solution and colligative properties class 12 notes pdf
|
Unacademy
Page 12. 12. Solutions. 6. No chemical reaction between solute and solvent. 7. It From the colligative properties of solution which one is the best method ... |
|
Solution And Colligative Properties
6 Apr 2020 Notes: The solution: (1) liquid in gas (2) Solid in gas not possible ... An aqueous solution of sucrose C12H22O11 containing 34.2 g/L has an ... |
|
Aspirations Institute - Solutions
in solution to the theoretically calculated value of the colligative property. 12 |
|
Solutions
Another important class of solutions of particles present in the solution. Such properties are called colligative properties (colligative: from Latin: co ... |
|
SOLUTIONS & COLLIGATIVE PROPERTIES
How is the concentration of a solute present in trace amount in a solution expressed? *12. N. 2 and O. 2 gases have K. |
|
Class - XII Multiple Choice Question Bank [MCQ ] Term – I & Term-II
gases in liquids solid solutions |
|
LECTURE NOTES
Colligative properties are the properties of only dilute solution which are supposed to behave 12 m : molality of the solution. Molal boiling point elevation ... |
|
Lech102.pdf
In dilute solutions it is equal to concentration. You will study more about it in higher classes. 2.1 Electrochemical. Electrochemical. Cells. Fig. 2.1: |
|
Chemistry Notes for class 12 Chapter 2 Solutions
Colligative properties are those properties which depends only upon the number of solute particles in a solution irrespective of their nature. Relative |
|
Chapter 5 - Colligative Properties
The result is often derived in physical chemistry books. In applying boiling point elevation to polymer solutions we should realize that polymer solu- tions |
|
Solutions
studying the properties of the solutions like vapour pressure and colligative properties. We will begin with 12 × 2 + 1 × 6 + 16 × 2 = 62 g mol–1. |
|
Chemistry Notes for class 12 Chapter 2 Solutions
Colligative properties are those properties which depends only upon the number of solute particles in a solution irrespective of their nature. Relative Lowering |
|
Class 12th Chemistry Notes: Solutions (Part – III) Colligative
Colligative properties are those properties which depends only upon the number of solute particles in a solution irrespective of their nature. Relative Lowering |
|
Solutions
studying the properties of the solutions like vapour pressure and colligative properties. We will begin with 12 × 2 + 1 × 6 + 16 × 2 = 62 g mol–1. |
|
Solutions and Colligative Properties
Used when both the components of solution are in liquid phase. •. Total volume of solutions is not equal to sum of volumes of solute and solvent as same solute |
|
Solution And Colligative Properties
Apr 6 2020 Notes: The solution: (1) liquid in gas (2) Solid in gas not possible because they do not form homogenous mixture. (B) On basis of on of ... |
|
Solutions-and-colligative-properties-iit-jee-questions.pdf
Chemistry Notes In Hindi Description Of me And Colligative Properties. 12th. In this section we will provide you with NEET question banks for Physics |
|
Colligative Properties
COLLIGATIVE PROPERTIES. V a p or Pressure. Solution. Temperature. Pure Solvent. 1 atm. T b. AT. Figure 5.1: Boiling point elevation effect is a consequence |
|
LECTURE NOTES
Colligative properties are the properties of only dilute solution which are 12 m : molality of the solution. Molal boiling point elevation constant or ... |
|
Solutions Notes for Class 12 Pdf Free Download Isotonic Solid
Colligative properties are those properties which depends only upon the number of solute particles in a solution irrespective of their nature. |
|
Solutions and Colligative Properties - Saptarshi Classes
Used when both the components of solution are in liquid phase • Total volume of solutions is not equal to sum of volumes of solute and solvent as same solute |
|
Solutions (Part – III) Colligative Properties [Colligatil1e : from Latin
Class 12th Chemistry Notes: Solutions (Part – III) Colligative Properties [Colligatil1e : from Latin = Co mean 'together'; ligare means 'to bind' ] |
|
Chemistry Notes for class 12 Chapter 2 Solutions
Colligative properties are those properties which depends only upon the number of solute particles in a solution irrespective of their nature Relative Lowering |
|
Solution And Colligative Properties
6 avr 2020 · Solution:A solution is a homogeneous mixture of two or more non reacting substances on molecular level Solute and Solvent: The contituent |
|
SOLUTIONS & COLLIGATIVE PROPERTIES - GGPS Bokaro
The properties that depend on the number of solute particles irrespective of their nature relative to the total number of particles present in the solution are |
|
SOLUTIONS AND COLLIGATIVE PROPERTIES - MasterJEE Classes
(a) Solutions are homogenous mixtures of two or more substances (b) We call what is being dissolved in the solution as a “solute”: for it is the active |
|
Solutions and Colligative Properties
Solution is a homogeneous mixture of two or more substances in same or different physical phases The substances forming the solution are called components |
|
Solutions Class 12 Notes CBSE Chemistry Chapter 2 - Vedantu
Colligative Properties Colligative qualities are those that are dependent on the number of solute particles present in a solution regardless of their type |
|
LECTURE NOTES - VSSUT
COLLIGATIVE PROPERTIES: A dilute solution is one in which the amount of the solute is very small in comparison to the amount of the solvent |
What are the short notes of solution and colligative properties?
