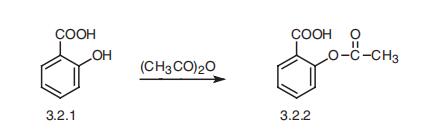synthesis of aspirin without acetic anhydride
|
8-Synthesis-of-Aspirin.pdf
If acetic anhydride is used instead of acetic acid the reaction is much faster and has a higher yield (since acetic anhydride is much more reactive than acetic |
|
A novel and facile one-pot green laboratory synthesis of aspirin
without using Acetic Anhydride. Malik Sarika and Ratnani Sonia. *. Ramjas This method embraces a safe and green synthesis due to absence of conventional ... |
|
An Efficient Microscale Procedure for the Synthesis of Aspirin
Jun 6 1998 The recommended procedures for the synthesis of aspirin use either acetic anhydride–concentrated sulfuric acid (1) or phosphoric acid (2) as ... |
|
5012 Synthesis of acetylsalicylic acid (aspirin) from salicylic acid
A mixture of 13.8 g (100 mmol) salicylic acid and. 12.8 g (11.9 mL 125 mmol) acetic anhydride is filled in the reaction flask and three drops of conc. |
|
Acetylation of alcohols phenols and salicylic acid by heteropoly
various of heteropoly acids in acetic anhydride as acetylating agent without solvent. Acetylation of salicylic acid gave acetyl salicylic acid or aspirin. |
|
Synthesis of Aspirin
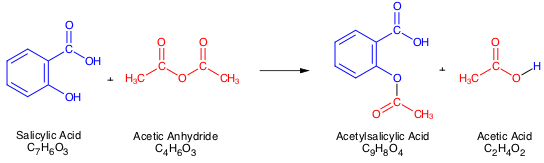
The by-product is acetic acid (Figure 5.1). +. →. +. H3PO4 acetic anhydride salicylic acid aspirin acetic |
|
The Synthesis of a Medicinal Agent- Aspirin By Walter Scharf and
A convenient way to obtain complete reaction in the preparation of an acetate ester is to use acetic anhydride. The reaction is catalyzed by concentrated |
|
Yin and Yang in Chemistry Education: The Complementary Nature
Aspirin was synthesized from the reaction of salicylic acid and acetic anhydride whereas wintergreen oil was made via the reaction of salicylic acid and |
|
SYNTHESIS OF ASPIRIN
The acetic anhydride will be added in excess so unreacted acetic anhydride will be present at the end of the reaction. In addition |
|
Chapter 5 Carboxylic Acids and Esters
Aspirin (acetylsalicylic acid). Produced by reacting salicylic acid with acetic anhydride giving a compound which is less acidic |
|
8-Synthesis-of-Aspirin.pdf
In this experiment the salicylic acid is the limiting reactant and the acetic anhydride is in excess. After the reaction heating period is over |
|
Experiment 5 - Synthesis of Aspirin
The synthesis reaction of aspirin is shown below: Since acetic acid is very soluble in water it is easily separated from the aspirin product. The aspirin. |
|
5012 Synthesis of acetylsalicylic acid (aspirin) from salicylic acid
5012 Synthesis of acetylsalicylic acid (aspirin) from salicylic acid and acetic anhydride without vacuum finally |
|
A Greener Approach to Aspirin Synthesis Using Microwave Irradiation
ration of aspirin from salicylic acid and acetic anhydride. As rin synthesis without microwave irradiation in terms of reaction time |
|
ACETIC ANHYDRIDE HAZARD SUMMARY IDENTIFICATION
perfumes explosives and aspirin. REASON FOR CITATION. * Acetic Anhydride is on the Hazardous Substance List because it is regulated by OSHA and cited by |
|
Experiment 5 - Synthesis of Aspirin
In the presence of moisture aspirin may decompose (hydrolysis) into salicylic acid and acetic acid. This reaction is the reverse of the synthesis reaction. The |
|
An Efficient Microscale Procedure for the Synthesis of Aspirin
06?/06?/1998 The recommended procedures for the synthesis of aspirin use either acetic anhydride–concentrated sulfuric acid (1) or phosphoric acid (2) as the ... |
|
Preparation of Solid Acid–Activated Carbon as Catalyst in Aspirin
Using atmospheric pressure reaction conducted at 353 K |
|
SYNTHESIS OF ASPIRIN
In this experiment aspirin will be produced by reacting salicylic acid with acetic anhydride |
|
Synthesis of paracetamol by acetylation
reaction with acetic anhydride. This organic experiment is done in our laboratory for more than 20 years (approximately 200 students per year) without any |
|
The Synthesis of a Medicinal Agent- Aspirin
Sep 8 2020 · Preparation of Aspirin In a 125 ml Erlenmeyer flask place 4 g of pure salicylic acid Add 10 ml of acetic anhydride in such a way as to wash down any material adhering to the walls of the flask and then 10 drops of syrupy (85 percent) phosphoric acid (CAUTION- concentrated phosphoric acid can cause chemical burns handle with care!) |
|
Searches related to synthesis of aspirin without acetic anhydride PDF
Mar 27 2016 · In this experiment you will synthesize aspirin from acetic anhydride and salicylic acid using phosphoric acid as a catalyst to speed up the reaction (it is not a reactant or product in the overall equation) The reaction for the synthesis is given below H3PO4 C4H6O3 + C7H6O3 ? C9H8O4 + HC2H3O2 |
How is aspirin made?
