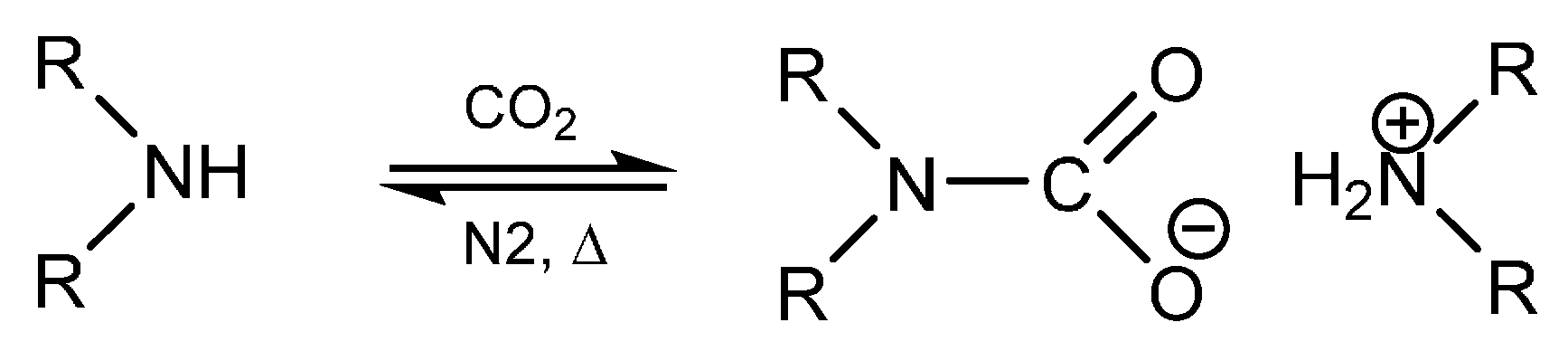
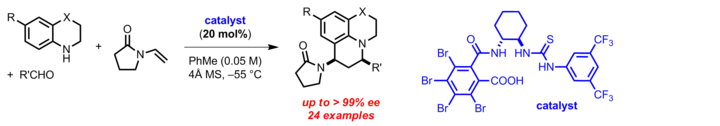
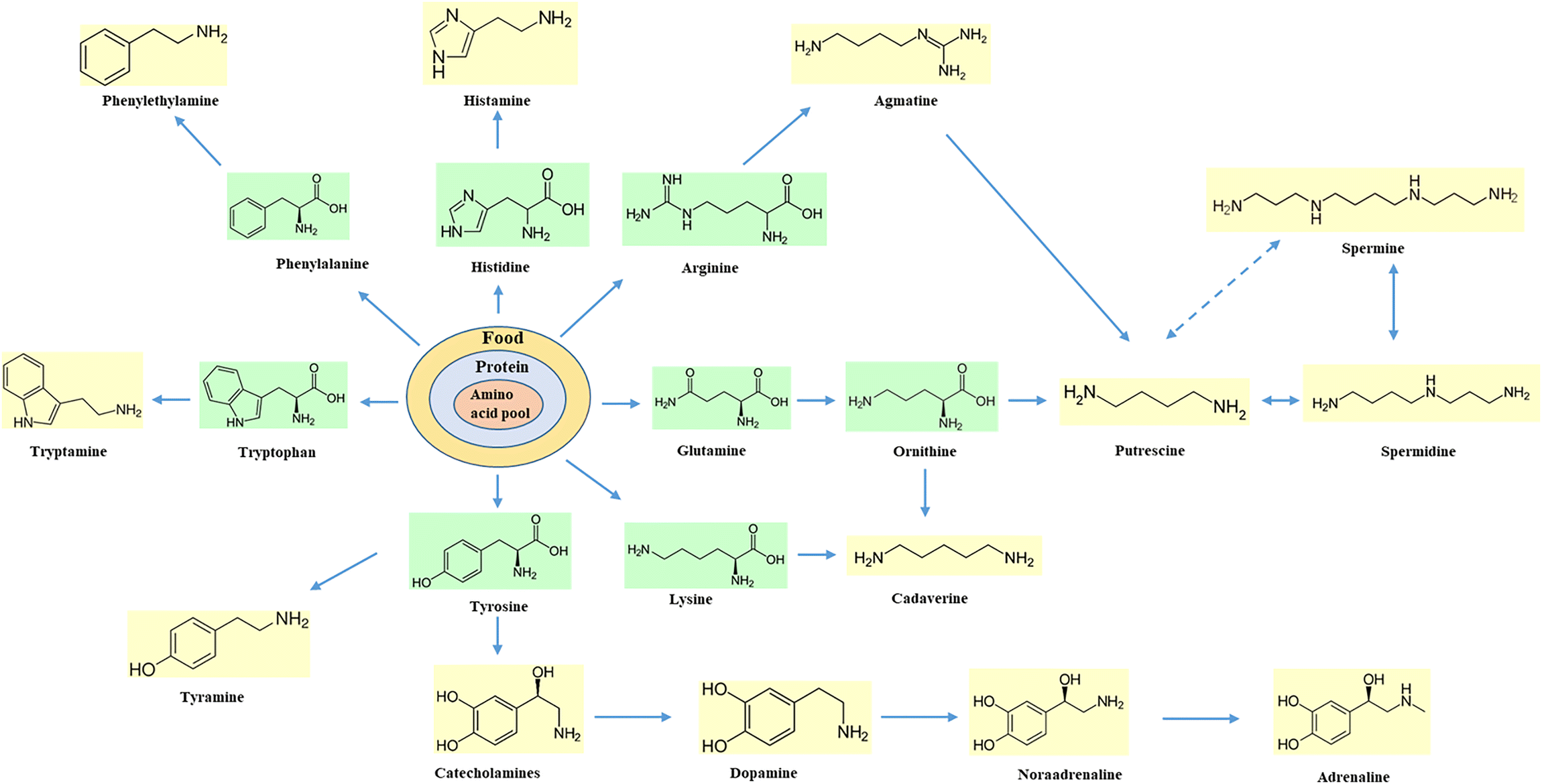
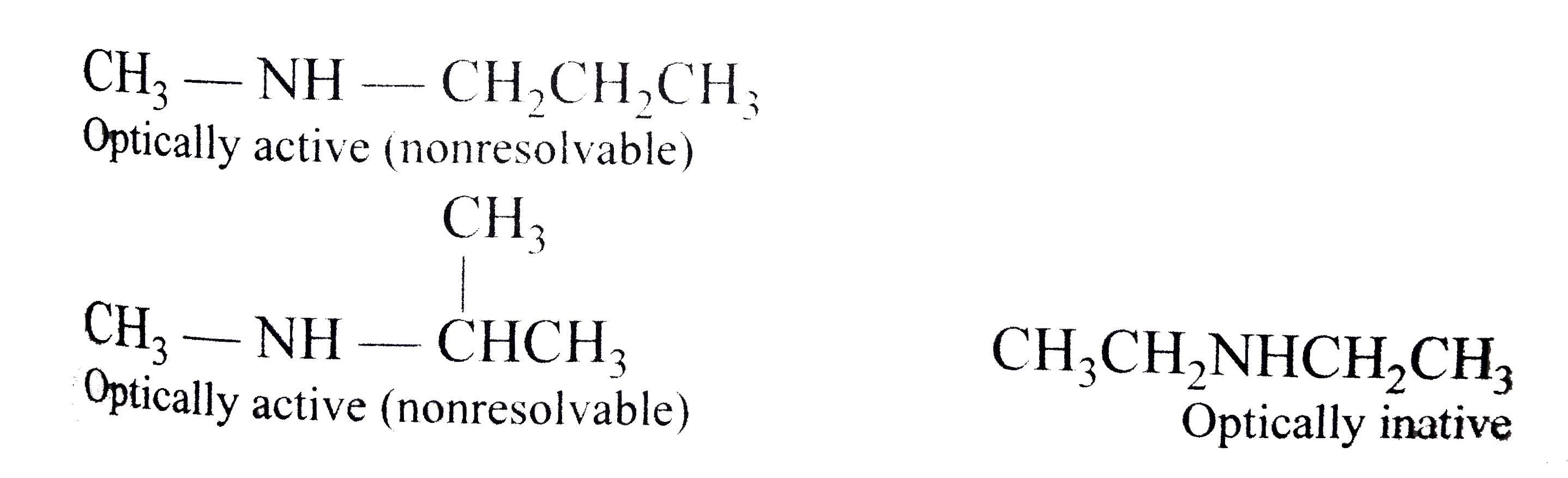secondary amine reaction with hcl
|
Maillard Reaction of Lactose and Fluoxetine Hydrochloride a
than formulations with starch as the diluent due to the Maillard reaction between the drug a secondary amine hydrochloride |
|
Maillard reaction of lactose and fluoxetine hydrochloride a
15 déc. 1997 than formulations with starch as the diluent due to the Maillard reaction between the drug a secondary amine hydrochloride |
|
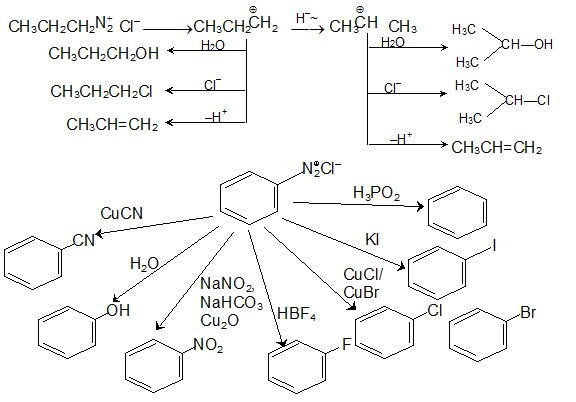
Reactions of Amines
But 1º 2º |
|
Reactions of Amines
But 1º 2º |
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to predict Amines are classified as primary (1°) |
|
Coupling Reagents
Secondary amine coupling . Wash the combined filtrates with 4% HCl(30 mL x 2) saturated NaHCO3 solution (25 mL x 3) and brine. |
|
Some Secondary Amines in the Sommelet Reaction
