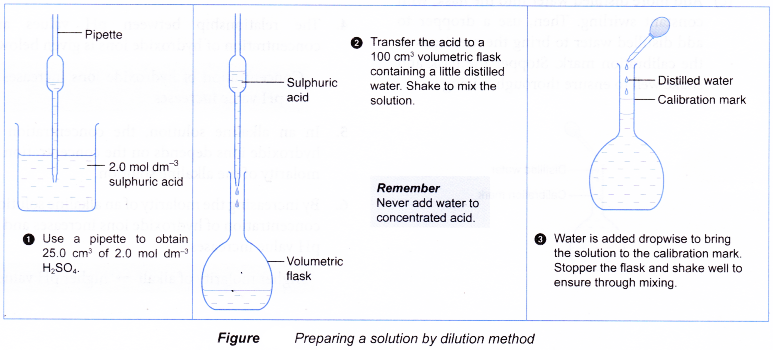
steps in preparing a standard solution
|
Laboratory Solution Preparation
Standard Solution: A very precise solution usually to 3–4 significant you through the steps of determining the correct procedure for making the solution. |
|
Solutions Preparation ‐ A solution that has a known accurate
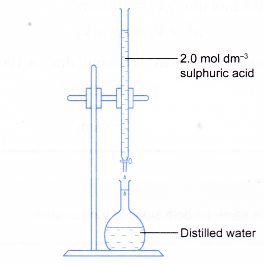
To make a standard solution using dilution follow the following steps. Step 1: Calculate the volume of the stock solution needed to make the solution. C1V1 = |
|
Preparation of Standard Solutions
Nov 20 2000 standard preparation logbook |
|
Practical notes - CP4 Preparation of a Standard Solution and
Prepare a standard solutionfrom a solid acid salt and find the concentration of a solution of. NaOH by titrationusing your prepared standard solution. |
|
Preparing Standard Sodium Hydroxide Solution*
Before we begin titrating that wine sample we have one more important step standardization of NaOH solution. Standardization simply is a way of checking |
|
Laboratory Solution Preparation
Standard Solution: A very precise solution usually to 3–4 significant you through the steps of determining the correct procedure for making the solution. |
|
EXPERIMENT 12 A: STANDARDIZATION OF A SODIUM
Therefore it is not possible to prepare a standard solution of sodium hydroxide of accurately known concentration by weighing NaOH. A sodium hydroxide |
|
Guidance for Preparing Standard Operating Procedures (SOPs)
Apr 6 2007 Where possible break the information into a series of logical steps to avoid a long list. The level of detail provided in the. SOP may differ ... |
|
A Guide to Titration
Repeat steps 1 2 |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
The source of dilution material for each step comes from the diluted material of the previous. Example 1: To prepare a liter of a simple molar solution from a ... |
|
Laboratory Solution Preparation
5. Divide the number of moles (step 4) by the volume in liters of the solution to find the molarity of the solution. Example |
|
Preparation of Standard Solutions
20 nov 2000 Follow step 4 to prepare matrix spike working solution using a separate source from that used for calibration. 6. Prepare pesticide calibration ... |
|
Preparation of Calibration Curves - A Guide to Best Practice
Any error in the preparation of the stock solution will propagate through the other standards leading to a bias in the calibration. A procedure sometimes used |
|
Making Standard Solutions
When making up a standard solution it is important that the correct mass of Work out the mass of substance needed using your answers from steps. |
|
Solutions Preparation ? A solution that has a known accurate
Preparing a standard solution from a solid. Step 1: Calculate what mass of solid is needed to make the required volume and concentration of solution. |
|
How to prepare standard solutions
- Use primary grade / analytical reagent grade chemical. - Molecular Weight (MW) of K2Cr2O7 = 294g. - weight of reagent for 1L of 0.0417M solution g. |
|
Thermo Scientific Orion ISE Calibration: Preparing Ammonia
Mix the solution well. 2. Prepare a 10 ppm ammonia standard by measuring 100 mL of the 100 ppm ammonia standard from Step 1 using a graduated cylinder. |
|
TITRIMETRIC ANALYSIS
(A) Preparation of 0.1M Standard Solution of Oxalic Acid. Follow the procedure as described in Experiment No. 2.1. (B) Titration of Sodium Hydroxide and Oxalic |
|
EXPERIMENT 12 A: STANDARDIZATION OF A SODIUM
It cannot be weighed accurately. Therefore it is not possible to prepare a standard solution of sodium hydroxide of accurately known concentration by weighing |
|
EZ1003 Ammonia Analyser
Method and reagent sheets Analysis method . ... Prepare a standard solution for calibration according to the following table: take accurately x. |
|
