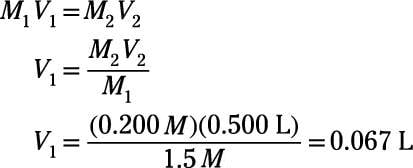solution dilution calculator percentage
|
Medical Calculations
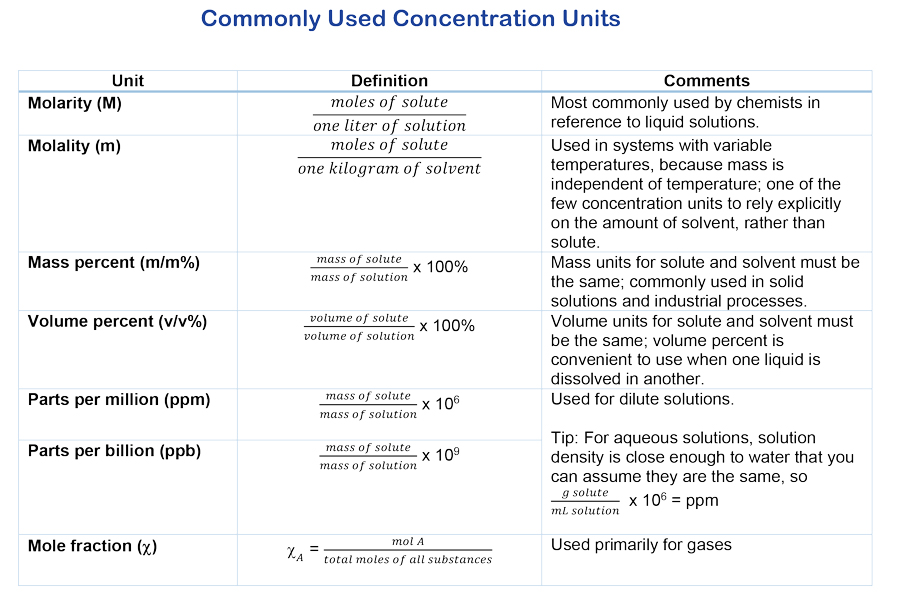
Medical Calculations. Dilutions and Solutions (Cont.) When calculating the percent strength of a solution (w/v) use the following formula:. |
|
Dilution of solutions for nurses
Similarly for a percentage stock strength solution the equation will be as in the following example. Example Calculate the amount of. (i) stock solution |
|
Pharmaceutical calculation
16-07-2021 If an injection containing a medication 50 mg/10 mL |
|
This is the table of contents and a sample chapter from WSO Water
Moles and Molarity. 6. Equivalent Weights and Normality. 7. Dilution Calculations. 8. Standard Solutions. 12. Chapter 2 Operator Math. |
|
Calculating Liquid Chemical Dilutions
Example No. 1 - Dry Powder-weight Dilutions. Assume desired concentration of citric acid solution is 15 percent (%). Concentration of stock dry powder. |
|
(2) Preparation and Dilution of Solutions
Understanding how to prepare solutions and make dilutions is an essential skill for biochemists which W/V% ? Weight/Volume Percentage Concentration. |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
The formula below is a quick approach to calculating such dilutions where: V = volume C = concentration; in whatever units you are working. (source solution |
|
Laboratory Math II: Solutions and Dilutions
When making dilutions you will know three of the four variables from the equation C1V1 equals C2V2. You will know your initial concentration |
|
DRUG CALCULATIONS 2
Using this to calculate the percentage of an adult dose should only be used intermediary solution such that when the intermediary solution is diluted 1 ... |
|
Conserve O Gram Volume 6 Issue 4: Making Percent Solutions of
Making Percent Solutions Of Chemicals while submerged in a strongly alkaline solution. ... To use non-metric quantities first calculate. |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS - MGEL
The formula below is a quick approach to calculating such dilutions where: V = volume C = concentration; in whatever units you are working (source solution |
|
PHARMACY CALCULATIONS MULTIPLE DILUTIONS - Mathcentre
Calculating the amount of substance needed in an initial concentration to produce a final desired dilution and volume Example 1 |
|
Solution Dilution Calculator - Sigma-Aldrich
This solution dilution calculator tool calculates the volume of stock concentrate to add to achieve a specified volume and concentration using the formula |
|
(2) Preparation and Dilution of Solutions
how to calculate the concentration of the diluted solutions if they unknown ? ? First: find the D F: Dilution factor (D F) = final volume / aliquot volume |
|
Laboratory Math II: Solutions and Dilutions
While you may already make solutions in the lab by following recipes we hope this Webinar will help you understand the concepts involved so that you can |
|
Calculating Dilutions and Site Size
You want to make 100 gallons of a 1 percent spray using water as the diluent You have a 2 EC formu- lation (pesticide label tells you that this is 2 pounds |
|
How to Make Simple Solutions and Dilutions
Ib conrert ftom solution to molerity multiply the solution by l0 to exprcss the percent solution Siams/L then divide by the formula rveight Molarit) = |
|
Dilution Conversions - Dosatron
Percentage Ratio Ounce = 29 57 Milliliters 1 Gallon = 3 79 liters Dilution Conversions 2090 Sunnydale Blvd Clearwater Florida 33765 USA |
|
Dilution Calculator Tocris Bioscience
The Tocris dilution calculator is a useful tool which allows you to calculate how to dilute a stock solution of known concentration |
|
Dilution Calculator - Molarity Percent - PhysiologyWeb
If you wish to perform dilution factor or fold dilution calculations for solutions with molarity or percent concentration units use our Dilution Factor |
How do you make a 2% solution?
