Metal_Catalysts_on_Bond_Cleavage_Reactions_of_Lignin_Model_Compounds_in_Supercritical_Water/links/5fc0bedc92851c933f659cd0/largepreview.png)