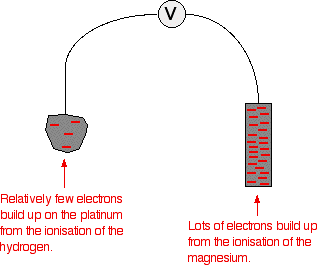
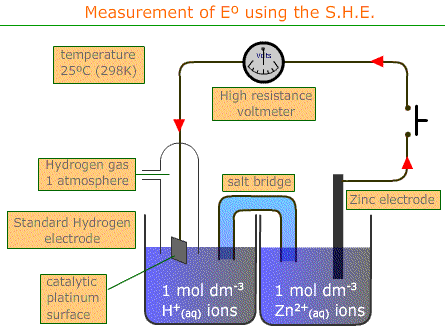
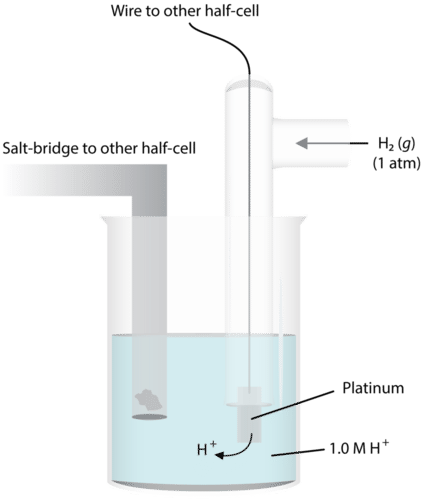
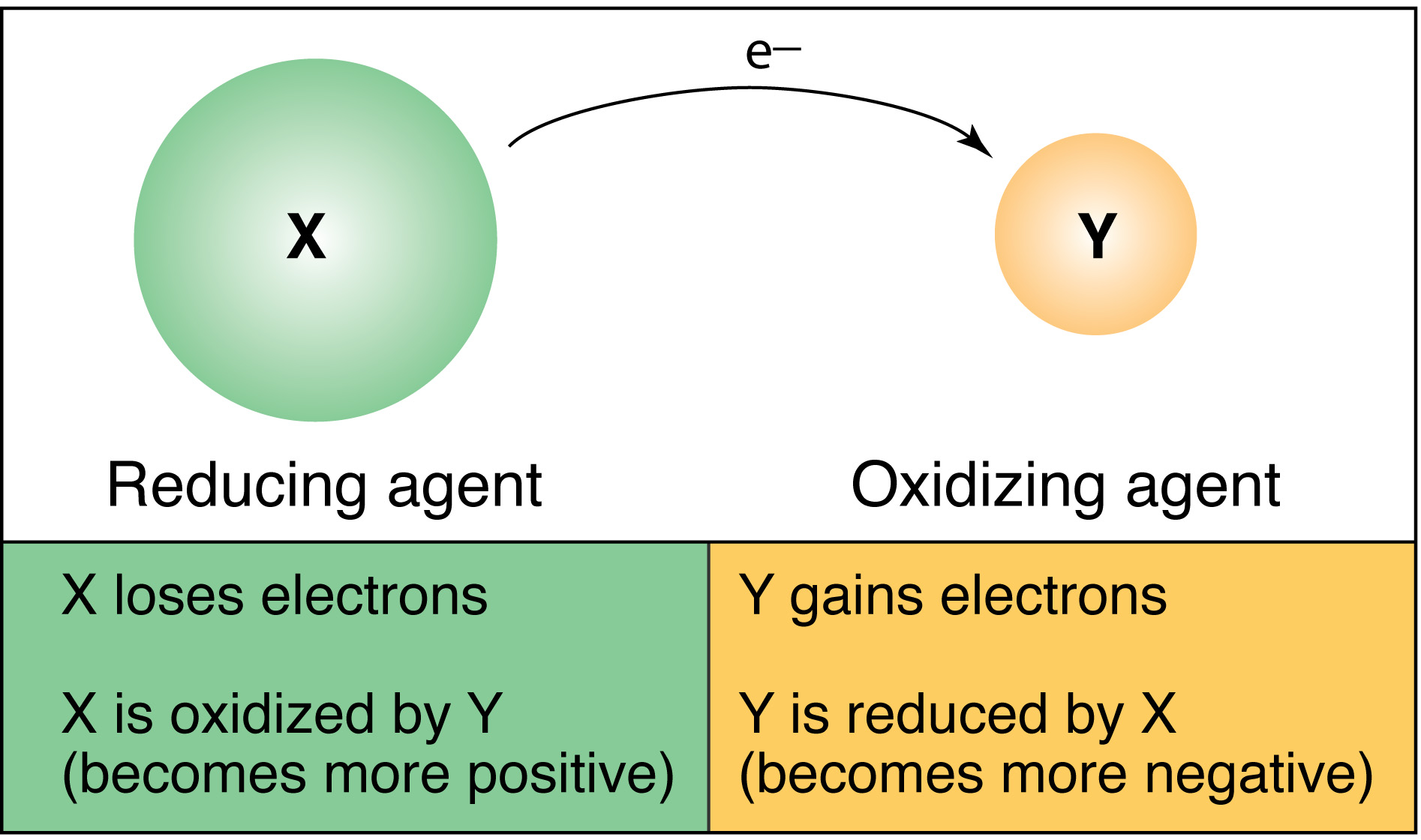
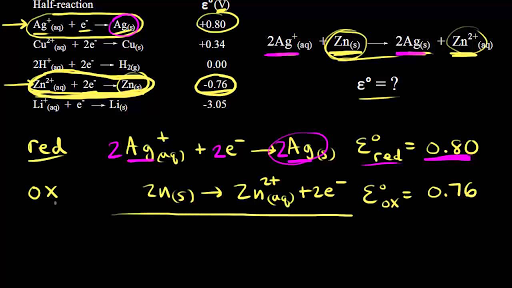standard cell potential problems
|
Test4 ch19 Electrochemistry Practice Problems.pdf
Using the Table of Standard Reduction Potentials table shown above what is the standard cell potential for an electrochemical cell that has iron (Fe) and |
|
Untitled
Ch.17- Electrochemistry: Practice Problems I Given that the standard reduction potential for Cr2O72?2C³ is 1.33 V what is E°Red for I2(aq)?. |
|
Ch 11 Practice Problems
The standard potential for the reaction Zn + 2Ag+ ? Zn2+ + 2Ag is 1.56 V. Given that the standard reduction potential for Ag+ + e– ? Ag is 0.80 V |
|
Electrochemical Cells Worksheet
Calculate the standard cell potential produced by a galvanic cell consisting of a nickel electrode in contact with a solution of Ni2+ ions and a silver |
|
Test4 ch19 Electrochemistry Practice-answers-Marked
Extra Practice Problems. Oxidation Numbers For the cell shown the standard reduction potentials are +0.80 V for Ag+ and –0.76 V for Zn2+. Based on the. |
|
Chapter 13 Cells and Batteries Solutions to Practice Problems 1
Aug 24 2007 Write the half reactions. Find the standard reduction potential values from Table 13.1. Calculate the standard cell potential using the equation ... |
|
CHEM1101 Answers to Problem Sheet 10 1. (a) 2Al(s) + 3Sn2+(aq
Electrons flow from the anode to the cathode. (c). 2Ag+(aq) + Pb(s) ? Ag(s) + Pb2+(aq). The standard cell potential E° = ((+0.80) + (+0.13)) V = 0.93 V. |
|
A.P. Chemistry Practice Test - Ch. 17: Electochemistry MULTIPLE
Feb 20 2018 19) The standard cell potential (Eecell) for the voltaic cell based on the ... on T. For temperature change in this problem |
|
?G° = -nFE°cell ?G° = -96.5nE°cell
Balance the reaction and find ?G° given the reduction potentials shown Caution: K values often work out to be enormous (calculator problems). |
