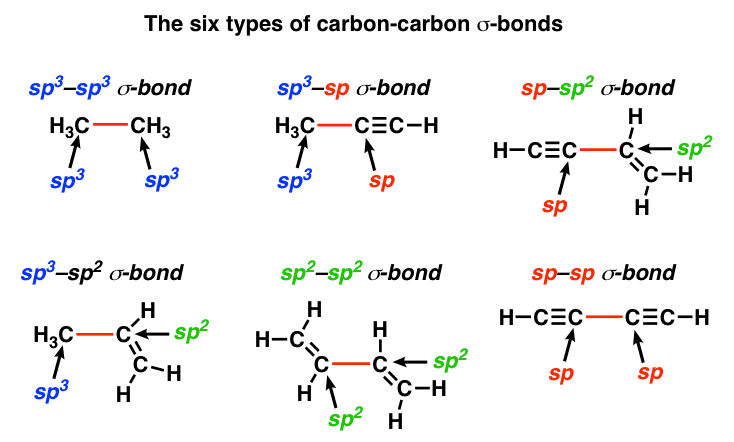
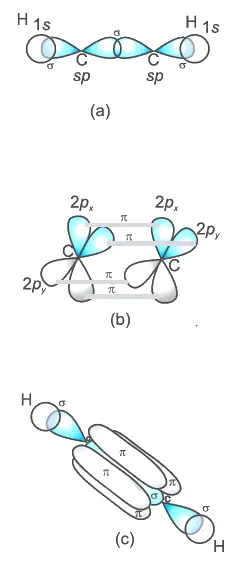
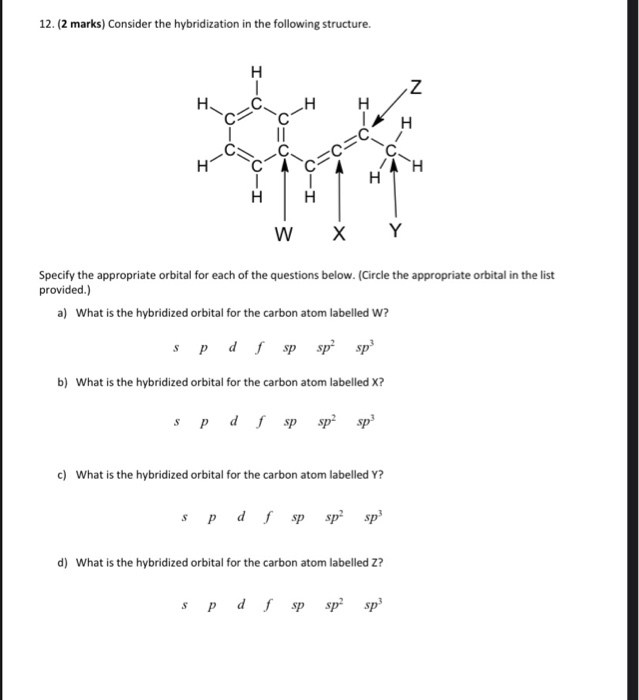
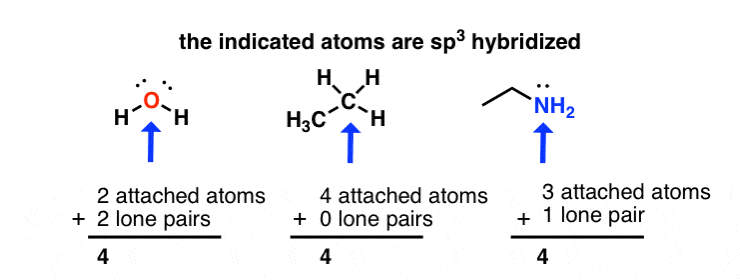
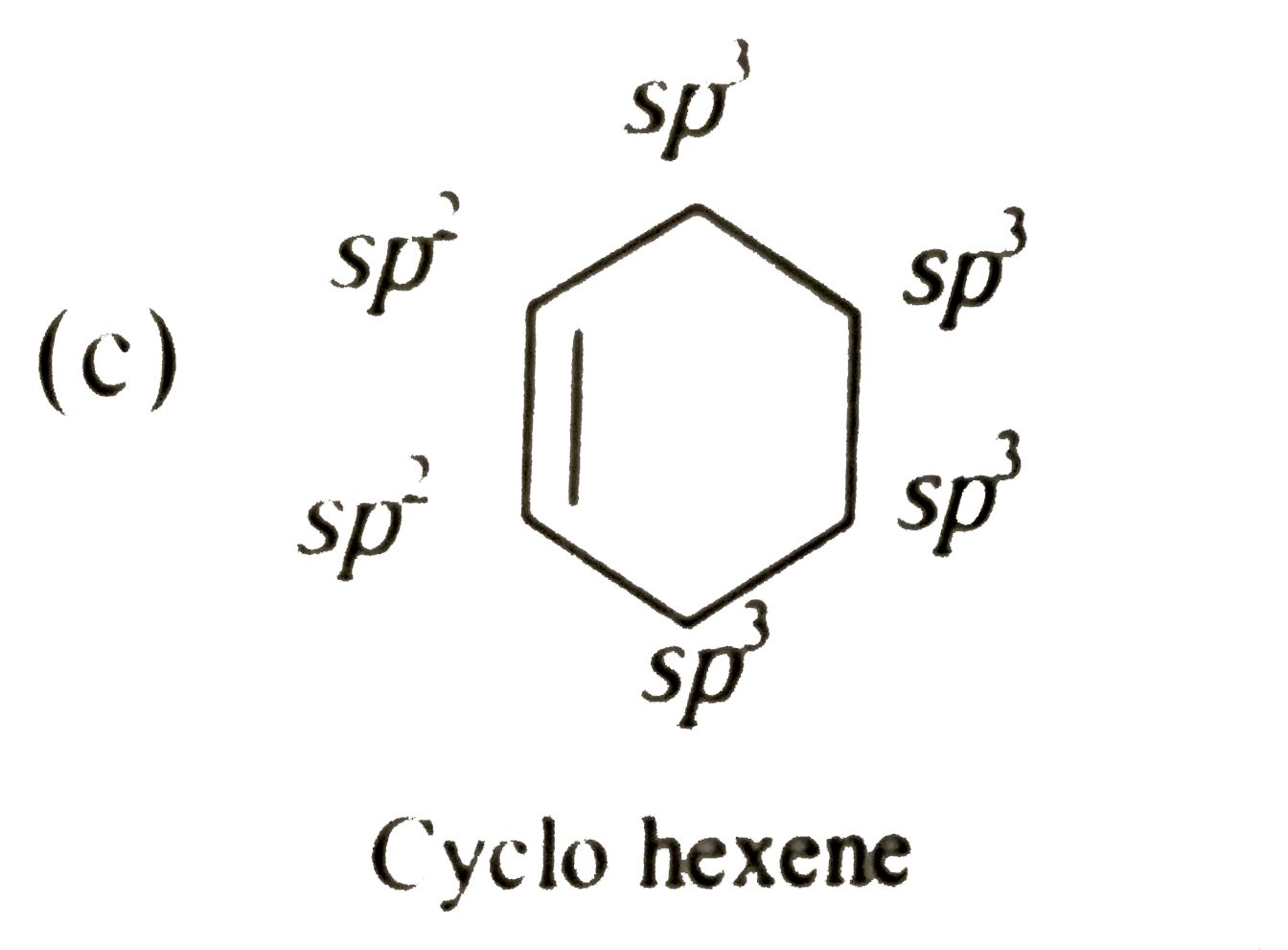
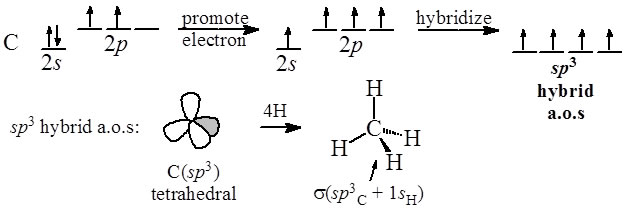
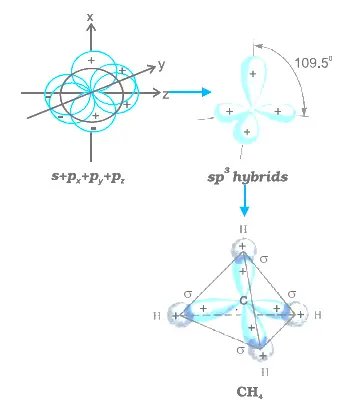
sp2 hybridized carbon
|
Carbon-Carbon bonds: Hybridization
2011. 5. 5. In the following sp3- and sp2- hybridization will be explained in detail. 2.1 sp3-Hybridization. Figure 1: [1] sp3-hybrid orbital. A carbon ... |
|
Sp2/sp3 Hybridized Carbon as an Anode with Extra Li-Ion Storage
2020. 7. 21. The sp2/sp3 hybridized orbital design concept will help to develop advanced electrode materials. ? INTRODUCTION. Lithium-ion batteries (LIBs) ... |
|
Sp2-Hybridized Carbon-Containing Block Copolymer Templated
2019. 3. 4. amphiphilic block copolymers with high content of sp2-hybridized carbon in the hydrophobic segments that were relatively stable and could be. |
|
The Nature of Interlayer Binding and Stacking of sp-sp2 Hybridized
Hybridized Carbon Layers: A Quantum Monte Carlo Study layer bindings of sp- and sp2-bonded carbon networks are significantly underestimated. |
|
PCF-Graphene: A 2D sp2-Hybridized Carbon Allotrope with a Direct
2019. 1. 30. a COT-based 2D carbon allotrope PCF-graphene |
|
Single-site pyrrolic-nitrogen-doped sp2-hybridized carbon materials
Integrating nitrogen species into sp2-hybridized carbon materials has proved an efficient means to improve their electrochemical performance. |
|
The Nature of Interlayer Binding and Stacking of sp-sp2 Hybridized
Hybridized Carbon Layers: A Quantum Monte Carlo Study layer bindings of sp- and sp2-bonded carbon networks are significantly underestimated. |
|
A sp2+sp3 hybridized carbon allotrope transformed from AB
2018. 1. 30. New carbon allotropes can be designed by combining sp sp2 and sp3 three hybridiza- tion states. And the hybridization states or ... |
|
The Nature of Interlayer Binding and Stacking of sp-sp Hybridized
2017. 8. 2. ?-graphyne is a two-dimensional sheet of sp-sp2 hybridized carbon atoms in a hon- eycomb lattice. While the geometrical structure is similar ... |
