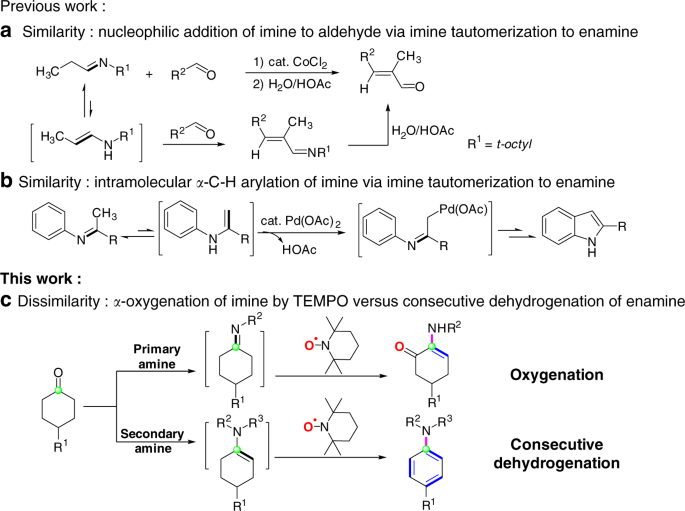
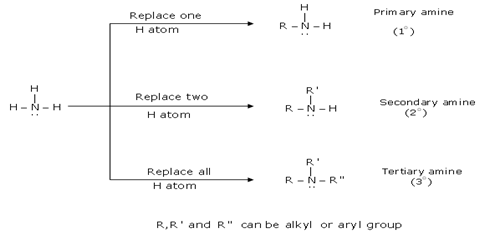
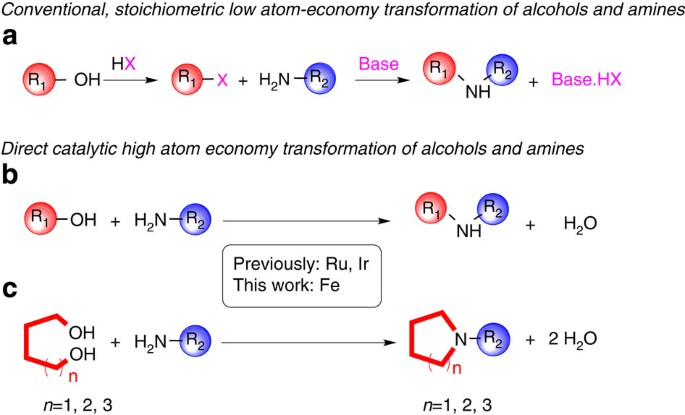
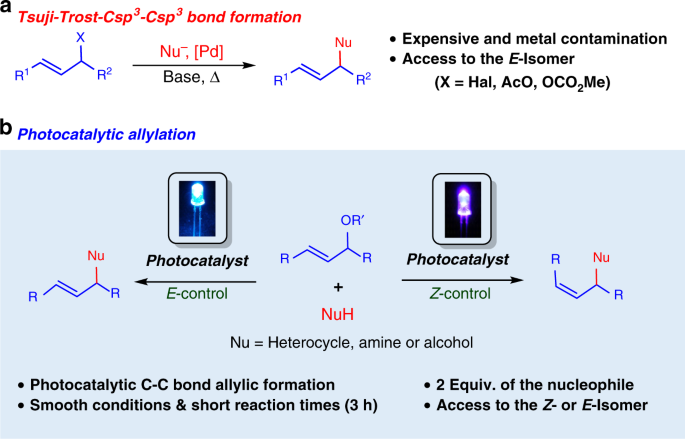
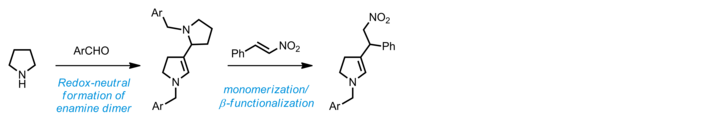
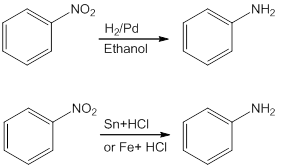
reactivity of amines
|
The Relative Reactivity of Amines in the Aminolysis of Amides1
Relative Reactivity of Amines in the Aminolysis of Amides. 657. Butylene with Benzene.—One mole of benzene and 8.4 g. of dihydroxyfluoboric acid were. |
|
A perspective approach of the influence of H bonds in epoxy
First the most reactive class of amine hardeners are aliphatic amines containing heteroatoms. The reactivity of amine hardeners could be indeed enhanced by the |
|
Concerning the reactivity of amines toward carbonium ions derived
Concerning the Reactivity of Amines toward Carbonium Ions of the carboniumion derived from ortho ester between amine generating an amine adduct as ... |
|
Unexpected Reactivity of N-Acyl-Benzotriazoles with Aromatic
18 déc. 2020 Unexpected Reactivity of N-Acyl-Benzotriazoles with Aromatic Amines in Acidic Medium (ABAA Reaction). European Journal of Organic Chemistry. |
|
Relative reactivity of amines and oxyanions toward proton
6.2 is 130-fold more reactive than an oxyanion with a conjugate acid of the same pAa. A comparison of the reactivity of tertiary amines and oxyanions. |
|
Reactions of phosphonic acid esters with nucleophiles. III. Reactivity
Abstract: Therate constants for the reactions of a series of amines with -nitrophenyl methylphosphonate mono- anion O-NPMP) in water have been determined. |
| Reactivity of Amines toward a Neutral Platinum (II) Complex |
| Reactivity of Amines toward a Neutral Platinum (II) Complex |
|
Chemical reactivity of graphene oxide towards amines elucidated by
29 avr. 2020 This study points out the absence of reactive COOH groups thus eliminating the possibility of the amidation reac- tion with amine derivatives |
|
ArC™ Amine Reactive Compensation Bead Kit
6 avr. 2009 The reactive dye can enter the compromised membranes of necrotic cells and react with free amines in the interior and on the surface of the cell ... |
