solved problems on colligative properties pdf
|
Solutions and Colligative Properties Solved Problems
( ) The molality of the solution is greater than 5.5 . Page 3. 3. © 2020 Montogue Quiz g Problem 2.1. |
|
WORKSHEET:SOLUTIONS AND COLLIGATIVE PROPERTIES SET
Find the molarity of all ions in a solution that contains 0.165 moles of aluminum chloride in 820. ml solution. Answer: [Al 3+]= 0.201 M (Cl-] = 0.603M. 2. |
|
Colligative Properties Problem Set Solutions
26 янв. 1999 г. The density of this solution is 0.80 g mL-1. Identify the unknown based upon the following: a.The boiling point of this mixture is 78.8 °C ... |
|
Colligative Properties Chemistry Questions with Solutions.docx
As a result of the lower van't hoff factor(i) value C6H12O6 has a higher vapour pressure. Q-2: 1 molal aqueous solution of an electrolyte A2B3 is ionised 60%. |
|
CHEMISTRY COLLIGATIVE PROPERTIES WORKSHEET ∆Tf = i Kf
molecules in a solution and not on the nature of those atoms ions or molecules. Freezing point depression and boiling point elevation are examples of |
|
Leep502.pdf
(iv) Phenol and aniline. 8. Colligative properties depend on ______. (i) the nature of the solute particles dissolved in solution. (ii) |
|
REACT Strategy: Efforts to Link Concept Colligative Properties
Colligative properties of solutions are one of the chemical concepts with broad appli- cations in solving everyday life problems. For example the use of the |
|
Colligative Property Problems
pressure of a solution containing 24.6 g of camphor (C10H16O) dissolved in 100. mL of benzene. The density of benzene is 0.877 g/mL. Xben = nben/(nben + |
|
Untitled
on the NUMBER of solute particles in solution and NOT the KIND of particle. These particles can be molecules or ions or both! Colligative properties include. :. |
|
CHEMISTRY 142 – Example Problems Solutions and Colligative
Solutions and Colligative Properties. To be taken up in class or solutions insulin if 20.0 mg is dissolved in 10.0 mL of solution to give a resulting osmotic ... |
|
WORKSHEET:SOLUTIONS AND COLLIGATIVE PROPERTIES SET
ml solution. Answer: [Al 3+]= 0.201 M (Cl-] = 0.603M. 2. Find the molarity of each ion present after |
|
Colligative Properties Problem Set Solutions
26 janv. 1999 The density of this solution is 0.80 g mL-1. Identify the unknown based upon the following: a.The boiling point of this mixture is 78.8 °C. b. |
|
Colligative Property Problems
pressure of a solution containing 24.6 g of camphor (C10H16O) dissolved in 100. mL of benzene. The density of benzene is 0.877 g/mL. Xben = nben/(nben + ncam). |
|
Acces PDF Colligative Properties Of Solutions Worksheet Answers
Murphy 2020-03-23. Thermodynamics Problem Solving in Physical Chemistry: Study Guide and Map is an innovative and unique workbook that guides physical chemistry |
|
SOLUTIONS & COLLIGATIVE PROPERTIES
Which solute will show greater elevation in boiling point and Why? Solution. 2 - MARK QUESTIONS. 13. Examine the following illustrations and answer the |
|
Read Free Chemistry Colligative Properties Practice Questions And
get the. Chemistry Colligative Properties Practice Questions And Answers member that we have the funds for here and check out the link. You could purchase guide |
|
Read Online Colligative Properties Of Solutions 163 (PDF) - covid19
Chemistry I practice problems online! As more and more students enroll in chemistry courses |
|
16.4 Calculations Involving Colligative Properties
32. Practice Problems Plus. Calculate the mole fraction of each component in a solution of 42 g. CH3OH |
|
File Type PDF Colligative Properties Of Solutions 163 [PDF
With problem-solving shortcuts and lots of practice exercises you'll build your chemistry skills and improve your performance both in and out of the |
|
Bookmark File PDF Solution And Colligative Property Related Mcq
Practice Citric Acid Cycle MCQ book PDF with answers test 4 to solve MCQ questions bank: Acetyl COA production |
|
WORKSHEET:SOLUTIONS AND COLLIGATIVE PROPERTIES SET A
WORKSHEET:SOLUTIONS AND COLLIGATIVE PROPERTIES SET A: 1 Find the molarity of all ions in a solution that contains 0 165 moles of aluminum chloride in 820 |
|
Colligative Properties Problem Set Solutions - Widener University
