synthesis of t butyl chloride theoretical yield
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride
How will 1 equiv of HCl + CaCl2 affect the yield and purity of tert pentyl chloride? acetate with octyl bromide for which the theoretical yield is 2.116 g. |
|
Experiment 3 Preparation of tert-Butyl Chloride
Calculate your percent yield and turn this in on your Organic Yield Report Sheet. Appendix 3.1 Distillation. Theory and Background: Distillation is the |
|
CH 2020/2270/2290 Post-lab: Synthesis of t-Butyl Chloride from t
What is the theoretical yield of t-butyl chloride? (Show your calculations not just the answer.) 3. What is your percent yield of t-butyl chloride? (Show |
|
Untitled
20 Apr 2005 t-butyl chloride by Friedel-Crafts Alkylation. ... 046 mole 14-di-t-butyl benzere is produced. 8.759. = theoretical yield 1 |
|
CH 2270 Post-lab: Synthesis of t-Butyl Chloride from t-Butanol Name
What is the theoretical yield of t-butyl chloride? (Show your calculations not just the answer.) 4. What is your percent yield of t-butyl chloride? (Show |
|
An Improved Synthesis of 1 3
https://www.journal.csj.jp/doi/pdf/10.1246/bcsj.36.747 |
|
1943] Synthesis of Tertiary Butyl Alcohol by the Hydration of
solution of sodium chloride. (DB315) increases the regeneration of isobutylene |
|
The comparison of Friedel-Crafts alkylation and acylation as a
Table 1: The theoretical and actual yield for the alkylation of m-xylene using t-butyl chloride. Theoretical yield (g). Actual yield (g). Per cent yield (%). |
|
The Exchange of Hydrogen and Chlorine between Bicyclo (22
https://pubs.acs.org/doi/pdf/10.1021/ja01206a013 |
|
THE PREPARATION AND PROPERTIES OF TERTIARY BUTYL
acetate we have succeeded in preparing pure tertiary butyl phenylacetate in yields equalto 65% of the theoretical. Since our procedure can be used to obtain |
|
Experiment 3 Preparation of tert-Butyl Chloride
The purpose of this experiment is to prepare tert-butyl chloride Calculate your percent yield and turn this in on your Organic Yield Report Sheet. |
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride
Multiply your crude product yield by the area percent indicated underneath your chromatogram. Identify correctly which peak is your ester product and which is. |
|
Untitled
Apr 20 2005 To synthesize 1 |
|
THE REACTIVITY OF ATOMS AND GROUPS IN ORGANIC
The yield of the mixed ethers was 82% of the theoretical. rapidly with water than does «-butyl chloride but that the tertiary chloride reacts more. |
|
Experiment Synthesis of t-Butyl Chlorid ee
Synthesis of t-Butyl Chloridee. Alcohols are critically important in the synthesis of other classes of organic compounds. With the exception of aromatics |
|
The comparison of Friedel-Crafts alkylation and acylation as a
Table 1: The theoretical and actual yield for the alkylation of m-xylene using t-butyl chloride. Theoretical yield (g). Actual yield (g). Per cent yield (%). |
|
4026 Synthesis of 2-chloro-2-methylpropane (tert-butyl chloride
4026 Synthesis of 2-chloro-2-methylpropane (tert-butyl chloride) from tert-butanol Yield: 639 mg (6.90 mmol 69%); bp 50–52 °C |
|
The Friedel-Crafts Reaction
Feb 27 2014 biphenyl (1) with two equivalents of tert-butyl-chloride (2) to ... isolated with a percent yield of 62% |
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride
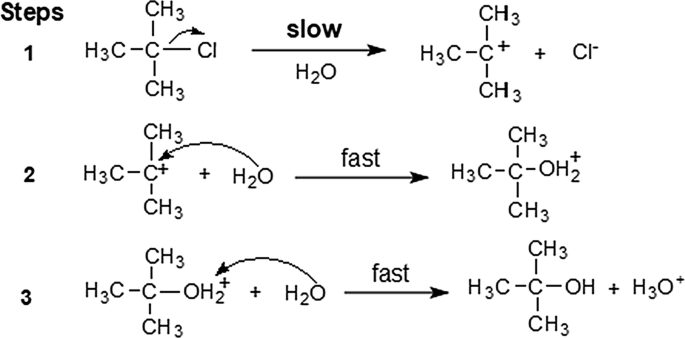
This lab experiment proposes the synthesis of an alkyl halide by reacting the corresponding alcohol with a hydrogen halide in an easy and inexpensive SN1 reaction 12 tert-Butanol reacts readily with HCl and forms the corresponding tert-butyl chloride at room temperature SN1 mechanisms are unimolecular because its slow step is unimolecular |
|
Synthesis and Reactivity of tert-butyl chloride - CHM2210 - Organic
