ISO 10993 – 1
|
ISO 10993-1 BIOLOGICAL EVALUATION – THE RISK
2017. 4. 10. Guideline/Standard. Scope. Description. ISO 10993-1. Medical devices. Biocompatibility evaluation and testing. ISO 10993-17. Medical devices. |
|
ISO 10993-1 “THE EARLY YEARS”
Page 1. ISO 10993-1 “THE EARLY YEARS”. JAMES M ANDERSON MD |
|
Update On Iso 10993 Nelson Labs
“Use of International Standard ISO 10993-1. 'Biological evaluation of medical devices - Part. 1: Evaluation and testing within a risk management process'” |
|
Statement regarding Use of ISO 10993-1:2009 Biological
2015. 10. 2. This document was produced by the Intemational Medical Device Regulators Forum. There are no restrictions on the reproduction or use of this ... |
|
위험관리 기반 의료기기 생물학적 평가 가이드라인[민원인 안내서]
2022. 12. 19. 이 안내서는 ISO 10993-1에 따라 위험관리 기반 의료기기 생물학적 평. 가에 대하여 알기 쉽게 설명한 것입니다. 본 안내서는 대외적으로 법적 효력을 ... |
|
Nelson Labs
[Based on ISO 10993-1:2018 and FDA 2016 Guidance on ISO 10993-1]. Device Categories. Contact Duration. Body Contact. Intact Skin. Biological Effect. ABC. ABC. A |
|
ISO-10993-1-2009.pdf
ISO 10993-1 was prepared by Technical Committee ISO/TC 194 Biological evaluation of medical devices. This fourth edition cancels and replaces the third edition |
|
위험관리 기반 의료기기 생물학적 평가 가이드라인[민원인 안내서]
2021. 12. 21. 전신독성 평가가 [표 1]에 명시되어 있는 경우 생물학적 시험이나. 위험사정은(ISO 10993-11:2017 |
|
Use of International Standard ISO 10993-1 Biological evaluation of
2023. 9. 8. This document supersedes “Use of International Standard ISO 10993-1. "Biological evaluation of medical devices - Part 1: Evaluation and testing. |
|
Use of International Standard ISO 10993-1 Biological evaluation of
2020. 9. 4. This document supersedes “Use of International Standard ISO 10993-1. "Biological evaluation of medical devices - Part 1: Evaluation and testing. |
|
Use of International Standard ISO 10993-1 Biological evaluation of
4 thg 9 2020 This document supersedes “Use of International Standard ISO 10993-1 |
|
ISO-10993-1-2009.pdf
ISO 10993-1 was prepared by Technical Committee ISO/TC 194 Biological evaluation of medical devices. This fourth edition cancels and replaces the third edition |
|
Nelson Labs
1 "Tissue" includes tissue fluids and subcutaneous spaces [Based on ISO 10993-1:2018 and FDA 2016 Guidance on ISO 10993-1]. |
|
ISO 10993-1
STANDARD. ISO. 10993-1. Fourth edition. 2009-10-15. Biological evaluation of medical devices —. Part 1: Evaluation and testing within a risk. |
|
?ISO 10993-1:2018?
ISO 2018. Biological evaluation of medical devices —. Part 1: 10993-1. Fifth edition. 2018-08. Reference number. ISO 10993-1:2018(E). Corrected version. |
|
INTERNATIONAL STANDARD ISO 10993-1
10993-1. Fifth edition. 2018-08. Reference number. ISO 10993-1:2018(E). Corrected version. 2018-10. iTeh STANDARD PREVIEW. (standards.iteh.ai). |
|
COMMISSION IMPLEMENTING DECISION (EU) 2020/438 of 24
24 thg 3 2020 EN ISO 10993-1:2009. Biological evaluation of medical devices - Part 1: Evaluation and testing within a risk management process. |
|
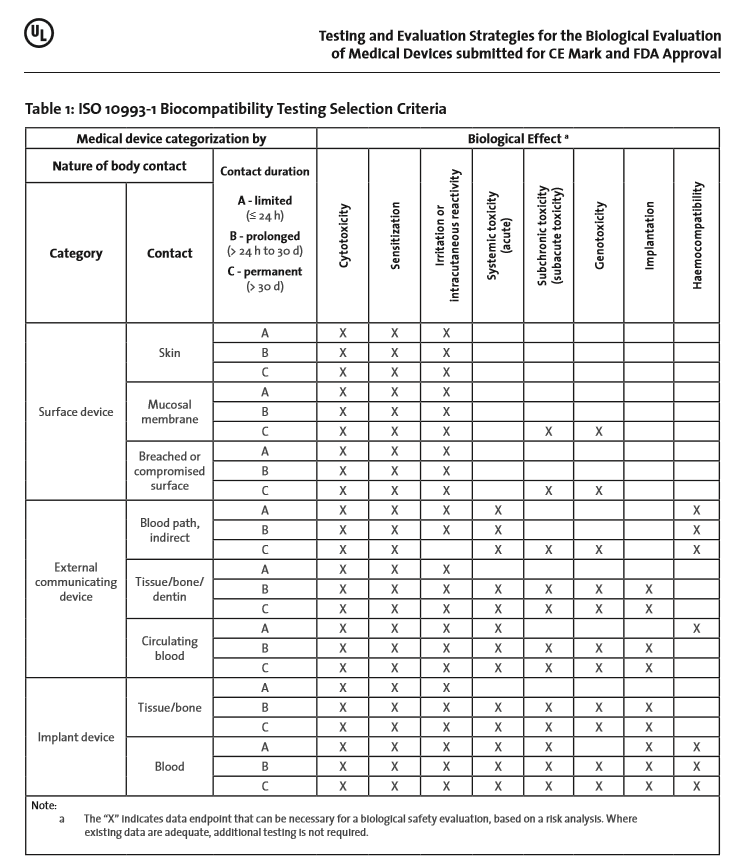
Biocompatibility Testing Matrix
Test for Consideration (Based on ISO 10993-1:2018 & FDA 2016 Guidance on ISO 10993-1). DEVICE CATEGORY. BIOLOGICAL EFFECTS. Body Contact. Contact Duration. |
|
Biological Evaluation of Medical Devices – Assessment of
In accordance with ISO 10993-1:2018 a medical device or material is biocompatible when it is able “to perform with an appropriate host response in a specific |
|
Commission communication in the framework of the implementation
18 thg 1 2011 EN ISO 10993-1:2009. Biological evaluation of medical devices —. Part 1: Evaluation and testing within a risk. |
|
ISO-10993-1-2009pdf