|
Ch 06 Amines and Amides - Angelo State University
Amines and amides are abundant in nature They are a major component of proteins and enzymes nucleic acids alkaloid drugs etc (Alkaloids are N-containing weakly basic organic compounds; thousands of these substances are known ) Aminesare organic derivatives of ammonia NH3 in which one or more of the three H’s is replaced by a carbon group |
|
Amine-Reactive Probes - Thermo Fisher Scientific
Basicity-Aliphatic Amines CH3NH2 NH3 CH3CH2 NH2 C6H11NH2 (CH3)2NH (CH3CH2)2NH (CH3)3N (CH3CH2)3N Tertiary Amines diethylamine dimethylamine cyclohexylamine ethylamine methylamine Secondary Amines Primary Amines Ammonia Amine Structure pKa trimethylamine triethylamine 9 26 10 64 10 81 10 66 10 73 10 98 9 81 10 75 pKb 4 74 3 36 3 19 3 34 3 27 3 |
|
Chemical Reactions of Amines
Preparation of Amines Classification of Amines On the basis of the presence of replaceable hydrogen atoms we can classify amines into three types: Primary Amines: The amine is primary amine when an alkyl or Aryl group replaces one of three h ydrogen atoms Secondary Amines: It is a secondary amine when the alkyl or |
|
Reactions of Amine- Reductive Amination - Department of Chemistry
or*enamines*canbereducedto2°*or3°*aminesrespectively Onewayishydrogen addition*using*a*metal*catalyst *** O O H H2N OH N H OCH3 H H OCH3 H H OH2 N H OH N H HHOCH 3 |
|
Searches related to reactivity of amines filetype:pdf
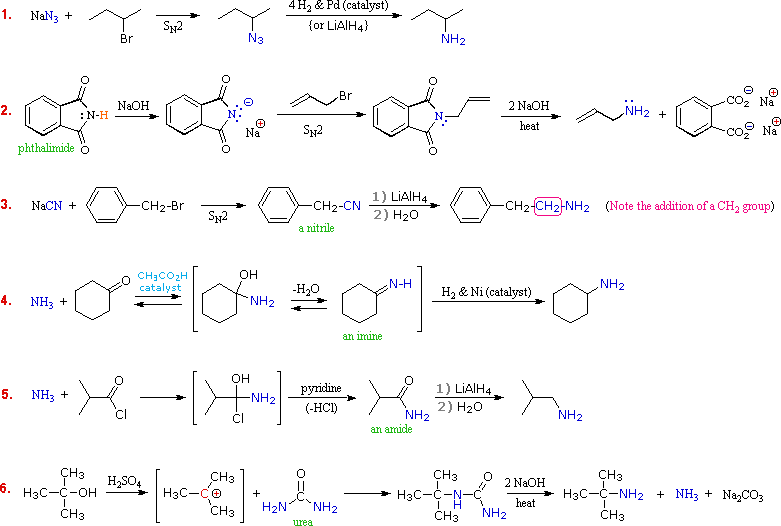
The order of reactivity of halides with amines is RI > RBr >RCl Write chemical equations for the following reactions: Example 13 1 Reaction of ethanolic NH with C 32H5Cl Ammonolysis of benzyl chloride and reaction of amine so formedwith two moles of CH3Cl Solution 3 Reduction of nitriles |
What reagents are used to label amines?
- There are three major classes of commonly used reagents to label amines: active esters (which include succinimidyl esters (SE), sulfosuccini midyl esters (SSE), tetrafluorophenyl esters (TFP), and sulfodichlorophenol esters (SDP)), isothiocyanates (ITC), and sulfonyl chlorides (SC).
