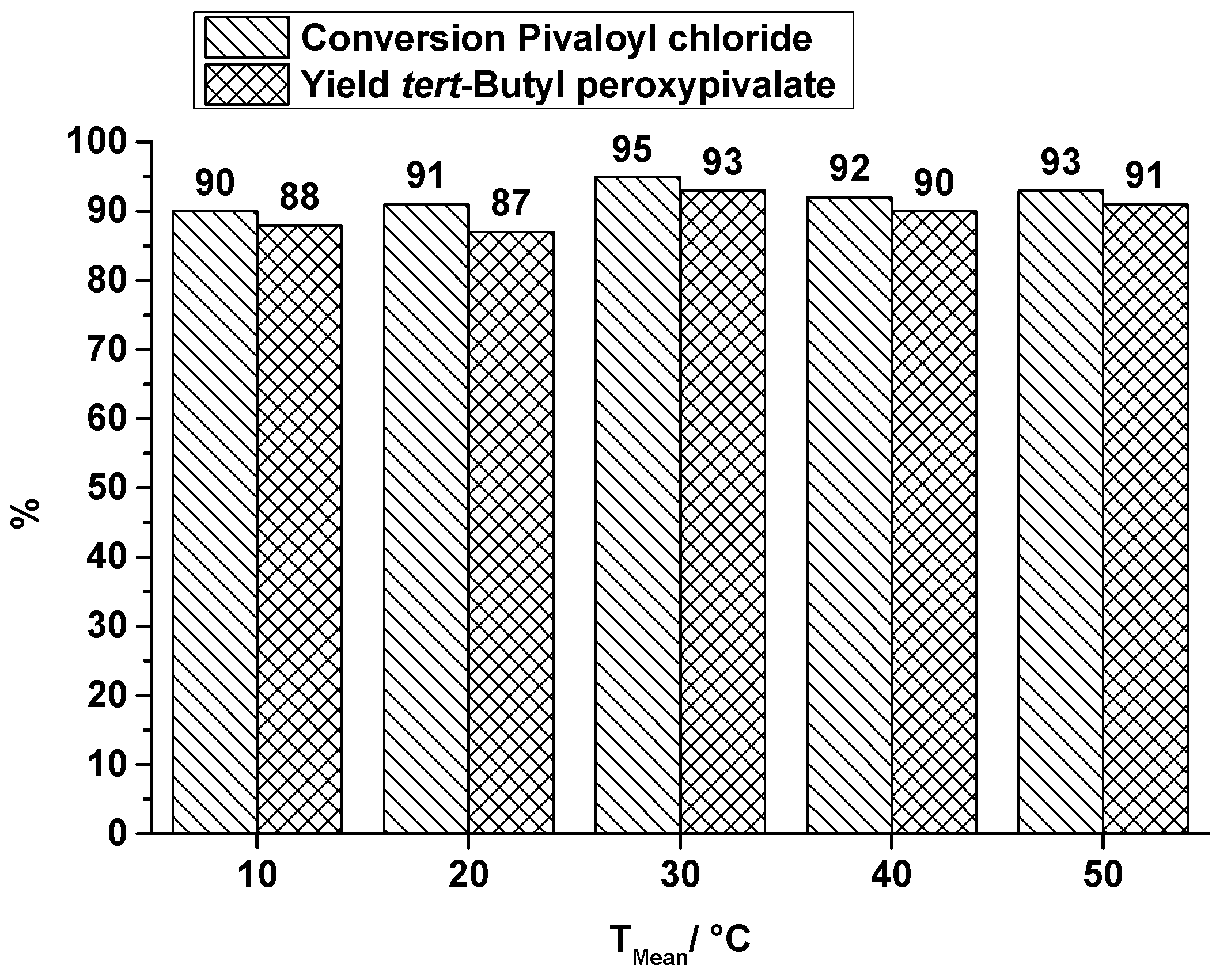
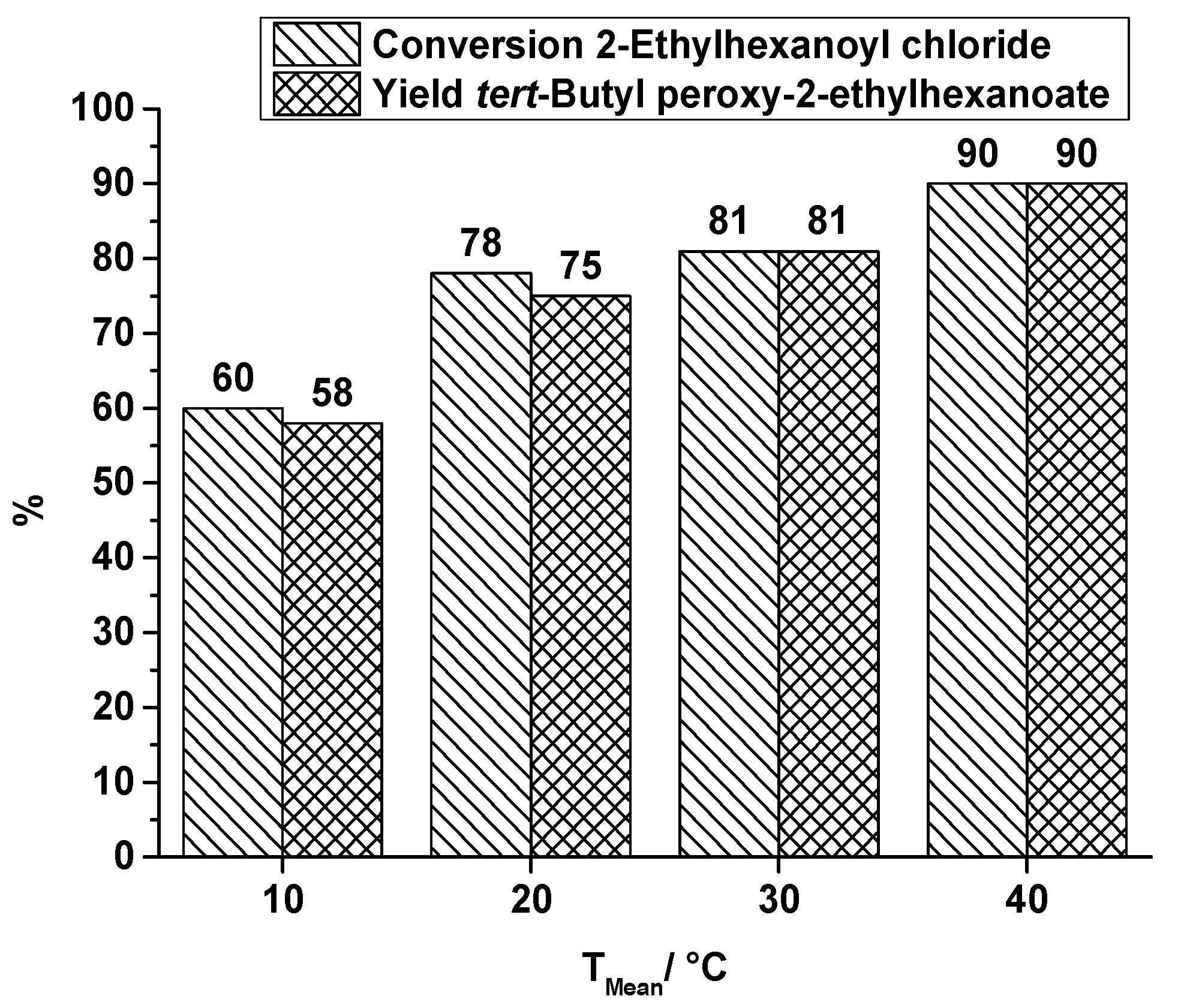
synthesis of tert butyl chloride lab report
| A SN1 Reaction: Synthesis of tert-Butyl Chloride |
|
Experiment 3 Preparation of tert-Butyl Chloride
Calculate your percent yield and turn this in on your Organic Yield Report Sheet. Appendix 3.1 Distillation. Theory and Background: Distillation is the |
|
TERT-BUTYL CHLORIDE FOR SYNTHESIS MSDS CAS-No.