CN group with MeOH/HCl aq. which could conceivably A number of secondary amines have been subjected to the Sommelet reaction |
|
New Strategies for Organic Catalysis: The First Highly
molecules as reaction catalysts2 despite the widespread avail- series of chiral secondary amine‚HCl salts. As revealed in Table. |
|
N-Dealkylation of Amines
20 mai 2022 metabolic reaction of drugs containing amines catalyzed by members of the ... led to the HCl salt of the corresponding secondary amines. |
|
QCM-Based HCl Gas Detection on Dimethylamine-Functionalized
10 fév. 2022 Secondary amine salts were formed on ... tertiary amine salt is formed by a chemical reaction between the tertiary amine groups of. |
|
10 Reactions of Alcohols Ethers Epoxides Amines and
N1 Reaction of Secondary and tertiary alcohols The mechanism of the substitution reaction depends on the structure of the alcohol Secondary and tertiary alcohols undergo S N1 reactions MECHANISM FOR THE S N1 REACTION OF AN ALCOHOL CH 3CO HH H Br CH 3 CH 3 + + H 2O + ? CH 3CH CH 3 CH 3 CH 3CB r CH 3 CH 3 CH 3C+ CH 3 CH 3 CH 3C CH 3 HBr CH 2 |
|
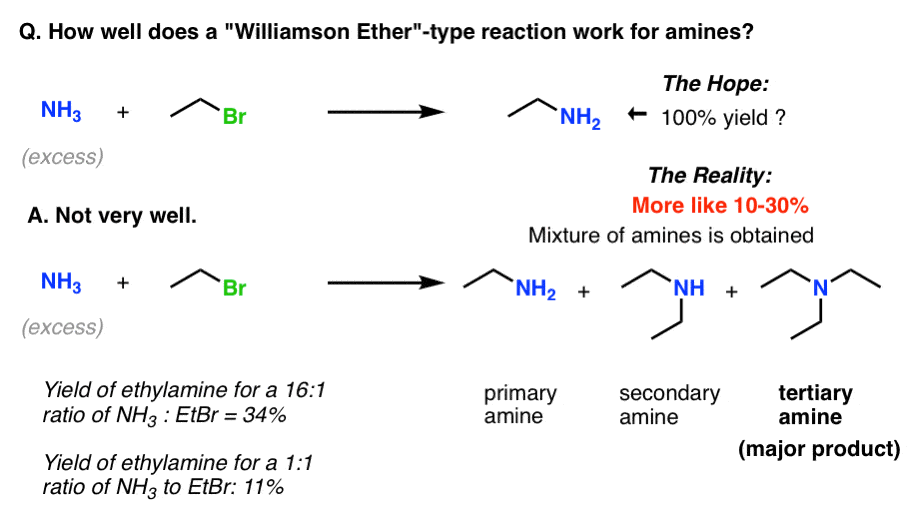
Reactions of Amines
Mechanism: Required (addition-elimination-deprotonation) Amine must have at least one hydrogen to begin But 1º 2º or NH3 all react well But 3º amines can’t work Some base is required for the deprotonation step and to absorb the HCl For cheap amines excess amine can simply be used |
| Covalent Scavengers for Primary and Secondary Amines - Thieme |
|
Synthesis and Characterization of Chiral Secondary Amine
Synthesis and characterization of chiral secondary amine catalysts (R)-Ethyl 22'-bis(methoxymethoxy)-11'-binaphthyl-3-carboxylate 11a To a solution of 10(15 g 40 mmol) in THF (600 mL) n-BuLi (24 mL 2 5 M in hexane 60 mmol) was added dropwise at -78 oC under argon atmosphere |
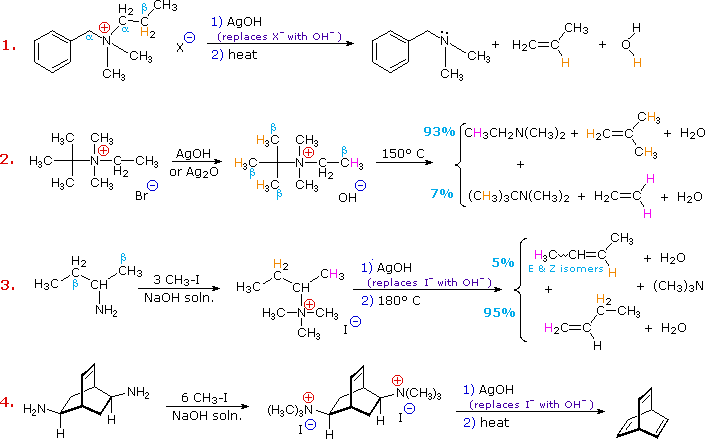
Why do tertiary amines react with quaternary ammonium ions?