|
Jasperse Electrochemistry Extra Practice Problems
Given the electrochemical reaction shown if the standard reduction potential of Zn2+ ? Zn is –0 76 V what is the standard reduction potential of Mg2+ ? Mg? |
|
Cell Potential
18 mar 2020 · The standard cell potential equals to the difference between the abilities of the two electrodes to act as reducing agents E° cell = E° |
|
Ch 11 Practice Problems - UCSB CLAS
The standard potential for the reaction Zn + 2Ag+ ? Zn2+ + 2Ag is 1 56 V Given that the standard reduction potential for Ag+ + e– ? Ag is 0 80 V |
|
Ch 17 Electrochemistry Practice Problems I - keypdf
Given that the standard reduction potential for Cr202 ?2C³ is 1 33 V what is E°Red for I2(aq)? +6 (+3) Ecell = {cattade = 8° gain Beti reduction |
|
Chapter 18: Electrochemistry
Standard cell potential : The cell potential under standard state conditions [ions] = 1 M T = 25°C 1 atm gas pressure Page 8 Zn(s) Zn2+ (1 M) Cu2+ (1 |
|
Exercise 182 - Standard Cell Potentials
DIRECTIONS: Write the oxidation and reduction half reactions Calculate the standard cell potential for the following electrochemical cells 3 Ag+(aq) + Fe(s) |
|
Electrochemical Cells Worksheet
Calculate the standard cell potential produced by a galvanic cell consisting of a nickel electrode in contact with a solution of Ni2+ ions and a silver |
|
Chapter 13 Cells and Batteries Solutions to Practice Problems 1
24 août 2007 · The balanced redox reaction is given Plan Your Strategy Write the half reactions Find the standard reduction potential values from Table 13 1 |
|
Electrochemical Cells Practice Problems
Calculate the standard cell potential produced by a voltaic cell consisting of a nickel electrode in contact with a solution of Ni2+ ions and a silver electrode |
|
Topic 9 Electrochemistry
23 jan 2018 · 17 2 Standard Reduction Potentials ? Reaction in galvanic cell is oxidation-reduction split into two half-reactions |
|
Test4 ch19 Electrochemistry Practice Problems