|
First-principles investigation on the bonding mechanisms of two
2020. 12. 9. sp3- sp2- and sp-hybridized carbon atoms owing to the versatile. exibility of carbon atom. When the sp2 carbon bonds in gra-. |
|
Carbon-Carbon bonds: Hybridization
5 mai 2011 · For carbon the most important forms of hybridization are the sp2- and sp3- hybridization Besides these structures there are more possiblities |
|
Hybridization
Dr Solomon Derese 149 sp2 hybridization of a carbon ground state excited state sp2 hybridized state 2p z sp2 2s 2p 2s 2p unhybridized p orbital |
|
Hybridization - Organic Chemistry Second Edition
The carbon has three sp2 hybridized orbitals and can form three ? bonds one of which is to the oxygen The oxygen has one sp2 orbital which is used in the ? |
|
Review of an Alkene Two sp2 hybridized carbon atoms 1 ? bond 83
Two sp2 hybridized carbon atoms 1 ? bond 83 Kcal/mol 1 ? bond 63 Kcal/mol The carbon-carbon double bond prevents this rotation because the ? bond |
|
CHAPTER 14 COVALENT BONDING: ORBITALS
Assuming all atoms are hybridized the carbon and oxygen atoms are sp2 hybridized and the two chlorine atoms are sp3 hybridized The two C?Cl ? bonds are |
|
(PDF) UNIT-II: sp 2 hybridization in alkenes and sp - ResearchGate
4 fév 2021 · PDF Hybridization:-Hybridization and general principles explain how covalent bonding in The sp2 hybridization of carbon orbital |
|
(PDF) Sp2- and sp3-hybridized carbon materials as catalysts for
PDF The catalytic properties of sp2- and sp3-hybridized carbons represented by graphene and diamond in the selective oxidation of cyclohexane were |
|
Hybrid sp2+sp3 carbon phases created from carbon nanotubes
These hybrid phases consists of atoms in three - and four- coordinated (sp2+sp3-hybridized) states The initial structure of the carbon phases was constructed |
|
Chapter II Hybridization in hydrocarbons
%2520Shoubra/Engineering%2520Mathematics%2520and%2520Physics/2974/crs-13602/Files/ch2-hybridization.pdf |
|
1 The sp2 Nanocarbons: Prototypes for Nanoscience and