12 Feb 2019 tert-BUTYL CHLORIDE FOR SYNTHESIS (507-20-0). Viscosity kinematic. 0.586 mm² ... Results of PBT and vPvB assessment. No additional information ... |
|
The synthesis of 44-di-tertbutyl biphenyl: a sophomore organic
Neckers4 has reported the alkylation of biphenyl with tert-butyl chloride as an organic laboratory experiment. In his experiment the alkylation may be |
|
Exp.07: preparation of t-butyl chloride
Organic Chemistry. 447 chem (Lab). 3. Laboratory Report. Name: ---------------------------------. Date: --------------------. Experiment Subject: -------------- ... |
|
Friedel-Crafts Alkylation: Synthesis of p-di-t-butylbenzene
The overall reaction is shown in Figure 3. Page 2. Figure 3: The Freidel Crafts alkylation of t-butylbenzene by t-butyl chloride in. |
|
CH 2270 Introduction to Organic Laboratory and Techniques
Pre-Lab for n-Butyl Bromide Synthesis. Post-Lab for t-Butyl Chloride Synthesis. Notebook Pages – t-Butyl Chloride Synthesis. Oral Report for Distillation. 7. |
|
NO LABS - SPRING BREAK NO LABS - SPRING BREAK
The syllabus experiments |
| SN1 reaction mechanisms of tert -butyl chloride in aqueous solution |
|
GUIDELINES AND MARKING SCHEME FOR PRACTICAL REPORT
1. Too brief (e.g. “Lab Report” “Mercury in Fish” |
| A SN1 Reaction: Synthesis of tert-Butyl Chloride |
| A SN1 Reaction: Synthesis of tert-Butyl Chloride |
|
Experiment 3 Preparation of tert-Butyl Chloride
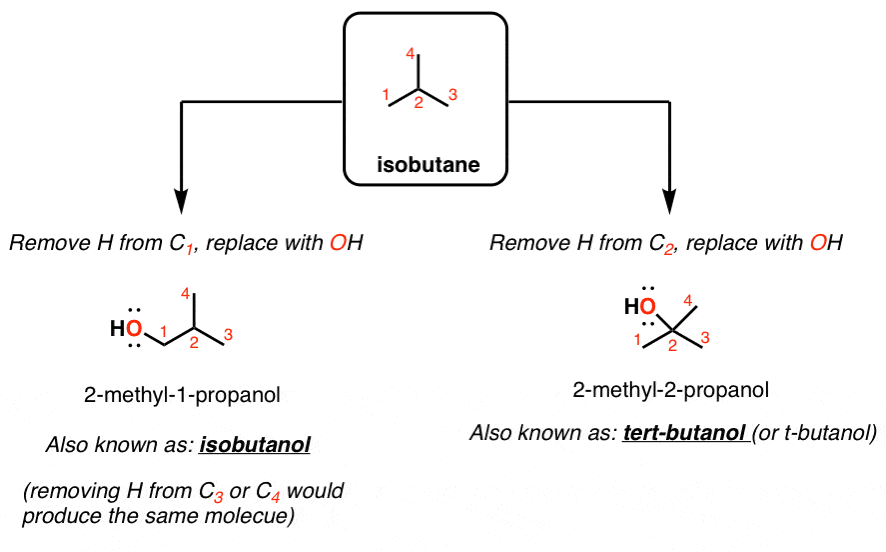
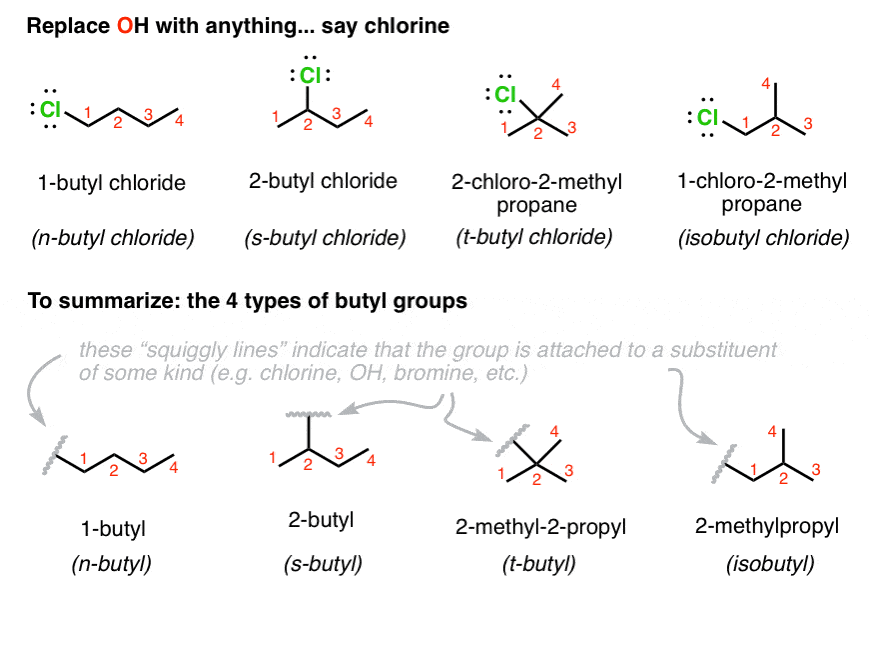
The purpose of this experiment is to prepare tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed |
|
Experiment 3 Preparation of tert-Butyl Chloride
The purpose of this experiment is to prepare tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed |
|
Exp.07: preparation of t-butyl chloride
447 chem (Lab). 1. Exp.07: preparation of t-butyl chloride. Objectives: -Preparetert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol |
|
4026 Synthesis of 2-chloro-2-methylpropane (tert-butyl chloride
http://www.oc-praktikum.de. March 2006. 1. 4026 Synthesis of 2-chloro-2-methylpropane (tert-butyl chloride) from tert-butanol. |
|
TERT-BUTYL CHLORIDE FOR SYNTHESIS MSDS CAS-No.