Preparation of Standard Solutions - CLU-IN
20 nov 2000 · Follow step 4 to prepare matrix spike working solution using a separate source from that used for calibration 6 Prepare pesticide calibration |
|
Preparation of Standard Solutions Theory:
Theory: the purpose of this experiment is Know Preparation of Standard Solutions from Solid Materials and Liquid Materials different concentrations |
|
Laboratory Solution Preparation
Determine concentration in percent by mass of the solute in solution Change to the decimal equivalent 3 Calculate the molar mass of the compound MM 4 |
|
Practical notes - CP4 Preparation of a Standard Solution and Titration
This is the standard solution Method - titration 1 Measure out 25 cm3 of the sodium hydroxide solution with an unknown concentration |
|
How to prepare standard solutions
This module describes procedure and a laboratory exercise for preparation of standard solutions Modules in which prior training is required to complete |
|
Preparation of standard solutions - Maddy Parsons - Academiaedu
In many cases it is difficult to make a choice as to what compound is best suited for these solutions No complete See Full PDF Download PDF See Full PDF |
|
Lab Report Preparation of Standard Solution - Titration - Scribd
Avis 43 |
|
Preparing a Standard Solution
To prepare a solution of known concentration from a solid solute we follow the steps outlined below 1 Calculate the mass of the solute required to |
|
Standard solutions
Such as preparation of approximately 0 1 M or 0 1 N HCl and standardising it with standard solution of Na2CO3 or Borax Standard solutions of Na2CO3 and |
What are the steps to prepare a standard solution?
A solution can be prepared by dissolving a known mass or volume of solute in a known amount of solvent. Concentration is expressed as grams of solute dissolved in one litre of solution. Example: Calculation for preparing 300 mL of a sucrose solution at a concentration of 5 g/L.What are the three steps in preparing a standard solution?
Solutions of known concentration can be prepared either by dissolving a known mass of solute in a solvent and diluting to a desired final volume or by diluting the appropriate volume of a more concentrated solution (a stock solution) to the desired final volume.
|
Preparation of Standard Solutions - CLU-IN
20 nov 2000 · Fill approximately one-tenth of the volumetric flask with deionized water; add an appropriate volume of concentrated nitric acid to produce 5 HNO3 at final volume; add the appropriate volume of the concentrated stock solution |
|
Solutions Preparation ‐ A solution that has a known, accurate
Preparing a standard solution from a solid Step 1: Calculate what mass of solid is needed to make the required volume and concentration of solution n = cv |
|
Laboratory Solution Preparation
Determine the mass in grams of one mole of solute, the molar mass, MMs Decide volume of solution required, in liters, V Decide molarity of solution required, M Dissolve 93 52 g of NaCl in about 400 mL of distilled water, then add more water until final volume is 800 mL |
|
3 PREPARATION OF A STANDARD SOLUTIONS Objectives Pre
To overview the most important concentration units and their use to calculate the mass of solid necessary to prepare a standard solution To practice the |
|
CP4 Preparation of a Standard Solution and Titration - Physics
Prepare a standard solutionfrom a solid acid salt and find the concentration of a solution of NaOH by titrationusing your prepared standard solution Method |
|
Preparation of standard solutions - Dairy Knowledge Portal
Molar solution is one, which contains one molecular weight of the reagent in one litre of the solution Molarity is expressed as M Normal solution Normal solution |

























/assortment-of-laboratory-glassware-flasks-680805969-59db9d15845b340012f6d3e5.jpg)
