regular solution model
|
Lecture 3: Models of Solutions
?GM the essential term in all thermodynamic models for solutions. Ideal Solution In the regular solution model |
|
A generalized regular solution model for asphaltene precipitation
Alboudwarej |
|
The effect of the regular solution model in the condensation of
We assume thermodynamic equilibrium and solve for the condensation sequence using the regular solution model. (DeHoff 1993) for the behaviour of solid solutions |
|
Regular Solution Model for Asphaltene Precipitation from Bitumens
Akbarzadeh |
|
Lecture 17 8/06/14 A. Regular Solutions
6 août 2014 A. Regular Solutions: A Simple Example of a Real Solution. • The simplest non-ideal solution model that works beyond the Henry's Law model. |
|
A regular-solution model with applications to carbonatite magma +
The regular-solution model of carbonate melts can be applied to problems of carbonatite magma + vapor equilibria and of extrapolating liquidus equilibria to |
|
Thermodynamic modelling of solid solutions
Guggenheim or Redlich–Kister Simple Mixture and Regular Solution models. Guggenheim (1937) suggested that the molar excess Gibbs energy of mixing of a |
|
Extending the Modified Regular Solution Model To Predict
20 déc. 2019 The main disadvantages are that (1) the model is limited to liquid?liquid equilibrium (2) only asphaltenes and resins can partition to the. |
|
Application of the Modified Regular Solution Model to Crude Oils
24 juil. 2020 The two classical thermodynamic approaches used to model asphaltene precipitation are equations of state (EoS) and the modified regular solution ... |
|
A REGULAR-SOLUTION MODEL FOR INTERSTITIAL SOLUTIONS
A REGULAR-SOLUTION. MODEL FOR INTERSTITIAL. SOLUTIONS IN HCP METALS. FIATS HILLERT and 4lhGXUS JARL. The Royal Institute of Technology. |
|
Lecture 3: Models of Solutions
In the regular solution model the enthalpy of mixing is obtained by counting the different kinds of near neighbour bonds when the atoms are |
|
Lecture 17 8/06/14 A Regular Solutions - University of Washington
6 août 2014 · This lattice-based theory of solutions is sometimes called regular solution theory A regular solution is non-ideal but the interactions between |
|
The Term •Regular Solution - Nature
A regular solution is one involving no entropy change when a small amount of one of its components is transferred to it from an ideal solution of the same |
|
The theory of regular solutions
The usual theory of regular solutions which is based on the assumption that all molecules are on the sites of a lattice is modified |
|
A “SUB-REGULAR” SOLUTION MODEL AND ITS APPLICATION TO
fractions; the model may be termed a “sub-regular” solution The equations for the terminal solubility curves of elements with the same lattice structure |
|
Chapter 9 Behavior of Solutions
9-10 Statistical (Quasi-chemical) Model of Solution 22 § 9-11 Subregular Solution The simplest mathematical form of regular solution is?HM =?'XAXB |
|
Regular Solution Theory PDF - Scribd
Regular Solution Theory - Free download as PDF File ( pdf ) Text File ( txt) or read online for free A report on regular solution theory on mixtures |
|
ASPECTS OF THE REGULAR SOLUTION MODEL AND ITS
For systems which complied with the regular solution behaviour the binary interaction energies between cations could be readily deduced from the published data |
|
Lecture 25: Ordering Transformation
Understanding of the concepts of “ideal solution” “regular solution” and “real by a so-called “quasichemical model” --- i e the heat of the mixing |
What is a regular solution model?
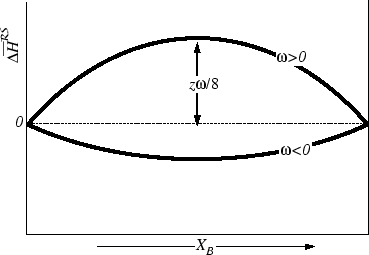
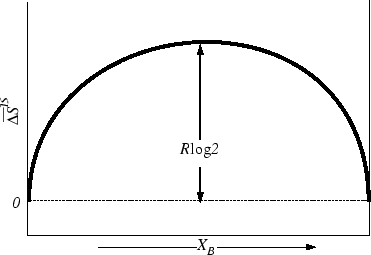
The regular solution model is a quantitative explanation of non-ideal behaviour. The model assumes that the entropy of mixing is the same as ideal mixing but that the enthalpy of mixing is not zero. We now examine how a non-zero enthalpy of mixing might depend on concentration.What is a regular solution in thermodynamics?
A regular solution is one involving no entropy change when a small amount of one of its components is transferred to it from an ideal solution of the same composition, the total volume remaining unchanged. '' I referred to the possibility of expressing the partial. molal enthalpy ( of component 2) by the Margules.How is the enthalpy of mixing regular solution derived?
In the regular solution model, the enthalpy of mixing is obtained by counting the different kinds of near neighbour bonds when the atoms are mixed at random; this information together with the binding energies gives the required change in enthalpy on mixing.- The Gibbs free energy of a mixture is defined as G=?j?jnj where ?j is the chemical potential of species j, and it is temperature and pressure dependent, and nj is the number of moles of species j.
|
Regular solutions - Faculty Washington - University of Washington
6 août 2014 · A Regular Solutions: A Simple Example of a Real Solution • The simplest non- ideal solution model that works beyond the Henry's Law model |
|
Thermodynamic modelling of solid solutions - Geosciences
Guggenheim or Redlich–Kister, Simple Mixture and Regular Solution models Guggenheim (1937) suggested that the molar excess Gibbs energy of mixing of a |
|
A REGULAR SOLUTION MODEL FOR PHASES WITH SEVERAL
Expressions for the partial Gibbs energy are derived The implementation of the model on a computer is discussed 1 INTRODUCTION The term regular solution |
|
Aspects of the Regular Solution Model and Its - Pyrocoza
For systems which complied with the regular solution behaviour, the binary interaction energies between cations could be readily deduced from the published data |
|
Model of the Excess Gibbs Energy for a Regular Solution
Introduction simplest solution Regular solution, models that have originally symmetric regular solution model is established by introducing the interaction |
|
SOLUTIONS AND SOLUTION MODELS
A few non ideal solution models of general interest for multicomponent systems, like the regular and subregular models, will then be examined before dis- |
|
Binary Solutions
Regular solutions: Heat of formation of a solution ➢ Real solutions: interstitial solid model called statistical or quasi-chemical model In this model the heat of |





![PDF] Regular Solution Theory and its Application : A Preliminary PDF] Regular Solution Theory and its Application : A Preliminary](http://pruffle.mit.edu/3.00/Lecture_32_web/img42.gif)










![4-Power Series Solution at Regular Singular Point-2 - [PDF Document] 4-Power Series Solution at Regular Singular Point-2 - [PDF Document]](https://upload.wikimedia.org/wikipedia/commons/thumb/1/1f/Entropy_of_Mixing.jpg/220px-Entropy_of_Mixing.jpg)





![The Mo−Ti (Molybdenum-Titanium) system - [PDF Document] The Mo−Ti (Molybdenum-Titanium) system - [PDF Document]](https://img.yumpu.com/7406078/1/500x640/alaras-2012-resume-in-pdf-format-liquid-mercury-solutions.jpg)











![Midterm Solution [pdf] - Caltech Midterm Solution [pdf] - Caltech](https://www.learncbse.in/wp-content/uploads/2019/05/NCERT-Solutions-for-Class-8-Maths.png)
