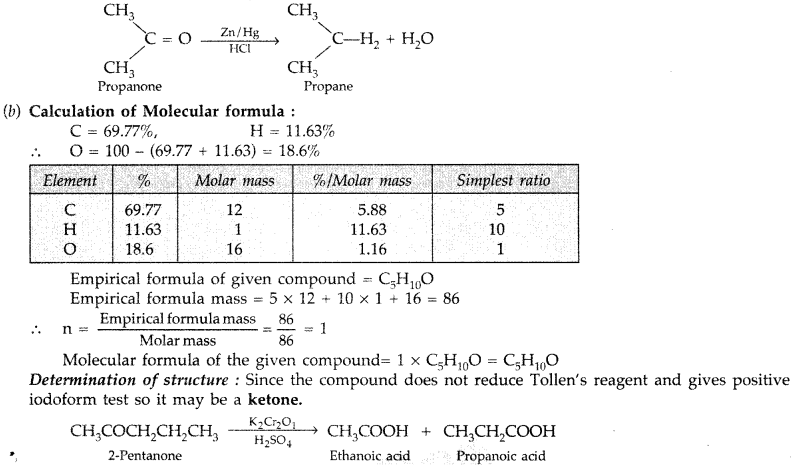
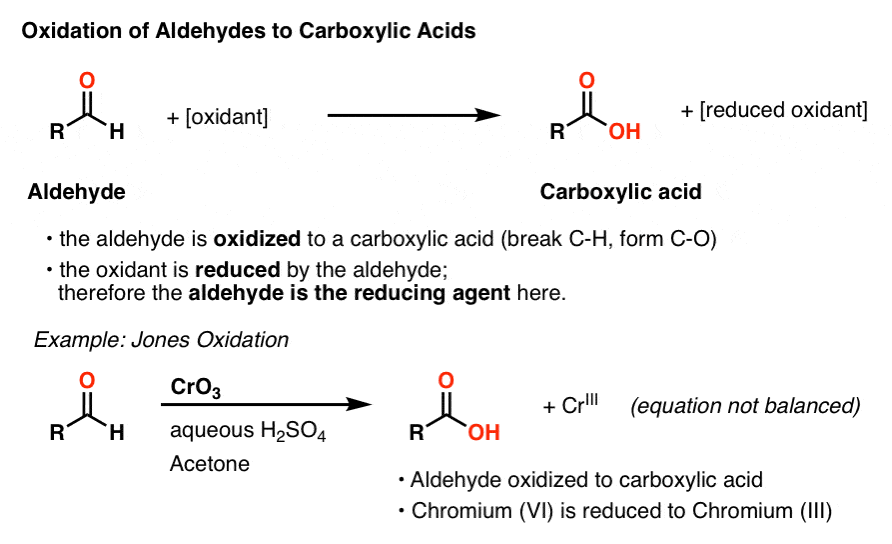
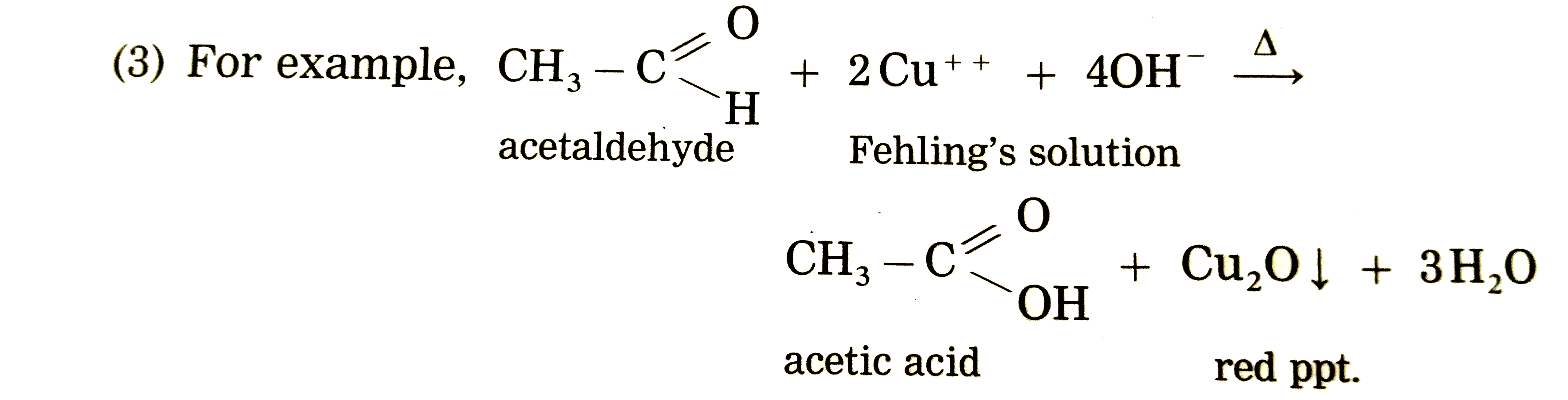test to distinguish between carboxylic acid and aldehyde
|
Testsforfunctionalgroups - inorganiccompounds
This is a test that distinguishes carboxylic acids from phenols. RCOOH + (v) How will you distinguish between phenol and benzoic acid? (vi) Why does ... |
|
Chemical tests to distinguish carbonyl compounds
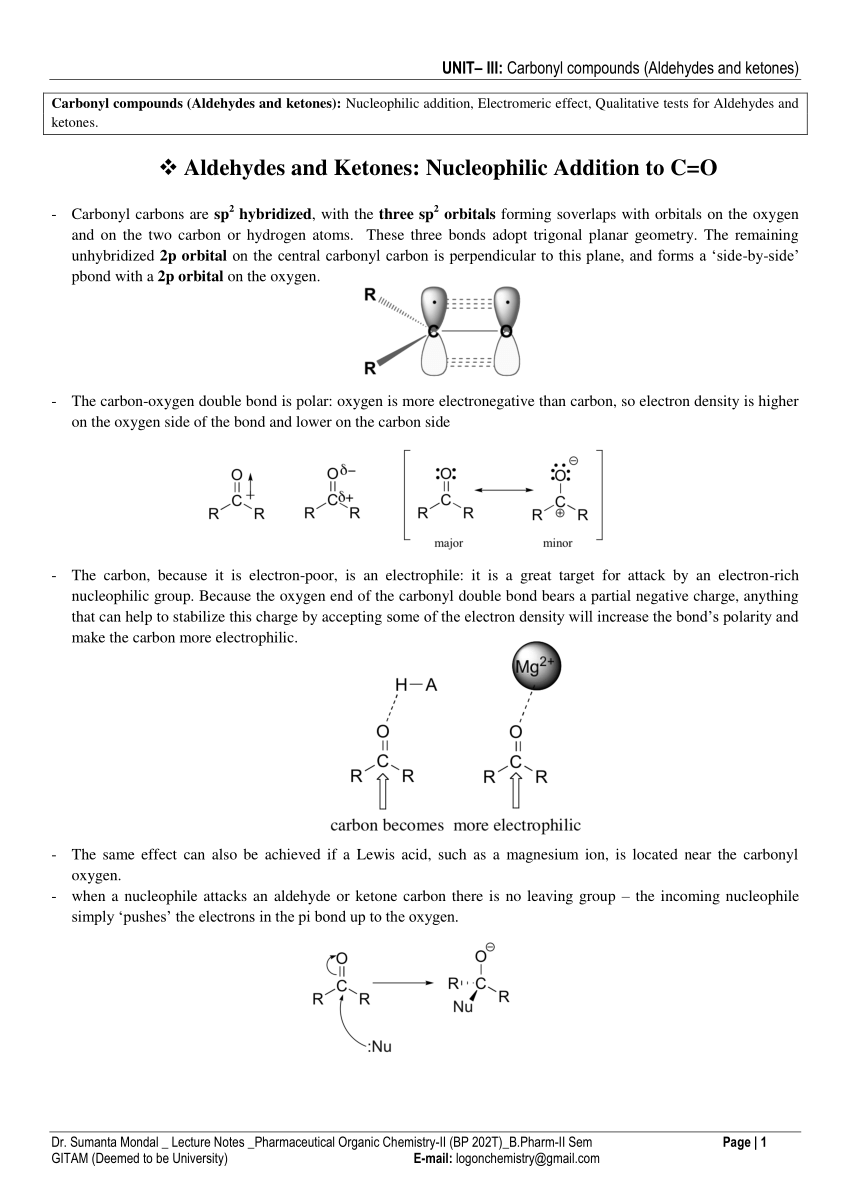
Recall that alcohols can be oxidized to form aldehydes ketones and carboxylic acids. • the pi-bond between C and O atoms breaks |
|
Topic 13
DISTINGUISHING BETWEEN DIFFERENT ORGANIC COMPOUNDS. Many of the organic compounds d) test for carboxylic acids. Carboxylic acids are acids and can liberate ... |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