For the cell shown, the standard reduction potentials are +0 80 V for Ag+ and – 0 76 V for Zn2+ Based on the reduction potentials, the electrode is where the |
|
Ch 17 Electrochemistry Practice Problems I - keypdf
Write the net ionic equation for the overall reaction that occurs as the cell operates and calculate the value of the standard cell potential, Ecell c Determine the |
|
Ch 11 Practice Problems - UCSB CLAS
The standard potential for the reaction Zn + 2Ag+ → Zn2+ + 2Ag is 1 56 V Given that the standard reduction potential for Ag+ + e– → Ag is 0 80 V, determine the |
|
Electrochemistry example problems
25 avr 2017 · The hydrogen electrode is then buffered to have pH 5 57 Calculate the resulting cell potential The reduction potential of the standard hydrogen |
|
Solutions for Electrochemistry Problem Set
24 avr 2001 · From the table of reduction potentials, we can find Eanode0 7996 volt Ecathode0 3402 volt And then calculate the standard cell potential |
|
Nernst Equation Example Problems - These Pages Have Moved
1 mar 2002 · Since the Cu2+ concentration is less than under standard conditions, the equlibrium will shift to the left This means that the cell potential will be |
|
Electrochemistry
Standard Electrode Potentials It is impossible to Standard Reduction Potential 16 E o cell = E o cathode + E o anode Eo cathode See problem set for the |
|
The Standard Electrode Potential, E0 - İYTE
18B Electrochemical cells * An electrochemical cell consists of two conductors called electrodes, each of which is immersed in an electrolyte solution |
|
2 Electrochemistry
One of common problems, that will become be the solution to the problem: Calculate the standard cell potential of the Daniell cell, in which reaction (2 2) |
|
CH1101 General & Physical Chemistry 2012 Basic Electrochemistry
PROBLEM: Diagram, show balanced equations, and write the notation for a voltaic cell that consists Table 21 2 Selected Standard Electrode Potentials ( 298K) |