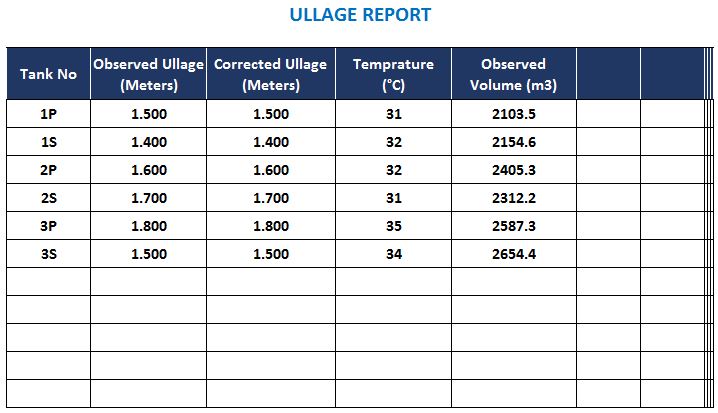
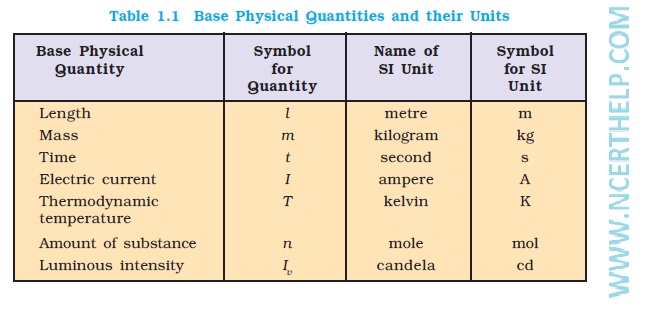
simple volumetric calculations chemistry
|
AS 03 Ans.p65
e) Number of moles in solution = molarity × volume (in dm3). = 0.0982 × 0.5 = 0.0491 (1). Mr. (NaOH) = 40 (1) SIMPLE VOLUMETRIC CALCULATIONS. |
|
Crystal Structures
Simple Cubic. 1. Find the volume of the space occupied in the unit cell: a) Count the number of atoms(spheres) inside the unit cell. b) Calculate the total |
|
CHAPTER 4:The Material Balance for Chemical Reactors General
Equation 4.5 applies whether the reactor volume is constant or changes during simple then analytical solutions of the material balance are possible. |
|
The Simple Method to Calculate Urban Stormwater Loads
The Simple Method estimates pollutant loads for chemical constituents as a product of annual runoff volume and pollutant concentration as:. |
|
Chapter 1 fundamental calculations in analytical chemistry
Basic operations for dilution. avoid misunderstanding. Thus to prepare a 1:4 acetic acid–ethanol solution |
|
Common Equations Used in Chemistry Equation for density: d= m v
Avogadro's law - Constant P and T: V = kn. Ideal Gas equation: PV = nRT. Calculation of changes in pressure temperature |
|
Basic Tools of Analytical Chemistry
Molality is used in thermodynamic calculations where a temperature in- dependent unit of concentration is needed. Molarity is based on the volume of solution |
|
EIIP Vol. II: Chapter 16 Methods for Estimating Air Emissions from
from Chemical Manufacturing Facilities 6-1 Calculating the moles of gas from volume temperature |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
A simple dilution is one in which a unit volume of a liquid material of interest is below is a quick approach to calculating such dilutions where:. |
|
CHEMISTRY (862)
(v) Chemical equivalents volumetric calculations Terms used in volumetric calculations such as ... Simple calculations on the above topics. |
|
Moles and equations Chapter 1 - Cambridge
This exercise gives you practice in calculating volumes moles and concentrations It also revises calculations from titration results Remember that:amount(inmol)Concentration (in mol dm ?3) = volume(indm)In some questions you will have to rearrange this equation |
|
Calculations In Chemistry How To Use this E-Book - ChemReviewNET
The 1250 page General Chemistry edition of Calculations In Chemistryis currently available as three paperback volumes for class sets (but not for individual purchase) Volume 1 Modules 1-16( ISBN 978-0-393-12552-8 ) 416 pages Volume 2 Modules 17-27( ISBN978-0-393-12553-5 ) 416 pages Volume 3 Modules 28-39( ISBN978-0-393-12554-2 ) 432 pages |
|
Chapter 3 Stoichiometry - Michigan State University
Calculating Empirical Formulas Calculate the mole ratio by dividing by the smallest number of moles: 5 105 mol C: 0 7288 mol = 7 005 ? 7 5 09 mol H: 0 7288 mol = 6 984 7 ? Calculating Empirical Formulas These are the subscripts for the empirical formula: C7H7NO2 H2N O O- Elemental Analyses |
What is volumetric method of analysis?
Volumetric method of analysis or sometimes referred to as titrimetric method of analysis is a quantitative method of analysis that is based upon the measurement of volume. These methods are considered important since they are usually rapid, convenient and are often accurate.
What is the volume-volume stoichiometry of a gas?
Volume-Volume Stoichiometry Avogadro's hypothesis states that equal volumes of all gases at the same temperature and pressure contain the same number of gas particles. Further, one mole of any gas at standard temperature and pressure ((0^ ext{o} ext{C}) and (1 : ext{atm})) occupies a volume of (22.4 : ext{L}).
What are the basic requirements of a volumetric method?
The basic requirements or components of a volumetric method are: A standard solution (i.e., titrant) of known concentration which reacts with the analyte in a known and repeatable stoichiometry (i.e., acid/base, precipitation, redox, complexation).
What is the difference between volumetric analysis and volumetric titrimetry?
The terms volumetric analysis specifically involves the determination of the volume of the reagent solution needed for a complete reaction. Volumetric titrimetry: methods that require that a reagent solution of known concentration, standard solution or titrant, be used.
|
VOLUMETRIC TITRATIONS I Introduction A Volumetric methods
Volumetric methods - measuring the volume of a solution (of known The reaction with analyte is selective and described by a simple, balanced equation IV |
|
What is Volumetric Analysis
derived ? 6- Calculate the concentrations of all species participating in the titration reaction at any In volumetric analysis, a chemical called a titrant is added to a where a moles of analyte A contained in a sample reacts with t moles of the |
|
Volumetric analysis
Volumetric analysis is a general term for a method in quantitative chemical analysis in In simple acid-base titrations a pH Calculations of volumetric analysis: |
|
Moles formulae equations questions - Chemstuff
Moles, Formulae and Equations Edexcel Advanced GCE in Chemistry (9080) Edexcel Advanced GCE in Exercise 11b Simple volumetric calculations 109 |
|
Chapter 4: Calculations Used in Analytical Chemistry - İYTE
Chapter 4: Calculations Used in Analytical Chemistry quantities in terms of a few simple digits, pre-fixes are particles as represented by a chemical formula |
|
Volumetric Analysis - TCD Chemistry
Molarity: Moles of solute per litre of solution ○ If we know the molarity and litres of solution, we can calculate the moles (and mass) of solute present Solution |
|
TITRIMETRIC ANALYSIS - NCERT
chemical equation and the relative molecular masses of the reacting compounds standard solution can be prepared by direct weighing of a sample of primary |
|
Volumetric Analysis
5th Year Chemistry Higher Level Sinéad Nolan Volumetric Analysis No part of this publication may be copied, A focus on simple shortcuts to raise students' |






/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)














/606823-calculate-molarity-of-a-solution-FINAL-5b7d7e15c9e77c0050355d4e.png)