How do amines react with acids?
- Amines, being basic in nature, react with acids to form salts. Amine salts on treatment with a base like NaOH, regenerate theparent amine. Amine salts are soluble in water but insoluble in organic solventslike ether. This reaction is the basis for the separation of amines fromthe non basic organic compounds insoluble in water.
How do amines undergo alkylation?
- Amines undergo alkylation on reaction with alkyl halides (refer Unit10, Class XII). Aliphatic and aromatic primary and secondary amines react withacid chlorides, anhydrides and esters by nucleophilic substitutionreaction. This reaction is known as acylation. You can considerthis reaction as the replacement of hydrogen atom of –NH or >N–H
What is an example of an amine?
- Amines constitute an important class of organiccompounds derived by replacing one or more hydrogenatoms of ammonia molecule by alkyl/aryl group(s). Innature, they occur among proteins, vitamins, alkaloidsand hormones. Synthetic examples include polymers,dyestuffs and drugs.
|
Reactions of Amines
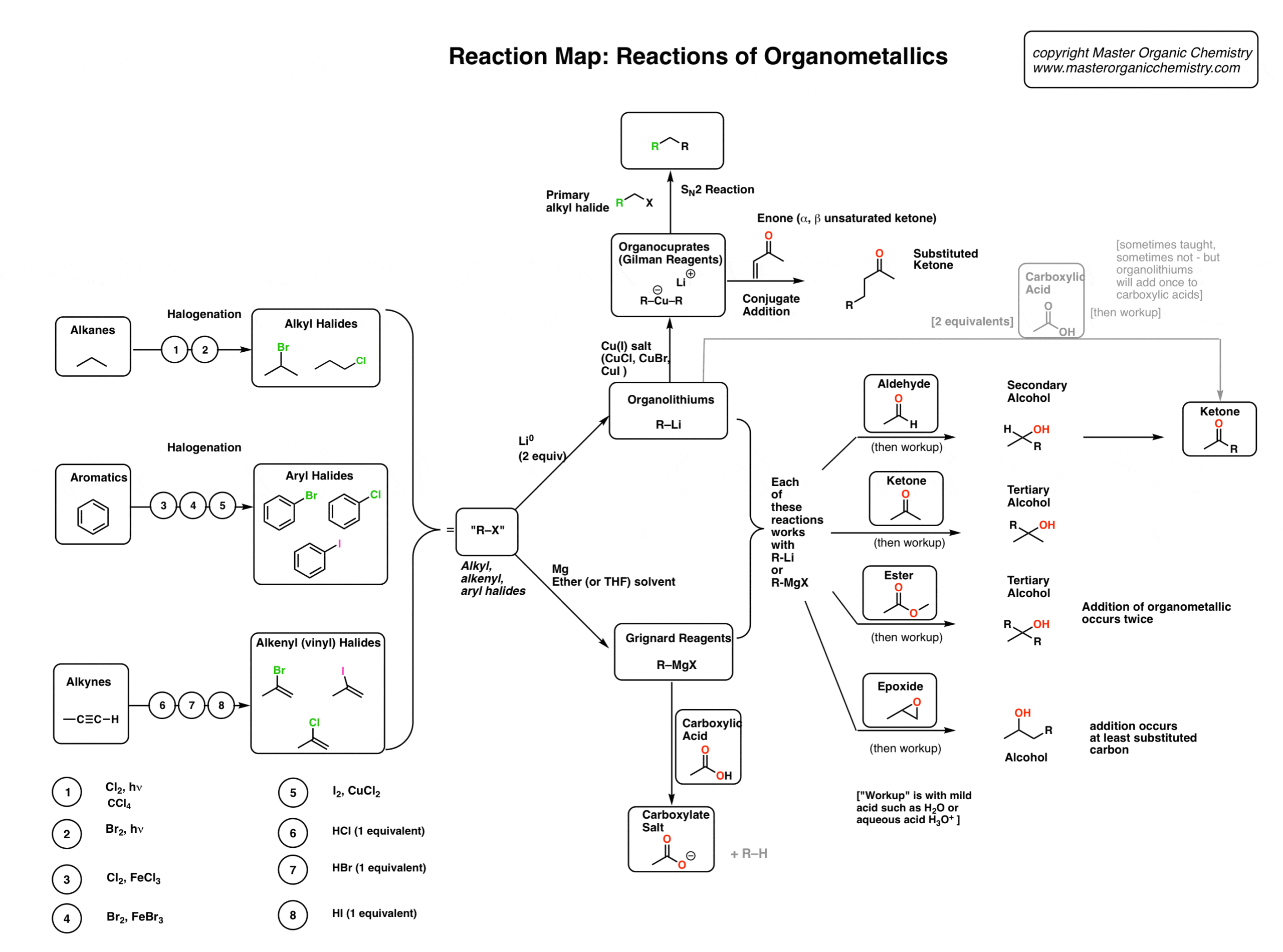
From 1º Alkyl Halides: Alkylation of Ammonia (Section 19-12, 19-21A) (See reaction 3) R Br excess NH3 R NH2 • Access: 1º Amines only |
|
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