Post-lab: Synthesis of t-Butyl Chloride from t-Butanol Name Section/TA 1 Draw the structures of t-butyl alcohol and t-butyl chloride 2 How many moles of t-butyl alcohol did you use in the synthesis of t-butyl chloride? (Show your calculations not just the answer ) 3 What is the theoretical yield of t-butyl chloride? (Show your calculations |
|
CH 2020/2270/2290 Post-lab: Synthesis of t-Butyl Chloride
Post-lab: Synthesis of t-Butyl Chloride from t-Butanol Name Section/TA 1 How many moles of t-butyl alcohol did you use in the synthesis of t-butyl chloride? (Show your calculations not just the answer ) 2 What is the theoretical yield of t-butyl chloride? (Show your calculations not just the answer ) 3 |
|
Searches related to synthesis of t butyl chloride theoretical yield PDF
The preparation of the activated electrophile begins with the attack of a unit of t-butyl-chloride on aluminum chloride (3)(formed by the interaction of aluminum metal and the dichloromethane solvent) Following this the t-butyl dissociates from the chlo-ride leaving a negatively charged Cl-AlCl3-complex (5)and an activated t-butyl-chloride |
What is a substitution reaction of tert-butyl chloride?
Synthesis and Reactivity of tert-butyl chloride - A substitution reaction occurs when the functional - Studocu post lab synthesis and reactivity of chloride via an sn1 reaction evan kirk minghui wang chm2210l:914 introduction in this experiment substitution reaction Skip to document Ask an Expert Sign inRegister Sign inRegister Home
What is the molecular weight of t-butyl alcohol?
Molecular weight of t-Butyl alcohol = 74.12g/mol Molecular weight of t-Butyl chloride = 92.57g/mol I'm really confused on how to go about calculating this as the only thing I've learnt about calculating percentage yield is (experimental value/ theoretical value) x 100%. Help would be greatly appreciated. Thank you very much!
What is the percent yield of tert-butyl chloride?
This experiments purpose was to extract and synthesize tert-butyl chloride using the proper SN1 reaction. In the experiment the percent yield came out to be 76%, this is significantly higher than the 50% mark. The possibility of getting a percent yield of 50% or less comes down to unseen error incorporated in the experiment.
How do you calculate the yield of a butyl chloride reaction?
From mass and the molar mass of the alcohol, the amount of substance (in mol) is available. This will define the maximal outcome of your reaction (100% yield). Calculate the amount of substance for 12 g of the butyl chloride. […] yield is (experimental value/ theoretical value) x 100%.
|
Experiment 3 Preparation of tert-Butyl Chloride
butyl alcohol (tert-butanol) using an acid catalyzed substitution reaction Calculate your percent yield and turn this in on your Organic Yield Report Sheet |
|
Preparation of t-butyl chloride - University of Illinois Archives
Preparation of t-butyl chloride (2-chloro-2-methylpropane) C CH3 CH3 CH3 OH HCl C CH3 CH3 CH3 Cl + H2 O t-butyl alcohol t-butyl chloride |
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride - The Royal Society
Scheme SM 2 1 1 1 – Mechanism for the formation of tert-Butyl Chloride from tert- Butyl calculated, making note of the limiting reagent in this specific reaction |
|
CH 2270 Post-lab: Synthesis of t-Butyl Chloride from t - Clemson
How many moles of t-butyl alcohol did you use in the synthesis of t-butyl chloride ? (Show your calculations, not just the answer ) 3 What is the theoretical yield of |
|
Chem342L Friedel_Craftspdf
t-butyl chloride 192 501,851-25 151-5210ml 1 092 Iflammable benzone limiting, Theoretical Yield i 1 Aspirin synthesized las week was placed in the bottle |
|
Introductory Organic Chemistry Laboratory (CHM 361 - MU BERT
23, 23A n-Butyl Bromide PE Sept 18 T 14 24, 24A 4-Methylcyclohexene - Sept 20 R 25 24 theoretical yield and percent yield These tables contain a detailed calculation of the theoretical and percent yields for the synthesis ) IV |