4-Methoxybenzoic acid (acid strength). 8.13 Give simple chemical tests to distinguish between the following pairs of compounds. (i) Propanal and Propanone. |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
4-Methoxybenzoic acid (acid strength). 12.13 Give simple chemical tests to distinguish between the following pairs of compounds. (i)Propanal and Propanone. |
|
QUESTION BANK UNIT 12 ALDEHYDES KETONES
(ii) Ethanal to 2-hydroxy propanoic acid. (iii) Toluene to benzoic acid. (b) Give simple chemical test to distinguish between: (i) Pentan-2-one and Pentan-3 |
|
Core practical 15: Analyse organic and inorganic unknowns
Write a plan for the safe conduct of this experiment to test for carboxylic acids. What further test could you do to distinguish between sulfate(VI) ions and ... |
|
AQA Chemistry A-level - Required Practical 6
Tests for alcohol aldehyde |
|
Identifying an Unknown Compound by Solubility Functional Group
The solubility of carboxylic acids and amines is so characteristic that solubility tests alone differentiate these functional groups from all the others in this |
|
INFRARED SPECTROSCOPY (IR)
Study the similarities and the differences so that you can distinguish between the two. A carboxylic acid functional group combines the features of alcohols ... |
|
Chemical tests to distinguish carbonyl compounds
can be used to help identify the specific aldehyde or ketone after purifying |
|
Testsforfunctionalgroups - inorganiccompounds
To identify the functional groups present in an organic compound. I. TESTS FOR This is a test that distinguishes carboxylic acids from phenols. |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
Aldehydes ketones and carboxylic acids are widespread in plants 12.13 Give simple chemical tests to distinguish between the following pairs of ... |
|
S42.CHP:Corel VENTURA