- Because a tertiary amine is a relatively poor leav- ing group, the reaction requires heat. the leaving group is a tertiary amine CH 3 CH CH 2 HO NCH 3 H 2 O CH 3 CH 3 CH 3 CH 2 CH 2 NCH 3 ++ CH 3 CH 3 + quaternary ammonium ion
What is the formula for ammonium hydroxide and amine synthesis?
- 3 3 + c. NH O CH 3 CH 3 CH 3 + pRoBLem 48 SoL veD Describe a synthesis for the following compound, using the given starting material and any necessary reagents: CH 3 CH 2 CH 2 CH 2 NH 2 CH 3 CH 2 CH CH 2 SoLutioN Although an amine cannot undergo an elimination reaction, a quaternary ammonium hydroxide can.
What is the leaving group of a tertiary amine?
- Because a tertiary amine is a relatively poor leav- ing group, the reaction requires heat. the leaving group is a tertiary amine CH 3 CH CH 2 HO NCH 3
What is the reaction between HOCl and OCL?
- Because HOCl is unstable, it is generated in the reaction mixture by an acid–base reaction between H and OCl (using CH 3 COOH and NaOCl). Secondary alcohols are oxidized to ketones, and primary alcohols are oxidized to aldehydes. OH RR CH secondary alcohol ketone O C R R NaOCl CH 3 COOH 0 C CH 2 OH primary alcohol aldehyde O C R H R NaOCl CH 3
|
Reactions of Amines
But 1º, 2º, or NH3 all react well • But 3º amines can't work • Some base is required for the deprotonation step and to absorb the HCl For cheap amines, excess |
|
CHAPTER 7 AMINES
Naming Secondary and Tertiary Amines When there is more A molecule with two amine groups can react with two molecules of HCl or other acid NH2(CH2) |
|
AMINES
vanillin, or fural, and hydrochloric acid are most commonly used Some- times secondary aromatic amines react as well, but these give a weaker color (10) |
|
NITROGEN CONTAINING COMPOUNDS Amines - Patna Science
The reaction probably goes through the formation of an imine and tertiary amines can also be synthesized by reductive amination if a primary and secondary |
|
Amines - Hodder Education
Primary aliphatic amines such as methylamine (CH3NH2) can be prepared behave as either a base and react with the HCl produced: Secondary amine N |
|
610 Amines - chemrevise
As the reaction is carried out in HCl the salt C6H5NH3 +Cl- will be Reaction of primary Amines with halogenoalkanes forming secondary amines CH 3 |
|
FACTFILE: GCE CHEMISTRY - CCEA
ammonia, primary, secondary, tertiary aliphatic amines and phenylamine using the availability of the lone pair on the nitrogen atom; 5 7 7 recall the reaction of |
|
Experiment 16 Qualitative Analysis of Amines and Amine Unknown
This layer, however, will dissolve in 5 hydrochloric acid RNH R' Cl S O O KOH Figure 16 2 Reaction of Secondary Amines with Benzenesulfonyl chloride |
|
Have an NH2, amine group Amines are derivatives of ammonia
Phenylamine reacts with nitric acid: Questions 1 The excess ammonia reacts with the HCl formed: NH3 Preparation of secondary / tertiary aliphatic amines: |
|
Reaction of glucose with some amines - CORE
acid, showing the presence of a secondary amine and thus eon- firming the glueosidie hydrochloric acid, but became brown with alkali treatment and liberated |