Simple secondary and tertiary amines are named in common nomenclature by designating the organic Reactions of Amines with Nitrous Acid • Nitrous acid |
|
Amines Amines - NCERT
In this degradation reaction, migration of an alkyl or aryl group takes place from carbonyl carbon of the amide to the nitrogen atom The amine so formed contains Chemicals of this type: Aniline |
|
145 Chapter 24: Amines Amines: Nitrogen containing organic
Gabriel amine synthesis: reaction of potassium phthalimide with alkyl halides or tosylates via an SN 2 reaction The resulting N-substituted phthalimide can be |
|
Amines Organic derivatives of ammonia, NH3 Nitrogen atom have
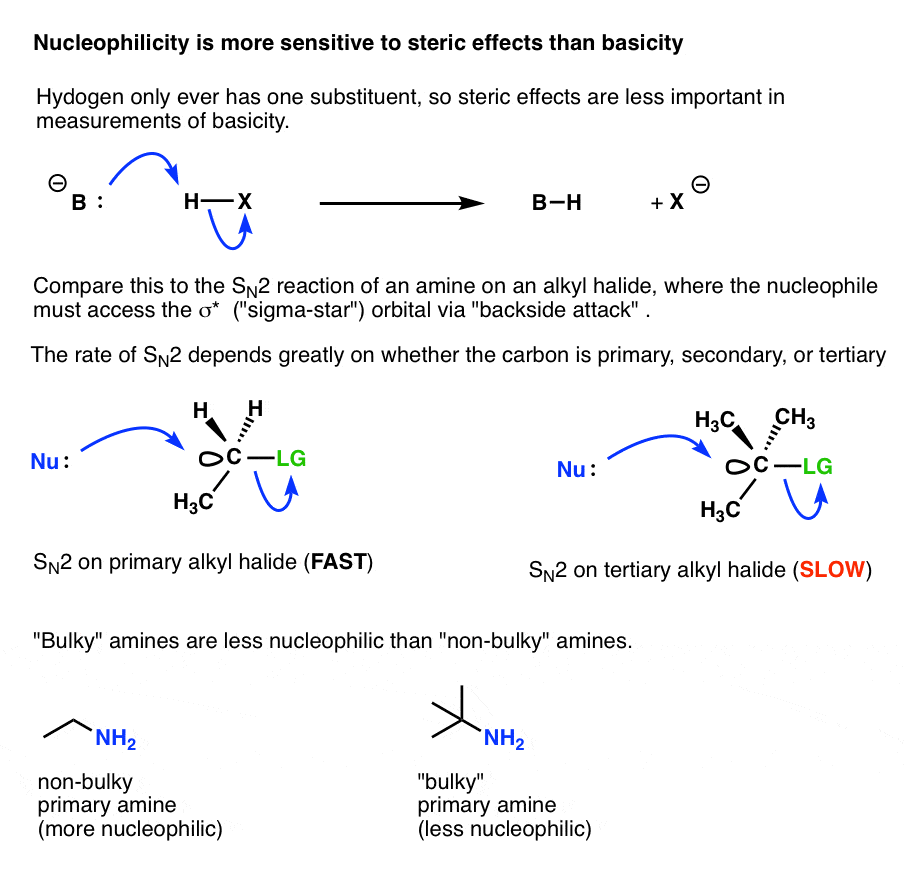
yielding alkyl amines 1°, 2°, and 3° amines all have similar reactivity; the initially formed monoalkylation product can undergo further reaction |
|
Nucleophilic Reactivities of Primary and Secondary Amines in
24 nov 2009 · However, for some reactions of secondary amines with quinone methides, the plots of kobs versus amine concentration showed an up- ward |
|
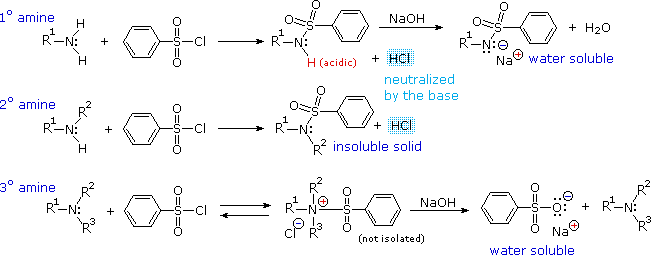
Amines
No reaction with tertiary (3°) amines • This reaction is used for the distinction of 1 °, 2° and 3° amines Electrophilic Substitution Reactions • Aniline is a resonance |