We tested the ability of human subjects to distinguish between aliphatic odorants 1-Alcohols n-aldehydes |
|
Lab 14: Qualitative Organic Analysis
Organic chemists often must identify unknown compounds. one major functional group (alcohol ketone |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
Aldehydes ketones and carboxylic acids are widespread in plants 12.13 Give simple chemical tests to distinguish between the following pairs of ... |
|
Topic 13
DISTINGUISHING BETWEEN DIFFERENT ORGANIC COMPOUNDS test for carboxylic acids ... Primary alcohols can be oxidized to form aldehydes. |
|
Identifying an Unknown Compound by Solubility Functional Group
In some cases these tests will be sufficient to identify the functional group(s) such as alcohols |
|
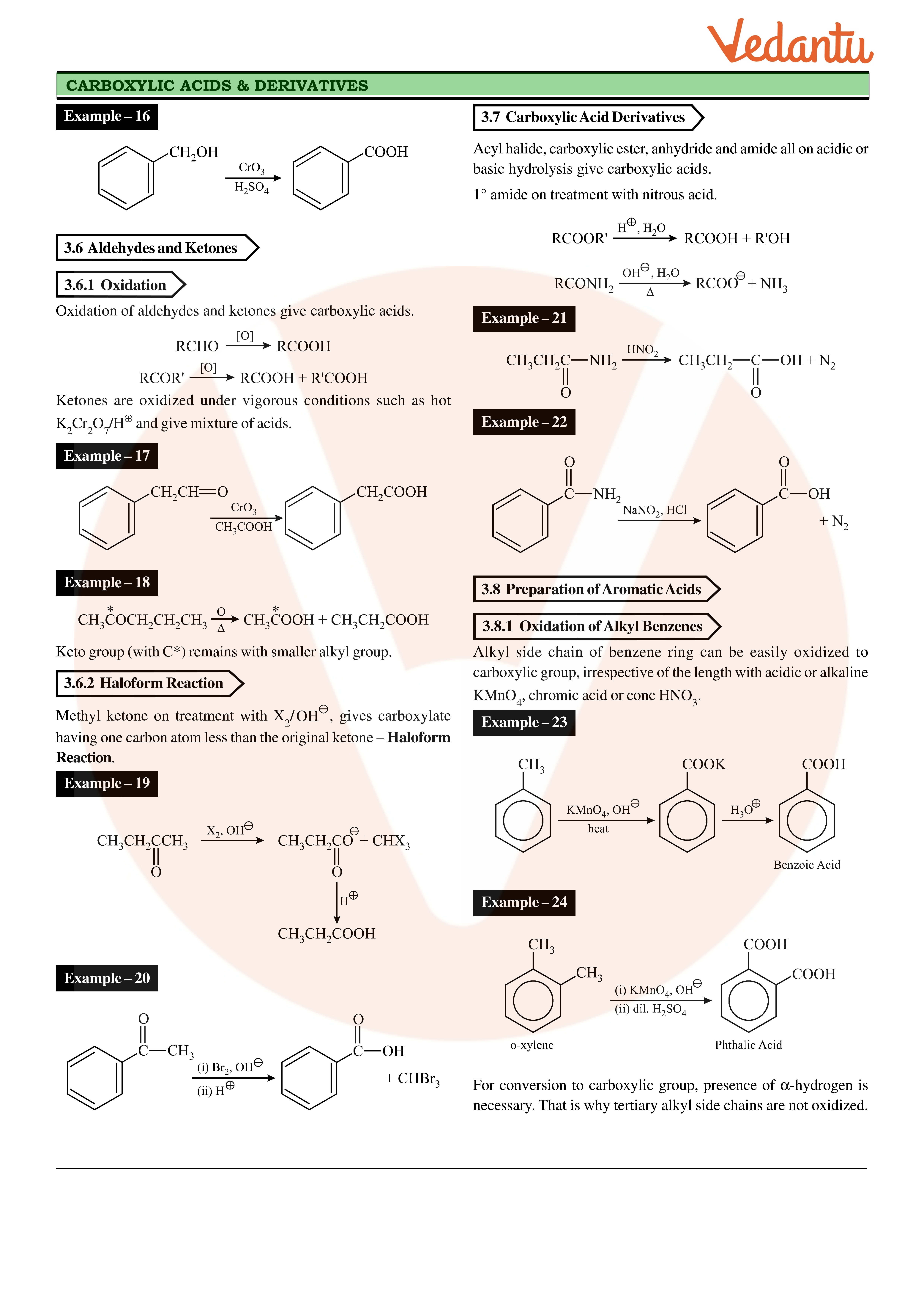
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters carbon chain in between the two carboxyl groups ... to 1° alcohols |
|