ISO 10993-1 was prepared by Technical Committee ISO/TC 194 Biological evaluation of medical devices This fourth edition cancels and replaces the third edition |
|
ISO 10993-1:2018(fr) Évaluation biologique des dispositifs médicaux
pdf [36] ISO/TR 10993-19 Évaluation biologique des dispositifs médicaux — Partie 19: Caractérisations physicochimique |
|
ISO 10993-1:2018(en) Biological evaluation of medical devices
This document was prepared by Technical Committee ISO/TC 194 Biological and clinical evaluation of medical devices This fifth edition cancels and replaces the |
|
INTERNATIONAL STANDARD ISO 10993-1
ISO 10993-1:2018(E) ISO 10993-3 Biological evaluation of medical devices — Part 3: Tests for genotoxicity carcinogenicity and reproductive toxicity |
|
Use of International Standard ISO 10993-1 Biological evaluation of
4 sept 2020 · This document specifically covers the use of ISO 10993-1 but also is relevant to other biocompatibility standards (e g other parts of the ISO4 |
|
NORME INTERNATIONALE ISO 10993-1 - iTeh Standards
10993-1 Cinquième édition 2018-08 Numéro de référence ISO 10993-1:2018(F) Version corrigée 2018-10 iTeh STANDARD PREVIEW (standards iteh ai) |
|
ISO-10993-1-2018pdf - iTeh Standards
ISO 2018 Biological evaluation of medical devices — Part 1: 10993-1 Fifth edition 2018-08 Reference number ISO 10993-1:2018(E) Corrected version |
|
Analyse de la nouvelle ISO 10993-1 (2018) - Biom Advice
La nouvelle norme ISO 10993-1 (2018) « Évaluation biologique des dispositifs médicaux - Partie 1: Évaluation et essais au sein d'un processus de gestion du |
|
ÉVALUATION BIOLOGIQUE DES DISPOSITIFS MÉDICAUX SELON
DES DISPOSITIFS MÉDICAUX SELON LA NORME ISO 10993-1 A INTRODUCTION Ce guide est applicable à tous les dispositifs médicaux quels que soient leur classe |
|
Évaluation biologique des DM : analyse de la nouvelle ISO 10993-1
de la nouvelle ISO 10993-1 (2018) Aurélien Bignon directeur de BioM Advice membre du réseau DM Experts Si la révision de la norme ISO 10993-1 ne |
|
ÉVALUATION BIOLOGIQUE DES DISPOSITIFS - Biom Advice
La nouvelle norme ISO 10993-1 (2018) « Évaluation biologique des dispositifs médicaux - Partie 1: Évaluation et essais au sein d'un processus de gestion du |
|
ISO 10993-1
ISO 10993-1 was prepared by Technical Committee ISO/TC 194, Biological evaluation of medical devices This fourth edition cancels and replaces the third edition |
|
Foire Aux Questions - Webinars LNE
Biocompatibilité : Comment démontrer la maîtrise du risque biologique selon la norme NF EN ISO 10993-1 (2010) ? Juin 2017 Page 2 Biocompatibilité : |
|
Analyse de la nouvelle ISO 10993-1:2018 - DM Experts
Aurélien Bignon, directeur de BioM Advice, membre du réseau DM Experts Si la révision de la norme ISO 10993-1 ne révolutionne pas le processus d'évaluation |
|
INTERNATIONAL STANDARD ISO 10993-1
ISO 10993-1:2018(E) ISO 10993-3, Biological evaluation of medical devices — Part 3: Tests for genotoxicity, carcinogenicity and reproductive toxicity |
|
Applying the New ISO 10993 - Nelson Labs
US FDA guidance document “Use of International Standard ISO 10993- 1, ' Biological evaluation of medical devices - Part 1: Evaluation and testing within a risk |
|
ISO 10993-1 BIOLOGICAL EVALUATION - Society of Toxicology
10 avr 2017 · FDA guidance (2016) on use of ISO 10993-1 – Highlights – Risk management framework • Biologics in delivery device systems – Challenges |





![ISO 10993-1 Biocompat Testing - [PDF Document] ISO 10993-1 Biocompat Testing - [PDF Document]](https://webstore.ansi.org/cover-pages/small/ISO/ISO+10993-5-1999.jpg)





























![Biocompatibility FDA and ISO 10993 - [PDF Document] Biocompatibility FDA and ISO 10993 - [PDF Document]](https://image.isu.pub/160525051124-b1a037279517bbba758eeea35a7aa86e/jpg/page_1.jpg)