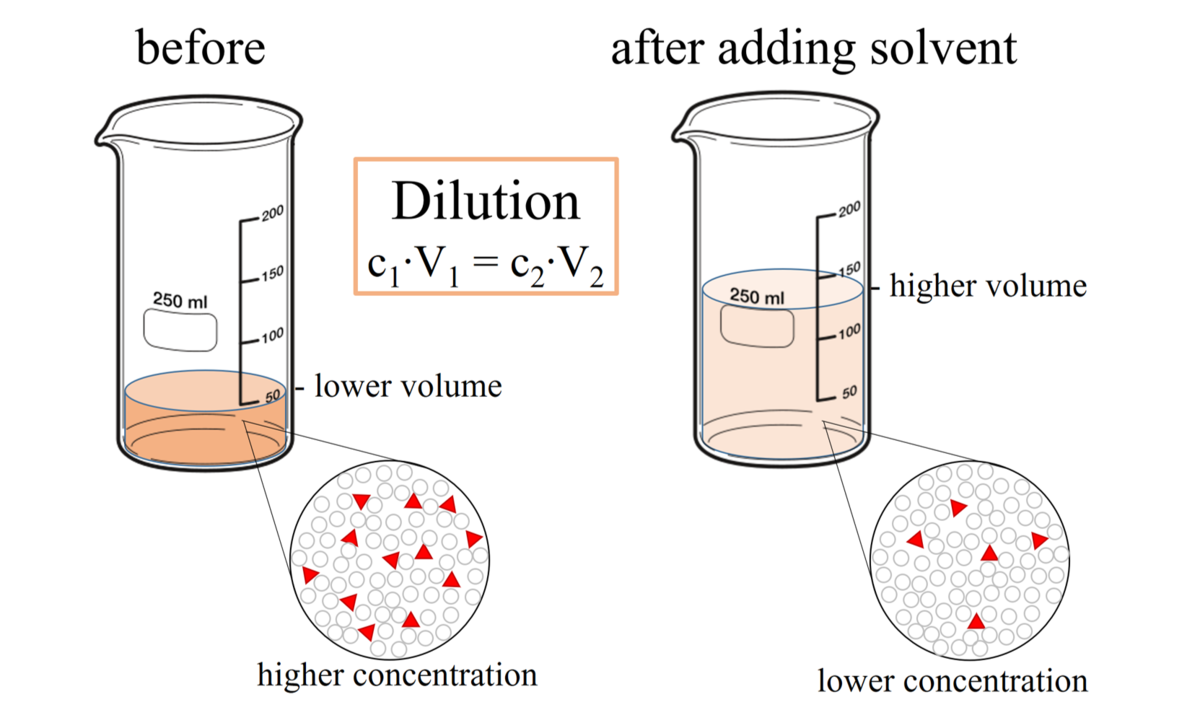
To make a proper 2 percent solution, they should specify a solvent but the general way to make a 2 percent solution is to get 2 grams of solvent and dissolve it in 98-100 ml of water.How do you dilute a 10% solution to a 1% solution?
You simply take 10?mL of the 10% solution, and dilute this up to a 100?mL volume with FRESH SOLVENT, i.e. a tenfold dilution that reduces the concentration to 1% .How do you dilute a 1% solution?
Measure the volume V1 of the solution with concentration C1. Then, add enough diluting liquid (water, etc.) to make a total volume V2. This new solution will have your desired concentration (C2).- So in this case, you would take 5mls (total volume) and divide by 1mls (the volume added). The result is 5, meaning that you added 1 part to a total of 5 parts… this is a 1 to 5 dilution or 1:5 dilution.
|
Diluting a % solution - Mathcentre
Calculating how much base to add to a product to achieve a solution to reduce it in strength to a 4 v/v solution? Method Step 1: percentages cancel out |
|
Dilution of solutions for nurses - Mathcentre
This leaflet explains how dilution calculations are performed When stating the strength of solution required it may be expressed in percentage strength, |
|
Solution dilution calculator percent
Solution dilution calculator percent Dilution Calculator of Molar Concentration Concentration: Volume: Stock Solution: mol mmol umol nmol m^3 L ml ul yd^3 |
|
Dilution and Concentration
Calculating the percentage or ratio strength of a solution made by diluting or con- If 50 mL of a 1:20 w/v solution of aluminum acetate is diluted to 1000 mL, |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
Anderson's How to Make Simple Solutions and Dilutions webpage) Mr Anderson's text below is a quick approach to calculating such dilutions where: mixed as dry mass (g) per volume where #g/100 ml = percent concentration A 10 |
|
Solution Dilution Equation - Free eBooks in the Genres you Love
oz) imperial gallon, liquid (gal) imperial pint (pt) litre Dilution Calculator - for percent solutions Solution:20 g NaCl / 100 g solution x 100 = 20 NaCl solution |
|
Concentrations and Dilutions INTRODUCTION
Calculate dilutions of stock solutions INTRODUCTION Concentrations of many pharmaceutical preparations are expressed as a percent strength This is an |






/volumetric-flask-containing-solution-of-potassium-dichromate--vi---k2cr2o7--and-other-flasks-of-transition-metal-salts--dry-chemicals-and-solutions--compounds-with-transition-metals-are-often-colored-702545755-5a579379eb4d520037b7a397.jpg)