Identifying an Unknown Compound by Solubility Functional
A pH of 4 or lower indicates a carboxylic acid A pH of 8 or higher indicates an amine Water-soluble compounds are tested with 5 sodium hydrogen carbonate (NaHCO3) to determine whether or not they are carboxylic acids Carboxylic acids react with NaHCO3 to produce carbon dioxide bubbles as shown below in Equation 3 |
|
Searches related to test to distinguish between carboxylic acid and aldehyde PDF
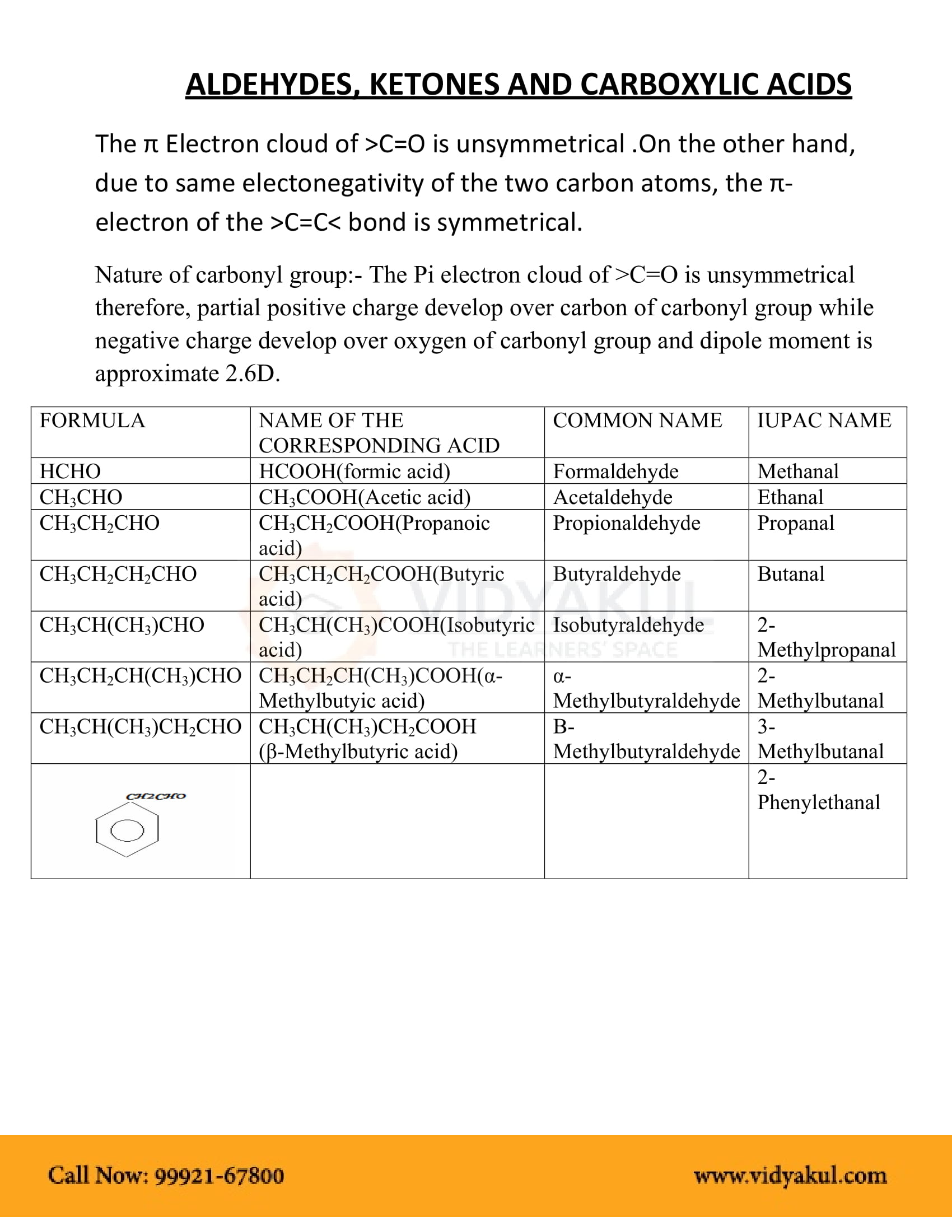
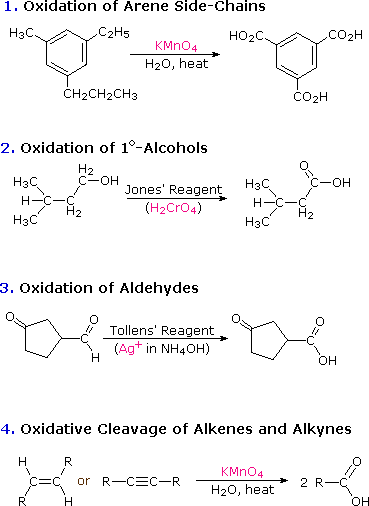
Aldehydes Ketones and Carboxylic Acids MODULE - 7 Chemistry of Organic 27 1 2 Preparation of Aldehydes and Ketones Compounds You have already studied most of the methods used in the synthesis of aldehydes and ketones in the previous lesson Let us now refresh them 1 Oxidation of Primary and Secondary Alcohols |
How do you identify a carboxylic acid from aldehydes and ketones?
