arrhenius equation graph
|
Arrhenius Equation
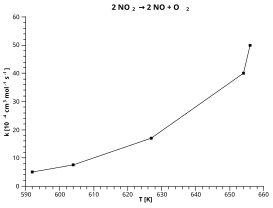
Arrhenius Equation The rate and E is the activation energy for the A graph of rate against absolute temperature is exponential as shown below Temp |
|
The Arrhenius equation indicates the relationship between the
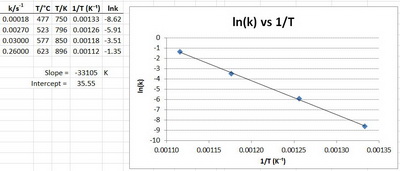
One straight line is obtained by plotting lnk against 1/T after measuring the reaction rate at different temperatures As is shown in Equation 2, the slope represents |
|
AP* Chemistry Arrhenius Made Easy
the activation energy, which must be overcome to produce a chemical reaction The collision If we graph the value of the rate constant, k, versus the Kelvin |
|
The development of the Arrhenius equation - ntc see result
known as the Arrhenius equation According to this, a where R is the gas constant and E is the activation energy The an Arrhenius plot of in h against 11T |
|
The Arrhenius Equation - Phoenix Tutoring
4) Once you know the gradient, you can use it to find both the activation energy and the Arrhenius constant Example: The graph below shows an Arrhenius plot |
|
Arrhenius Plots—Curves or Straight Lines?
Arrhenius (1889) showed that, under certain conditions, such a plot should yield a straight line with a slope proportional to the activation energy of the reaction |