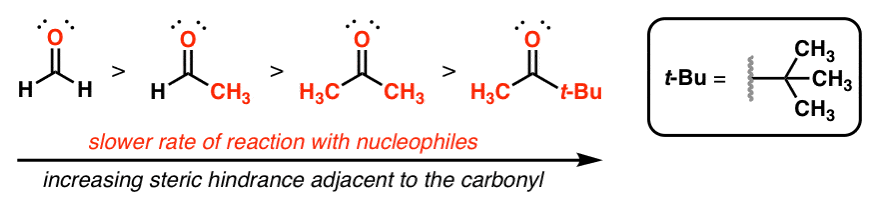
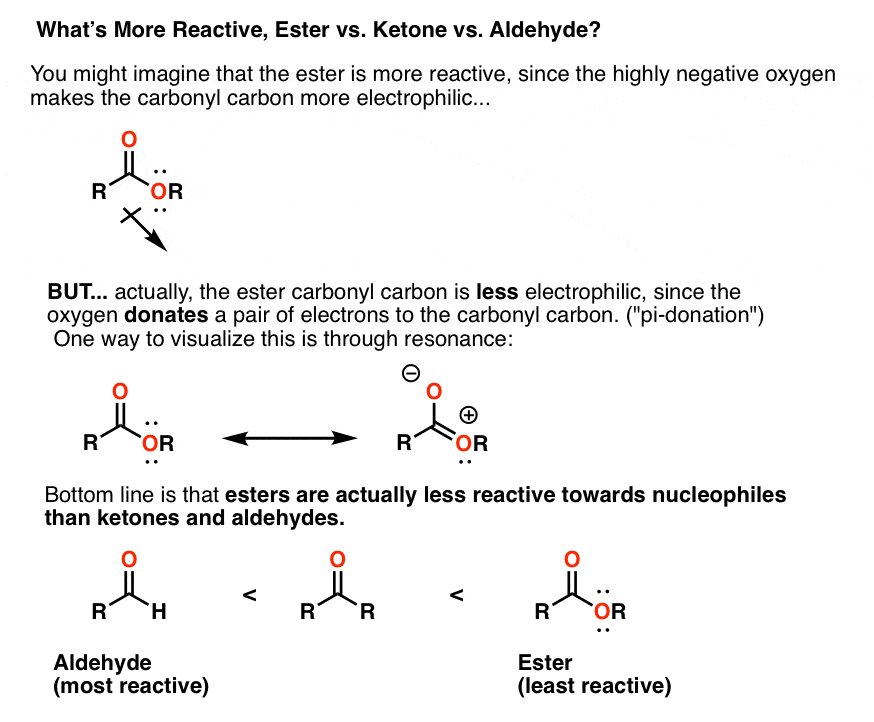
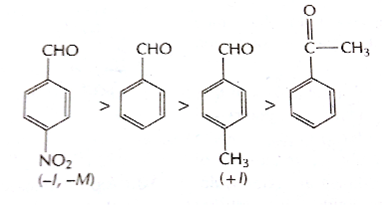
reactivity of carbonyl compounds towards nucleophiles
|
Chemistry II (Organic) Heteroaromatic Chemistry LECTURE 6
conjugated imines/carbonyl compounds: susceptibility to attack by reactivity: unreactive towards most electrophiles (E+); <<benzene (relative ... |
|
The sulfonamide motif as a synthetic tool
22 Oct 2010 to nucleophilic displacement of leaving groups at ... atom demonstrating their different reactivity compared to carbonyl compounds (Scheme 20).21. |
|
Gas phase nucleophilic displacement reactions of negative ions with
nucleophile toward the carbonyl substrate. All studies reported thus far Asubiojo Brauman / Displacement Reactions of Negative Ionswith Carbonyl Compounds. |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
Since aldehydes and ketones both possess the carbonyl functional group they undergo similar chemical reactions. 1. Nucleophilic addition reactions. Contrary to |
|
Profiling the reactivity of cyclic C-nucleophiles towards electrophilic
7 Oct 2015 thus shows similar reactivity to compounds listed in Chart 2). Next ... a carbonyl group with a sulfone moiety on 5-membered ring system C ... |
| Amide activation: an emerging tool for chemoselective synthesis |
|
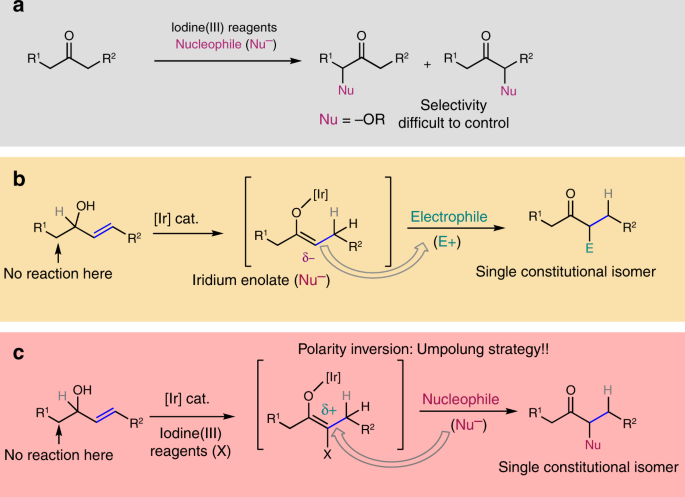
Umpolung of the Reactivity of Carbonyl Compounds Through Sulfur
Product 28 - 71 Reactivity of Carbonyl Compounds towards Nucleophiles (N") and Electrophiles (E"). Normal reactivity. Reactivity with. Umpolung. Sulfur ... |
|
A theoretical treatment of nucleophilic reactivity in additions to
Nucleophilic Reactivity in Additions to Carbonyl Compounds contributor to the curvature is the extent of odd-electron delo- calization in the charge |
|
Reactivity of the carbon–carbon double bond towards nucleophilic
pattern of an adjacent carbonyl group thereby leading to the well known result that the a |
|
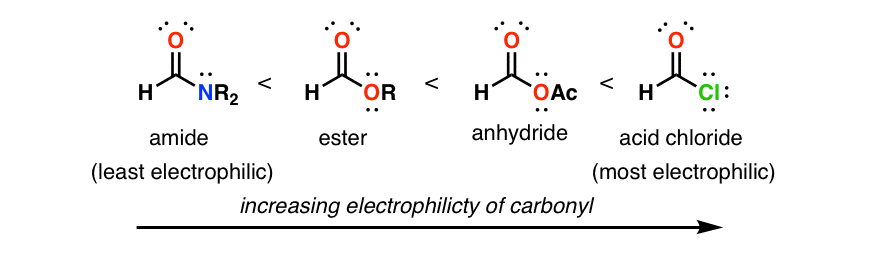
Carbonyl Reactivity
reactivity of the following carbonyl compounds towards nucleophilic attack. Most reactive. Least reactive. OH. O. NR. NR2. O. O. Carbonyls have a "resonance |
|
Carbonyl Reactivity
reactivity of the following carbonyl compounds towards nucleophilic attack. Most reactive. Least reactive. OH. O. NR. NR2. O. O. Carbonyls have a "resonance |
|
Reactivity of the carbon–carbon double bond towards nucleophilic
towards nucleophilic addition reactions. A wide family of molecules including The nucleophilic activation of the ab-unsaturated carbonyl compounds is a. |
|
Reactivity of the carbon–carbon double bond towards nucleophilic
towards nucleophilic addition reactions. A wide family of molecules including The nucleophilic activation of the ab-unsaturated carbonyl compounds is a. |
|
A theoretical treatment of nucleophilic reactivity in additions to
focusing on the problem of nucleophilic reactivity at carbonyl model carbonyl compounds in the gas phase6*1 theoretical studies. |
|
Conformational preferences of ?-fluoroketones may influence their
