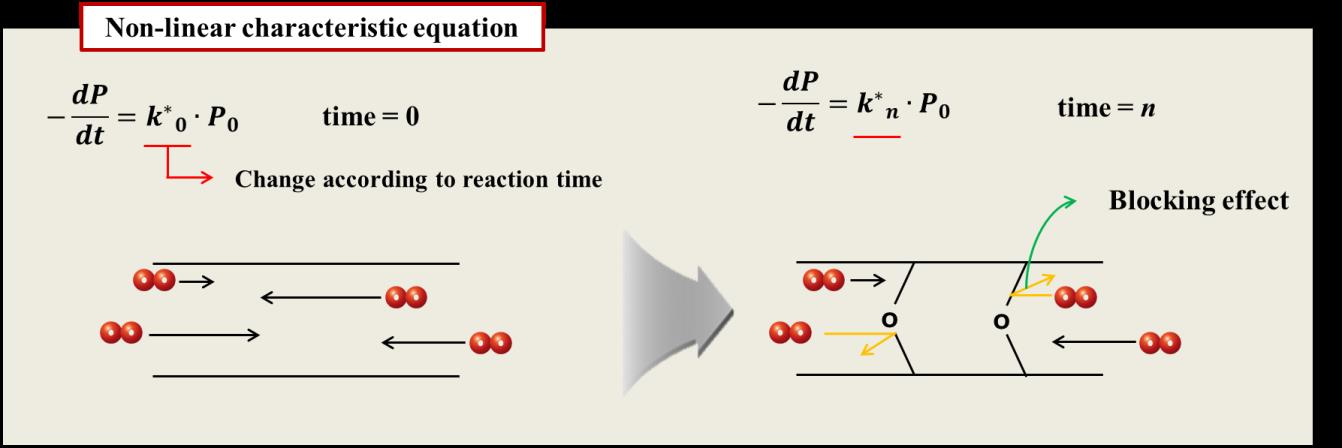
arrhenius equation r
|
Arrhenius Equation
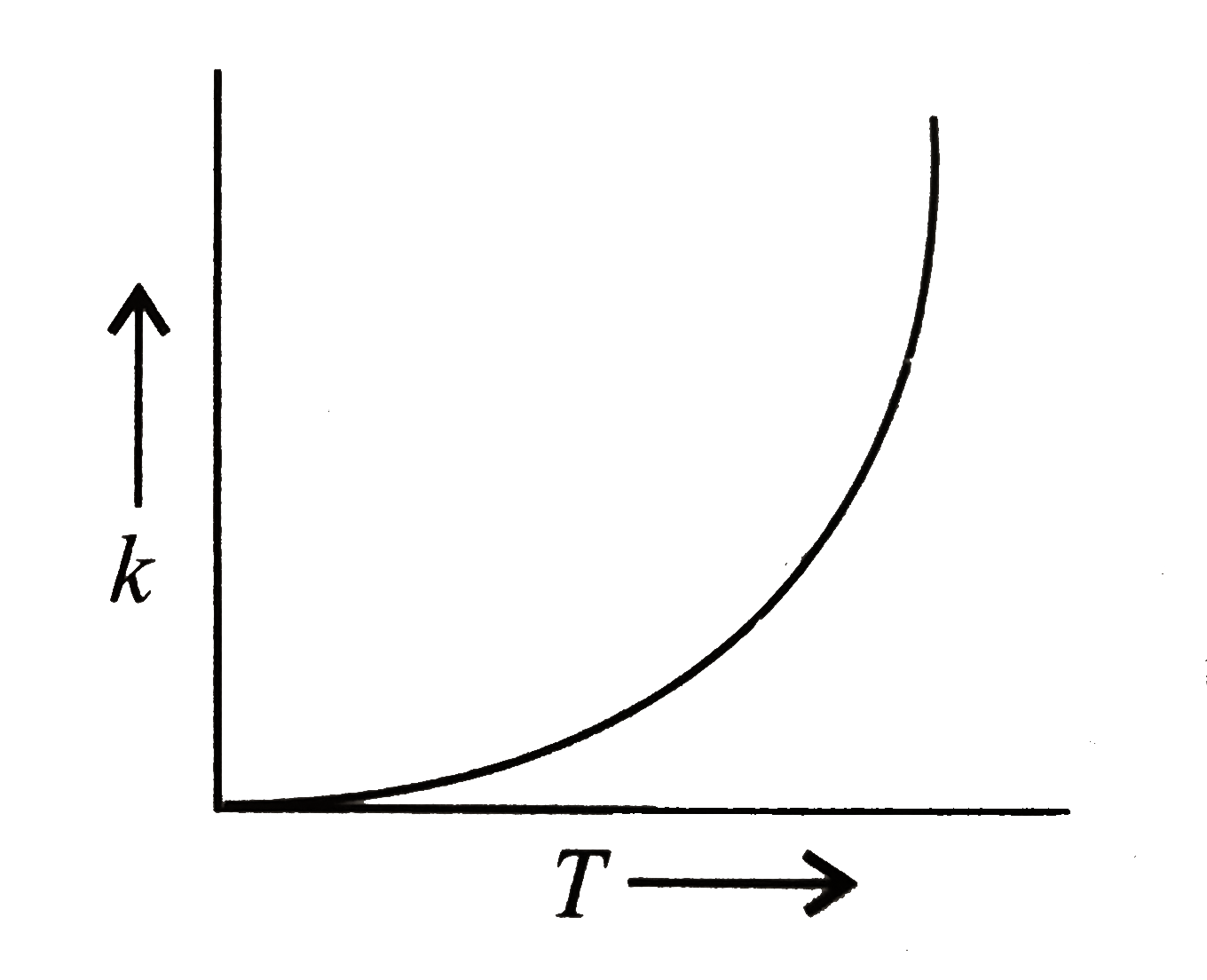
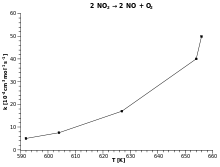
Once activation energy for a reaction is known; it is easy to predict the value for the rate constant k at a different temperatureT1 from its value k at another temperature T Equation 8 becomes:- ln( k1/k)=Ea/R*(1/T-1/T1) for a reaction taking place at T and T1, temperatures |
|
The development of the Arrhenius equation - ntc see result
known as the Arrhenius equation where R is the gas constant and E is the activation energy 1850, an equation relating the rate constant of a reaction to |
|
On the Validity of the Arrhenius Equation for Electron - CORE
where T is the temperature in energy units, A is a constant, and Ea is the so- called activation energy In numerous cases, activation energies have thus been |
|
Package rpmodel
4 déc 2019 · tion, and dark respiration rates as a function of the environment (temperature, CO2, Arrhenius kinetics are described by an equation of |
|
