saponification of ethyl acetate and sodium hydroxide in cstr lab report
|
CHEMICAL REACTION ENGINEERING LABORATORY LAB MANUAL
To determine the reaction rate constant for saponification of ethyl-acetate with NaOH at ambient conditions. THEORY: In an ideal CSTR (that is an ideal steady |
|
Analysis of Chemical Reactors for Saponification Project: Final
In the laboratory the reaction of ethyl acetate and sodium hydroxide to form The second reactor |
| Experimental Studies of Ethyl Acetate Saponification Using Different |
| Effect of Operating Conditions on CSTR performance: an |
|
Reaction Engg. & Thermodynamics Lab
reactor: Determine the reaction rate constant for the saponification of ethyl acetate with sodium hydroxide. [non-Equimolar]. |
| REACTION ENGINEERING 5.1. Determination of Kinetic Parameters |
|
Jawaharlal Nehru Engineering College
A] Aim: To study batch reactor kinetics for ethyl acetate - NaOH and find the A] Aim: To carry out saponification of ethyl acetate in given CSTR and to ... |
|
CHEMICAL REACTION ENGINEERING LAB EXPERIMENT
lutions employed were sodium hydroxide (0.2 M I 00 cm3. ) and ethyl acetate (0.25 M It is known that the ethyl acetate saponification reaction is. |
| Eric T Henderson |
|
Aspen Plus Simulation of Saponification of Ethyl Acetate in the
In this experiment saponification of ethyl acetate in the presence of sodium hydroxide is used to demonstrate the saponification reaction. The saponification |
|
Article Experimental Studies of Ethyl Acetate Saponification
Saponification of ethyl acetate using sodium hydroxide was considered to be a model reaction This is a well-investigated second-order homogeneous liquid phase reaction [4546]: CHCOOCH+NaOH ?CHCOONa+CHOH (2) where k denotes the reaction rate constant The conversion of sodium hydroxide was calculated: X = c?c c ?100 (3) |
|
Estimation of Parameters of Arrhenius Equation for Ethyl Acetate
hydroxide and ethyl acetate in a continuous-stirred tank reactor (CSTR) The saponification process is a process that produces soap usually from fats and lye In technical terms saponification involves base (usually caustic soda NaOH) hydrolysis of triglycerides which |
|
Experiment C5 Chemistry 114 Kinetics in Solution