26 jan 1999 · The density of this solution is 0 80 g mL-1 Identify the unknown based upon the following: a The boiling point of this mixture is 78 8 °C b |
|
Example Problems Solutions and Colligative Properties To be taken
Example Problems Solns and Colligatives 2013 doc Solutions and Colligative Properties To be taken up in class or solutions will be posted |
|
Solutions and Colligative Properties Solved Problems
g Problem 2 1 A perchloric acid solution contains 520 g of HClO4 per liter of solution and the solution has a density of 1 32 g/cm³ True or false? 1 |
|
Solution And Colligative Properties
6 avr 2020 · Solution:A solution is a homogeneous mixture of two or more non reacting substances on molecular level Solute and Solvent: The contituent |
|
Leep502pdf - NCERT
(iv) Phenol and aniline 8 Colligative properties depend on ______ (i) the nature of the solute particles dissolved in solution (ii) |
|
CHEMISTRY COLLIGATIVE PROPERTIES WORKSHEET ?Tf = i Kf
molecules in a solution and not on the nature of those atoms ions or molecules Freezing point depression and boiling point elevation are examples of |
|
Solutions and Colligative Properties - Saptarshi Classes
1 Dream Big Aim High Think Smart Solutions and Colligative Properties Introduction: solvent Types of solution: No Solute Solvent Examples 1 |
|
164 Calculations Involving Colligative Properties
32 Practice Problems Plus Calculate the mole fraction of each component in a solution of 42 g CH3OH |
|
Colligative Properties Chemistry Questions with Solutionsdocx
As a result of the lower van't hoff factor(i) value C6H12O6 has a higher vapour pressure Q-2: 1 molal aqueous solution of an electrolyte A2B3 is ionised 60 |
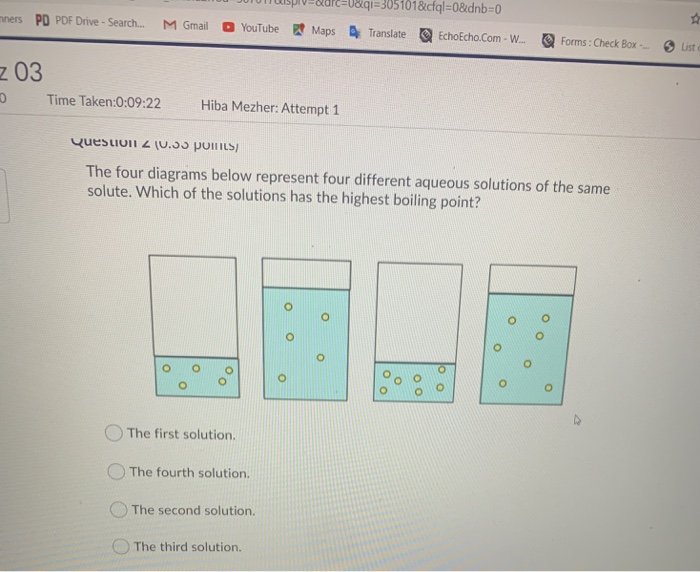
What are solved examples of colligative properties?
4) What is the boiling point of a solution that contains 1.25 mol CaCl2 in 1400 g of water? The solutions boiling point is elevated 101.36 °C.What is the boiling point of a solution that contains 1.25 mol CaCl2 in 1400g of water?
Colligative properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.What are 4 examples of colligative properties?
Colligative Properties Equations
The equations for the four colligative properties are as follows. Tf=?iKfm T f = ? i K f m where Tf is the change in freezing point of the solution, Kf is a constant, and m is the molality of the solution measured in kg/mol.
|
Example Problems on Solutions and Colligative Properties
Example Problems Solns and Colligatives 2013 doc Solutions Osmotic pressure is a colligative property that can be used to determine the molar mass of an |
|
WORKSHEET:SOLUTIONS AND COLLIGATIVE PROPERTIES SET
Find the molarity of all ions in a solution that contains 0 165 moles of aluminum chloride in 820 ml solution Answer: [Al 3+]= 0 201 M , (Cl-] = 0 603M 2 Find the |
|
Solutions - NCERT
pressure and colligative properties We will begin with Solvent determines the physical state in which solution exists One or Common Examples Gaseous |
|
164 Calculations Involving Colligative Properties
•Laboratory Manual, Lab 33 Technology 16 4 1 Solve problems related to the molality and mole fraction of a solution 16 4 2 Describe how Recall that colligative properties depend only upon solute concentration The unit molality and |
|
Colligative Properties Problem Set, Solutions - Widener University
26 jan 1999 · A mixture of the unknown is prepared by mixing 10 0 grams of unknown and diluting to 250 mL with ethanol The density of this solution is 0 80 g |
|
Colligative Property Problems
Colligative Property Problems Vapor pressure C Calculate the vapor pressure of a solution containing 24 6 g of camphor (C10H16O) dissolved in 100 mL of |
|
Chapter 13 Properties of Solutions Classification of Matter
Calculate the boiling point elevation and freezing point depression of a solution ➢ Calculate the osmotic pressure of a solution ➢ Use colligative properties of |
|
Lecture 4: Colligative Properties
By definition a colligative property is a solution property (a property of mixtures) Note that in the problem above, we determined the reduction in vapor pressure |
|
C:\Users\1\Desktop\Solution and colligative pdf\1xps - MT Educare
solution EVALUATION PAPER - SOLUTIONS AND COLLIGATIVE PROPERTIES Time : 30 Min Marks : 25 Page 41 ≥ π π Page 42 1 d 2 c 3 a |