12 Feb 2019 TERT-BUTYL CHLORIDE FOR SYNTHESIS. MSDS. CAS-No.: 507-20-0 MSDS. SECTION 1: Identification of the ... Results of PBT and vPvB assessment. |
|
CH 2020/2270/2290 Synthesis of t-Butyl Chloride from t-Butanol (An
In this experiment you will convert t-butanol to t-butyl chloride by reaction with hydrochloric acid. The overall reaction is. |
|
Friedel-Crafts Alkylation: Synthesis of p-di-t-butylbenzene
Figure 3: The Freidel Crafts alkylation of t-butylbenzene by t-butyl chloride in the presence of aluminum chloride. *Note: The reaction should be performed |
|
Headline
Sustainable Lab Practices in the CU- of hydrolysis of tert-butyl chloride”. Previously: ... Synthesis 2001 25 |
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride
This lab experiment proposes the synthesis of an alkyl halide by reacting the corresponding alcohol with a hydrogen halide in an easy and inexpensive SN1 reaction 12 tert-Butanol reacts readily with HCl and forms the corresponding tert-butyl chloride at room temperature SN1 mechanisms are unimolecular because its slow step is unimolecular |
|
Experiment 7 Post Lab- Synthesis and Reactivity of tert-Butyl Chloride
Preparation of tert-Butyl Chloride The purpose of this experiment is to prepare tert-butyl chloride (2-chloro-2-methylpropane) from tert-butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction (Note: the correct IUPAC name for this compound is 2-methyl-2-propanol) |
|
Exp07: preparation of t-butyl chloride - KSU Faculty
Experimental Procedure In your separatory funnel (250 mL) place 120 mL of concentrated hydrochloric acid that has first been cooled to 5°C in an ice bath Add 28 mL (22 g )of t-butanol and swirl gently with the stopper off the separatory funnel release the pressure after every swirlfor 20 minutes |
|
Experiment 3 Preparation of tert-Butyl Chloride
Preparation of tert-Butyl Chloride The purpose of this experiment is to prepare tert-butyl chloride (2-chloro-2-methylpropane) from tert-butyl alcohol (tert-butanol) using an acid catalyzed substitution reaction (Note: the correct IUPAC name for this compound is 2-methyl-2-propanol) |
How to prepare tert-butyl chloride?