Dec 29 2017 different ?-halogen atoms have on carbonyl reactivity. ... various ?-halogenated ketones towards nucleophilic addition. |
|
Amide activation: an emerging tool for chemoselective synthesis
Aug 28 2018 compared to other carbonyl and carboxyl derivatives |
|
Chemical reactivity of carbonyl compounds
into the reactivity scale towards nucleophiles: Acyl halides > anhydrides > aldehydes > ketones > esters = carboxylic acids > amides. |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones
nucleophilic addition to carbonyl compounds; however other nucleophiles can undergo the same reaction. carbon more reactive toward the alkyl group. |
|
Mechanism of the antigen formation of carvone and related α βâ
Both these compounds are still ketones but the reactivity towards nucleophilic groups in pro- teins is greatly changed compared to carvone; in 1 the reactive |
|
Transition metal catalysed reactions of alcohols using borrowing
Nov 21 2008 metal catalyst to generate an intermediate aldehyde or ketone. The so-formed carbonyl compound has a greater reactivity towards nucleophilic ... |
|
THE CHEMISTRY OF THE CARBONYL GROUP - University of Oxford
4) Enolisation of carbonyl compounds A) keto-enol tautomerism B) enols and enolates as nucleophiles C) condensation reactions with carbonyl groups D) conjugate additions 5) Making Alkenes • Wittig reaction Suggested Reading: Core Carbonyl Chemistry J Jones Oxford Primer Organic Chemistry Clayden Greeves Warren and Wothers |
|
Nucleophilic Addition: Definition Example and Mechanism
Abstract—Reactive carbonyl compounds (RCC) are a group of compounds with clearly pronounced electrophilic properties that facilitate their spontaneous reactions with numerous nucleophilic reaction sites in proteins lipids and nucleic acids |
|
NUCLEOPHILIC ADDITION TO CARBONYL-CONTAINING COMPOUNDS
NUCLEOPHILIC ADDITION TO CARBONYL-CONTAINING COMPOUNDS GRIGNARD REACTION Carbonyl-containing compounds such as aldehydes ketones carboxylic acid and acid anhydrides possess an electophilic carbon center which can be attacked by various nucleophiles (Figure 1) ? |
|
First Year Organic Chemistry THE CHEMISTRY OF THE CARBONYL
B) Factors that affect reactivity of C=O towards nucleophiles; leaving group ability; IR spectroscopy C) The reactivity of acid chlorides (RCOCl) D) The reactivity of anhydrides (RCO) 2 O E) The reactivity of esters COOR F) The reactivity of amides CONR 2 4) Enolisation of carbonyl compounds A) keto-enol tautomerism |
|
Lecture Notes Chem 51C S King I The Reactivity of Carbonyl
• The reactivity of an organometallic compound towards an electrophile depends on the polarity of the carbon-metal bond Preparation of Organometallic Reagents: Organolithium and organomagnesium (Grignard) Reagents are typically prepared by reaction of an alkyl halide with the corresponding metal Organolithium reagents: example: |
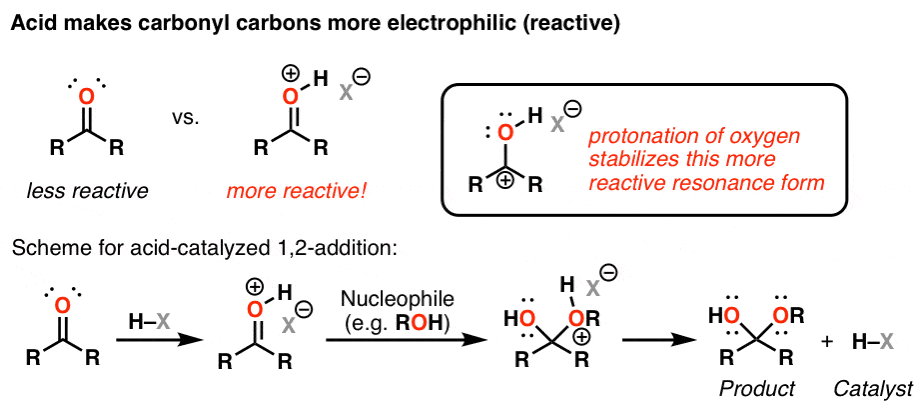
Why do carbonyl compounds undergo nucleophilic addition reactions?
- This reaction is essential in organic chemistry because it enables carbonyl compounds to form new products having various functional groups. Carbonyl compounds undergo nucleophilic addition reactions because of the C=O bond’s polarity.