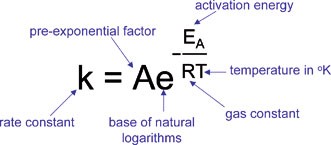
The Arrhenius equation indicates the relationship between the
The Arrhenius equation indicates the relationship between the reaction temperature (T) and the reaction rate constant (k) in Equation 1, where A represents the pre- exponential factor, Ea is the apparent activation energy, T and R stand for absolute temperature and universal gas constant, respectively |
|
Reaction Rates and Temperature; Arrhenius Theory Arrhenius Theory
a is the activation energy R is the ideal-gas constant (8 314 J/Kmol) T is the temperature in K constant increases according to the Arrhenius Equation |
|
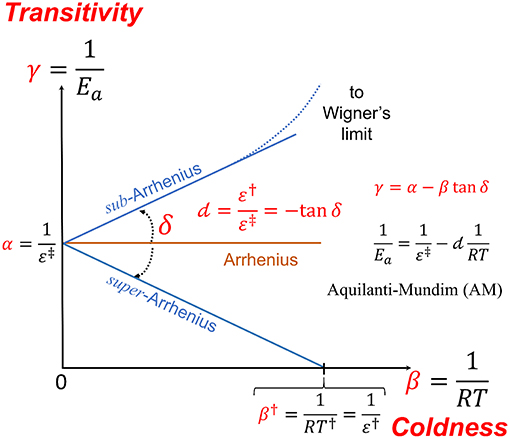
Evaluating the Simple Arrhenius Equation for the Temperature
18 juil 2020 · In this case, coupled chemical reactions would collapse into a common Arrhenius equation with one master activation energy and integrated |




















![PDF] BARC Exam Chemistry Syllabus PDF] BARC Exam Chemistry Syllabus](https://reader021.docslide.net/reader021/html5/20170802/575076d41a28abdd2ea06990/bg1.png)