Thus, we were able to prepare tert-butyl chloride by preforming a substitution reaction using tert-butyl alcohol and hydrochloric acid. The formation and reactivity of tert-butyl chloride are both achieved by an SN1 reaction. An SN1 reaction is a first order nucleophilic substitution which is unimolecular.
How does tert-butanol react with HCl?
tert-Butanol reacts readily with HCl and forms the corresponding tert-butyl chloride at room temperature. SN1 mechanisms are unimolecular because its slow step is unimolecular. The reaction proposed involves an initial step where the tert- butyloxonium ion is formed by protonation.
Why does tert-butyl chloride contain a tertiary alkyl halide?
This can be seen when comparing the results of the qualitative tests with the literature information. In test tubes one and two it is made clear that tert-butyl chloride contains a tertiary alkyl halide because it reacts and forms a precipitate with silver nitrate as the literature predicted.
What is arrow pushing in tert-butyl chloride?
In the preparation of tert-butyl chloride a mechanism called arrow pushing outlines the movement of elections throughout the course of a reaction to track the pacing and overall progression of the reaction.
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride - The Royal Society
This lab experiment proposes the synthesis of an alkyl halide by reacting the Scheme SM 2 1 1 1 – Mechanism for the formation of tert-Butyl Chloride from tert- Discussion of Student Results: Kinetic vsThermodynamic (Equilibrium) Effects |
|
Experiment 3 Preparation of tert-Butyl Chloride - Remondini
butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction (Note: the up the apparatus for distillation (see Figure 3 6 and discussion below) |
|
Exp07: preparation of t-butyl chloride
-Preparetert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol ( tert- butanol) using The first step of the overall reaction is an acid-base reaction between the t-butanol and the hydrochloric chem (Lab) 3 Laboratory Report |
|
Materials - Clemson
CH 2020/2270/2290 Synthesis of t-Butyl Chloride from t-Butanol (An SN1 Reaction) (adapted from Organic Chemistry: A Short Course, H Hart, L E Craine, |
|
Preparation of t-butyl chloride - University of Illinois Archives
Preparation of t-butyl chloride (2-chloro-2-methylpropane) C CH3 (CH 3 ) 3 CCl + H 2 O Reaction Mechanism? Theoretical Yield Experimental Yield |
|
Experiment 8 — Kinetics of SN1 Solvolysis
Pre-lab preparation (1) Textbook Ch 8 covers the SN2 and SN1 mechanisms Read/review as reaction In this lab we will be measuring the rate of solvolysis of tert-butyl chloride as a function detailed discussion of kinetic expressions ) |
|
ORGANIC CHEMISTRY
EXPERIMENT 8: Synthesis of borneol by reduction of camphor 60 INDEX The laboratory report: within which assessed are the order, clarity, correctness, rigor, and quantity Experiment 8: Synthesis of tert-butyl chloride 60 |
|
SN1 Reaction: Hydrolysis of tert-butyl chloride - NC State University
The concentration of t-butyl chloride can be changed to observe the change in rate with change in [tBuCl] Discussion: This experiment demonstrates a typical SN1 |
|
4026 Synthesis of 2-chloro-2-methylpropane (tert-butyl chloride
Reaction 3 0 g (2 6 mL, 30 mmol) conc hydrochloric acid are filled in a 10 mL two-neck flask equipped with a magnetic stir bar and a reflux condenser with |