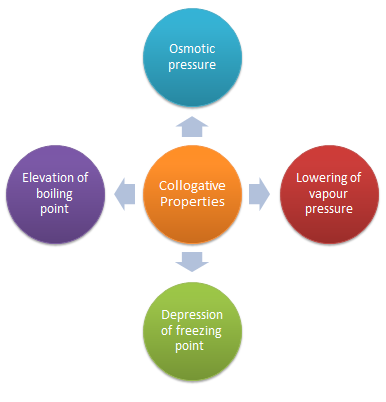
A colligative property is the property of a solution that depends only on the number of solute particles and not on their nature. Colligative properties of a solution include relative lowering of vapour pressure, elevation of boiling point, depression of freezing point, and osmotic pressure.What are the colligative properties of solution Class 12?
The four colligative properties that can be exhibited by a solution are given below:
Boiling point elevation.Freezing point depression.Relative lowering of vapour pressure.Osmotic pressure.What is a solution Class 12 chemistry Chapter 2?
Solution is a homogeneous mixture of two or more substances in same or different physical phases. The substances forming the solution are called components of the solution. On the basis of number of components a solution of two components is called binary solution. Solute and Solvent.- Colligative properties : The properties of solutions that depend only on the total number of soluble particles (molecules or ions), and not on nature of solute particles in solution are called Colligative properties.
|
Chemistry Notes for class 12 Chapter 2 Solutions - Ncert Help
Colligative properties are those properties which depends only upon the number of solute particles in a solution irrespective of their nature Relative Lowering of |
|
Solutions - NCERT
studying the properties of the solutions, like vapour pressure and colligative properties We will begin with 12 × 2 + 1 × 6 + 16 × 2 = 62 g mol–1 Moles of C2H6O2 Another important class of solutions consists of solids dissolved in liquid, for |
|
Solution And Colligative Properties
6 avr 2020 · Notes: The solution: (1) liquid in gas (2) Solid in DGT MH –CET 12th CHEMISTRY Study Material 1 Types of colligative properties: 1 |
|
Solutions and Colligative Properties
Depending upon the amount of solute dissolved in a solvent we have the following types of solutions: Unsaturated solution A solution in which more solute can be |
|
Solutions and Colligative Properties - Saptarshi Classes
Concentration of solutions: It is defined as the amount of solute dissolved in specific amount of solvent Dilute solutions: The solutions containing relatively less |
|
Colligative Properties - Patna Science College
The law states that partial vapour pressure of a solvent in a solution is equal to the vapour pressure of the pure solvent multiplied by its mole fraction in the solution |
|
Class 12th Chemistry Notes: Solutions (Part – III) Colligative
Colligative properties are those properties which depends only upon the number of solute particles in a solution irrespective of their nature Relative Lowering of |
|
LECTURE NOTES - VSSUT
Colligative properties are the properties of only dilute solution which are supposed to 12 m : molality of the solution Molal boiling point elevation constant or |
|
Lecture 4: Colligative Properties
By definition a colligative property is a solution property (a property of mixtures) for which it is the amount of solute dissolved in the solvent matters but the kind of |
|
Che Solution 12 Notes - DITP
Download CBSE class 12th revision notes for chapter 2 Solutions in PDF format Class 12 Chemistry Notes PDF - NCERT Solutions Colligative properties |






















![PDF]DOWNLOAD IIT JEE handwritten notes (Chemistry) PDF]DOWNLOAD IIT JEE handwritten notes (Chemistry)](https://www.kopykitab.com/blog/wp-content/uploads/2020/02/CBSE-Class-12-Chemistry.png)