When aspirin is ingested, it is broken down to salicylic acid by the basic conditions in the small intestine. It is then absorbed into the bloodstream. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. The phenol group on the salicylic acid forms an ester with the carboxyl group on the acetic acid.
How is acetic anhydride reacted with salicylic acid?
To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride. A small amount of a strong acid is used as a catalyst which speeds up the reaction. In this experiment, sulfuric acid will be used as the catalyst. The excess acetic anhydride will be quenched (reacted) with the addition of water.
What is the difference between salicylic acid and aspirin?
In modern times, salicylic acid is administered in the form of aspirin which is less irritating to the stomach than salicylic acid. To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride. A small amount of a strong acid is used as a catalyst which speeds up the reaction.
What happens when you mix aspirin with acetylsalicylic acid?
The acetylsalicylic acid will recrystallize, and the solid impurities (unreacted salicylic acid) should remain dissolved in the solution. The solid aspirin will again be collected using vacuum filtration and tested for purity. This aspirin should be more pure than the original aspirin.
|
8-Synthesis of Aspirin
Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid If no crystals appear, gently scratch the inside of the flask with a |
|
Experiment 5 - Synthesis of Aspirin
To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride A small amount of a strong acid is used as a catalyst which speeds up the reaction |
|
Synthesis of Aspirin - Carbon
Synthesis of salicylic acid from oil of wintergreen 2 Procedure 2 Carbon 4 Synthesis of aspirin from salicylic acid Salicylic Acid Acetic Anhydride Sodium Acetate (1D-13C), there are no tertiary carbons (DEPT-90) and there are three |
|
ASPIRIN SYNTHESIS - University of Helsinki
Which functional groups can be found in salicylic acid and aspirin? Salicylic acid: Catalyst increases reaction rate without being consumed in the process 4 Where do we get Why do we use acetic anhydride instead of acetic acid? |
|
Synthesis of Aspirin - Bellevue College
In this experiment you will synthesize aspirin from acetic anhydride and salicylic acid using phosphoric acid as a catalyst to speed up the reaction (it is not a |
|
Synthesis of Aspirin
Synthesis of Aspirin Synthesis Purification Characterization Mix salicylic acid (solid) with acetic anhydride (liquid) and a catalytic amount of H 2 SO 4 |
|
Synthesis of Aspirin
7 sept 2010 · Acetic anhydride is the preferred acid derivative to synthesize aspirin commercially If no crystals form, gently scratch the sides of the flask with |
|
From salicylic acid and acetic anhydride
5012 Synthesis of acetylsalicylic acid (aspirin) from salicylic acid 12 8 g (11 9 mL, 125 mmol) acetic anhydride is filled in the reaction flask and three drops of Without addition of sulphuric acid the yield of the crude product amounted to |
|
JCE1098 p1261 Synthesis of Aspirin: A General Chemistry Experiment
75 No 10 October 1998 • Journal of Chemical Education 1261 In recent years, examples acrid liquid acetic anhydride, salicylic acid reacts and dissolves |
|
Experiment 11 Synthesis and Analysis of Aspirin - CSUN
If the “corner” has no other marks, that means there is a carbon atom bonded to 2 of salicylic acid with acetic anhydride, using concentrated sulfuric acid as a |

![Experiment 5 - Synthesis of Aspirin - [PDF Document] Experiment 5 - Synthesis of Aspirin - [PDF Document]](https://0.academia-photos.com/attachment_thumbnails/36278700/mini_magick20180818-12561-1g9d5pm.png?1534579755)












![Aspirin Synthesis and Analysis - [PDF Document] Aspirin Synthesis and Analysis - [PDF Document]](https://www.coursehero.com/thumb/c0/dc/c0dc53e4be5989a9a39ac57751ec2d91b9980728_180.jpg)