Goals: To measure rate constants for the saponification of Ethyl acetate in sodium hydroxide solutions at several temperatures From these rate constants the activation energy and entropy of activation can be calculated Additional experiments will explore the order of the reaction and the saponification rate of other esters Background |
|
CHEMICAL ENGINEERING LABORATORY
Saponification of Isopropyl Acetate with Sodium Hydroxide Objective: The purpose of this experiment is to examine and determine the reaction kinetics of a simple homogenous liquid-phase system and to model this reaction in a continuously stirred-tank reactor (CSTR) |
|
Effect of Operating Conditions on CSTR performance: an
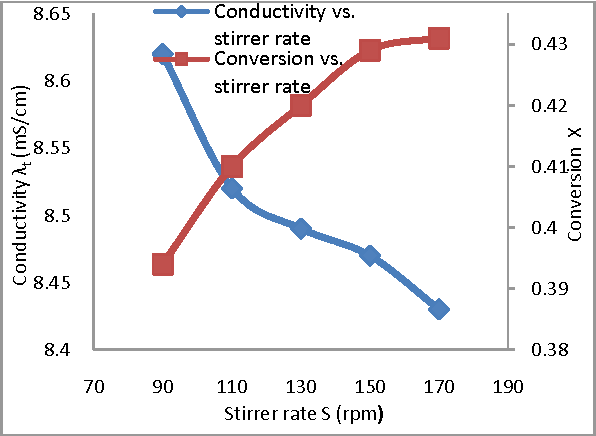
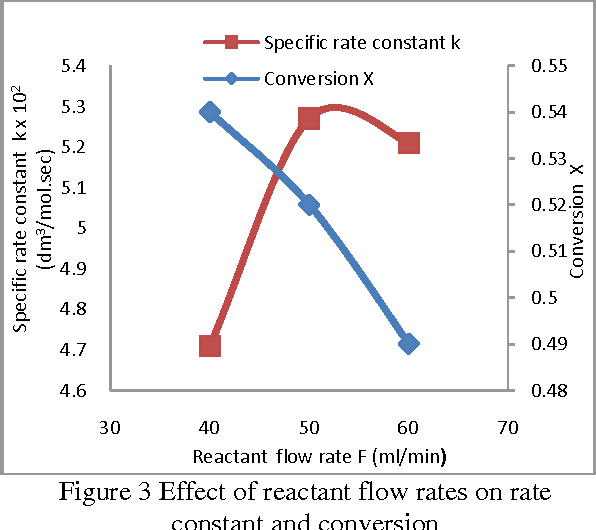
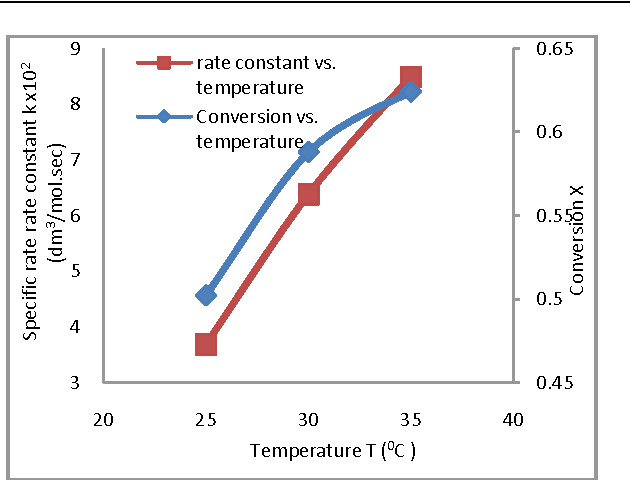
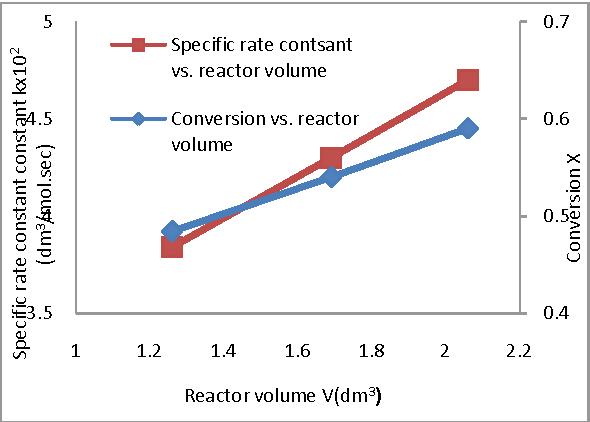
In this work Saponification reaction of ethyl acetate by sodium hydroxide is studied experimentally in a continuous stirred tank reactor at 1 atmospheric pressure The aim of this study is to investigate the influence of operating conditions on the conversion and specific rate constant |
|
Estimation of Parameters of Arrhenius Equation for Ethyl
In this scientific research a Saponification Reaction between Ethyl Acetate and caustic soda is carried out in a Batch Reactor at STP Conditions The aim of this scientific research is to estimate the parameters of Arrhenius equation which are rate constant and activation energy for ethyl acetate saponification |
Is saponification reaction of ethyl acetate and sodium hydroxide irreversible?
- Reaction Kinetics: Saponification Reaction of Ethyl Acetate and Sodium Hydroxide is an irreversible 2nd order overall, 1st order with respect to reactants furthermore reaction order decreases and become sequential rather than 2nd order when equimolecular concentrations of both reactants are used7-10.
What chemicals were used in the saponification reaction?
- Chemicals: Analytical reagent (AR) - grade chemicals were utilized to conduct the experiments using PFR and CSTR. Ethyl acetate with 99.5% purity and sodium hydroxide with 98.0- 100% purity were used to carry out the saponification reaction.
What is the alkaline hydrolysis of acetate esters?
- The alkaline hydrolysis of acetate esters, also know as the saponification reaction can be written as: AcO-R + Na+ + OH- ? AcO- + Na+ + R-OH (1) As the reaction proceeds, hydroxide ions are consumed and acetate ions are produced.