This test is used to identify a carboxylic acid from aldehydes and ketones. Carboxylic acids undergo acid-base reaction with carbonate and hydrogen carbonates to produce salt, water and carbon dioxide. No acid and base reactions occur between an aldehyde/ketone and carbonates.
How do you know if a compound is a carboxylic acid?
A pH of 4 or lower indicates a carboxylic acid. A pH of 8 or higher indicates an amine. Water-soluble compounds are tested with 5% sodium hydrogen carbonate (NaHCO3) to determine whether or not they are carboxylic acids. Carboxylic acids react with NaHCO3 to produce carbon dioxide bubbles, as shown below in Equation 3.
How are carboxylic acids prepared?
We prepare carboxylic acids by the oxidation of aldehydes or alcohols whose –OH functional group is located on the carbon atom at the end of the chain of carbon atoms in the alcohol: A chemical reaction with two arrows is shown.
What are carboxylic acids and hydroxyl halides?
The carbonyl compounds in whichcarbon of carbonyl group is bonded to carbon orhydrogen and oxygen of hydroxyl moiety (-OH) areknown as carboxylic acids, while in compounds wherecarbon is attached to carbon or hydrogen and nitrogenof -NH2 moiety or to halogens are called amides andacyl halides respectively.
|
Chemical tests to distinguish carbonyl compounds - WordPresscom
1: Detecting an aldehyde or ketone Aldehydes and ketones react with 2,4-dinitrophenylhydrazine (2,4-DNP or 2,4-DNPH) to form an orange or yellow precipitate No precipitate is formed with other carbonyl compounds such as carboxylic acids or esters |
|
Tests for functional groups
Add 1 cm3 of the 'unknown' and heat in a water bath at 50 ºC for a few minutes If a 'silver mirror' is formed on the inside of the test tube, this confirms the presence of an aldehyde ii) Reaction with water Carboxylic acids are weak acids, i e they are only partially ionised in aqueous solution |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
Aldehydes, ketones and carboxylic acids are widespread in plants 12 13 Give simple chemical tests to distinguish between the following pairs of compounds |
|
Lab 14: Qualitative Organic Analysis - California State University
identify a solvent system that will separate your unknown from any impurities After you have Both strong and weak acids (Carboxylic acids and phenols) will be deprotonated This reaction tests for the presence of aldehydes and ketones |
|
Organic Chemistry
Name alkyl halides, alcohols, phenols, aldehydes, ketones, carboxylic acids and Perform reactions typical of alcohols and distinguish between aliphatic Use basic diagnostic tests and prepare synthetic derivatives for the purpose of |
|
7-Aldehydes, Ketones, C Acids
Experiment 7 – Aldehydes, Ketones, and Carboxylic Acids Aldehydes of each compound in its own separate test tube the stirring rod between solutions |
|
Identification of an Unknown – Alcohols, Aldehydes, and Ketones
precipitates, can be used to differentiate alcohols, aldehydes, and ketones, and also to provide (Aldehydes oxidize to carboxylic acids in the presence of |
|
27 ALDEHYDES, KETONES AND CARBOXYLIC ACIDS - NIOS
distinguish between aldehydes and ketones on the basis of certain reactions and tests based on them; ○ give IUPAC names of carboxylic acids; ○ |
|
Identification of Aldehydes and Ketones
General test (2,4-Dinitrophenylhydrazine reagent) Both aldehydes Differentiation between aldehydes and ketones is achieved by taking the advantage carboxylic acid with an accompanying reduction of the silver ions from this reagent to |
|
Long Answer Questions - SelfStudys
Q Give simple chemical tests to distinguish between the following pairs of compounds: Ans (a) Butanal being an aldehyde reduces Tollens' reagent to give silver (b) Benzoic acid decomposes NaHCO3 to produce brisk effervescence due to |