21 oct 2010 · The concept of sp2 hybridization where hybridization means the mixing of valence electronic states is presented here Carbon has six |
What is sp2 hybridized carbon?
Carbon - sp2 hybridization. A carbon atom bound to three atoms (two single bonds, one double bond) is sp2 hybridized and forms a flat trigonal or triangular arrangement with 120° angles between bonds.What is sp2 hybridized?
The sp2 hybridization is the mixing of one s and two p atomic orbitals, which involves the promotion of one electron in the s orbital to one of the 2p atomic orbitals. The combination of these atomic orbitals creates three new hybrid orbitals equal in energy-level.- A typical example of a sp2-hybridized crystal structure is graphite (see Fig. 4, left). It consists of parallel carbon layers. Within a layer the planar sp2-hybrid orbitals align themself to a structure with strong binding.5 mai 2011
|
Supplement 41: Electronegativity Effects of Hybridization The Effect
Actually, the electronegativity of carbon depends on its hybridization state Carbons that are sp2-hybridized are somewhat more electronegative (about 0 2 |
|
Carbon-Carbon bonds: Hybridization - FU Physik
5 mai 2011 · In the following sp3- and sp2- hybridization will be explained in detail 2 1 sp3- Hybridization Figure 1: [1] sp3-hybrid orbital A carbon atom |
|
ORGANIC CHEMISTRY BY
three sp2 hybridized orbitals py pz Unhybridized p-orbital sp2 hybridization : one s-orbital and two p-orbitals Carbon atom of alkenes and aromatic compounds |
|
Chapter 14
atom is sp2 hybridized Two of the sp2 hydride orbitals, on the carbon, are used in C-H single bonds (σ bond) The C-H single bond is formed from the overlap of |
|
CHAPTER 14 COVALENT BONDING: ORBITALS
sp2 hybridization The two C−H sigma bonds are formed from overlap of the sp2 hybrid orbitals from carbon with the hydrogen 1s atomic orbitals The double |
|
Structural Analysis of Planar sp3 and sp2 Films: Diamond - CORE
The allotropes of carbon are characterized by the type of hybridized bonding forming its structure, ranging from pure sp2 as in graphene, carbon nanotubes and |