Can a carbonyl be a nucleophile?
- 13. base base, ROH heat (or acid) Organic Chemistry II Review Jasperse Enolate Chemistry 17 1. If one carbonyl lacks any ?-hydrogens, it can’t be converted to nucleophile and can only function as electrophile 2.
Which nucleophiles attack at the carbonyl group in cyclic sulfinic–carboxylic anhydr?
- Nucleophiles such as alcohols attack at the carbonyl group in the cyclic sulfinic–carboxylic anhydride (15) to give ?-acylsulfinic acids ?86KFZ843, 88IZV1633?. Alcoholysis of 1,2-oxathiolane 2,2-dioxide ( 14) proceeds by attack at the 5-carbon to give ?-alkoxysulfonic acids ?54LA (586)151?.
What is the role of the carbonyl group?
- The carbonyl group has received great attention, due in part to its varied reactivity and ubiquity across chemistry and biology. In addition to the enormous catalogue of chemical transformations supported by carbonyl groups, their intermolecular interactions play a paramount role in the organization of biological systems.
|
Reactions involving carbonyl compounds - NPTEL
When nucleophile adds to carbonyl group, its HOMO reacts with LUMO of carbonyl group So when this LUMO is more polarized i e , coefficient of LUMO at carbon is greater, then this interaction is better to filling of electrons to the antibonding π* orbital of carbonyl group |
|
Nucleophilic addition to the carbonyl group
However, special factors can shift the equilibrium towards the hydrated form even for ketones, particularly if the carbonyl compound is reactive or unstable |
|
Reactions of Carbonyl Compounds
ketones, esters, amides, and other carboxylic acid derivatives are intimately associ- In many reactions at carbonyl groups, a key step is addition of a nucleophile, of the reaction system is a more effective nucleophile toward the carbonyl |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
The reactions of aldehydes and ketones can be divided into two main categories: nucleophilic addition to carbonyl compounds; however, other nucleophiles can similar to acid chlorides, although they are much less reactive towards these |
|
Chemical reactivity of carbonyl compounds - International Journal of
into the reactivity scale towards nucleophiles: Acyl halides > anhydrides > aldehydes > ketones > esters = carboxylic acids > amides Properties of Aldehydes |
|
Nucleophilic Addition Reactions of Carboxylic Acid Derivatives
The most important reaction for aldehydes and ketones is nucleophilic addition Relative Reactivity of Carboxylic Acid Derivatives Towards Nucleophilic Attack |
|
UNIT 6 NUCLEOPHILIC ADDITION TO CARBONYL COMPOUNDS
In this unit we shall discuss the nucleophilic addition reactions'of aldehydes of the carbon-oxygen double bond get shifted towards the oxygen atom and the |
|
16: Addition and Substitution Reactions of Carbonyl Compounds
"Uncatalyzed" Addition of HOH to Carbonyl Compounds (16 3D) 16-17 Reactions of nucleophiles with carbonyl groups are among the most important We can shift the position of that equilibrium toward the acetal by removal of the product |