What is an example of a reverse saponification reaction?
- is an example of a saponification reaction (the reverse reaction would be esterification). This reaction may be either reversible or irreversible. For the irreversible case, the rate equation for a batch reactor may be written: where k is the rate constant and [A] and [B] are the concentrations of the reactants in the appropriate units.
|
CHEMICAL REACTION ENGINEERING LABORATORY - VSSUT
given sponification reaction of ethyl acetate in aqueous sodium hydroxide solution in To determine the reaction rate constant for saponification of ethyl acetate with NaOH In an ideal CSTR (that is an ideal steady state flow reactor) the contents in the reactor are well mixed and Note: report k at the reaction temperature |
|
Analysis of Chemical Reactors for Saponification - Tylar Farmer
In the laboratory, the reaction of ethyl acetate and sodium hydroxide to form ethanol and the sodium The second reactor, the CSTR, is an open system which has a continuous feed flow and quantified through a series of technical objectives in the lab 6 are used in the study to compare results between the reactors |
|
Reaction Engineering Lab
Material balance done for batch reactors results in the following governing equation: During the reaction, sodium hydroxide will dissociate into sodium ion and hydroxide ion while Carefully introduce to the reactor 90 mL of 0 1 M ethyl acetate To determine the order and reaction rate constant for liquid phase in CSTR |
|
Effect of Operating Conditions on CSTR performance - International
ABSTRACT In this work, Saponification reaction of ethyl acetate by sodium hydroxide is studied experimentally in a Keywords– Conductivity, Conversion, CSTR, Hydrolysis, Saponification I Introduction 0C) were determined and it is noticed that results obtained Experiment was conducted at flow rate of 40 ml/ min of |
|
EXPERIMENT 2 - UniMAP Portal
IN A CONTINUOUS STIRRED TANK REACTOR (CSTR) EXPERIMENT 3 : EFFECT OF In case of emergency, report to the Instructor in charge or saponification reaction between ethyl acetate and sodium hydroxide among other types of |
|
Aspen Plus® Simulation of Saponification of Ethyl Acetate in the
1 sept 2014 · Saponification of Ethyl Acetate in the Presence of Sodium Hydroxide in The results obtained from the simulation were verified by the experiment carried lower than the Continuously Stirred Tank Reactor (CSTR) volume for |
|
Saponification of Ethyl Acetate
Ethyl Acetate This experiment illustrates a bimolecular reaction for which a A solution containing sodium hydroxide and ethyl acetate undergoes a marked |
|
Chapter 5
Determination of Kinetic Parameters of the Saponification Reaction in a PFR Safety Issues: In this experiment, sodium hydroxide (NaOH) and ethyl acetate data collected during the experiment performed in a CSTR, and the results will be |
|
A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate Using
ABSTRACT: Alkaline hydrolysis of ethyl acetate is essentially an irreversible and second order reaction Reaction conversion in plug flow reactor was investigated, and experimental results after analysis Design of experiment is an efficient technique of arranging Startup and Transient Response of CSTR's in Series, |













![PDF] Effect of Operating Conditions on CSTR performance: an PDF] Effect of Operating Conditions on CSTR performance: an](https://0.academia-photos.com/attachment_thumbnails/44144301/mini_magick20190215-22363-bx8sts.png?1550218483)

![PDF] Effect of Operating Conditions on CSTR performance: an PDF] Effect of Operating Conditions on CSTR performance: an](https://docplayer.net/docs-images/65/54216378/images/5-2.jpg)
![PDF] Effect of Operating Conditions on CSTR performance: an PDF] Effect of Operating Conditions on CSTR performance: an](https://www.coursehero.com/doc-asset/bg/dea376696839ac7e92b730c580944a2c74608f02/splits/v9.2.qiv2.clean/split-0-page-2-html-bg-unsplit.png)


![PDF] Effect of Operating Conditions on CSTR performance: an PDF] Effect of Operating Conditions on CSTR performance: an](https://www.coursehero.com/thumb/77/71/7771ba08f6a0c4ac54c9a58f318c5eada348bf24_180.jpg)