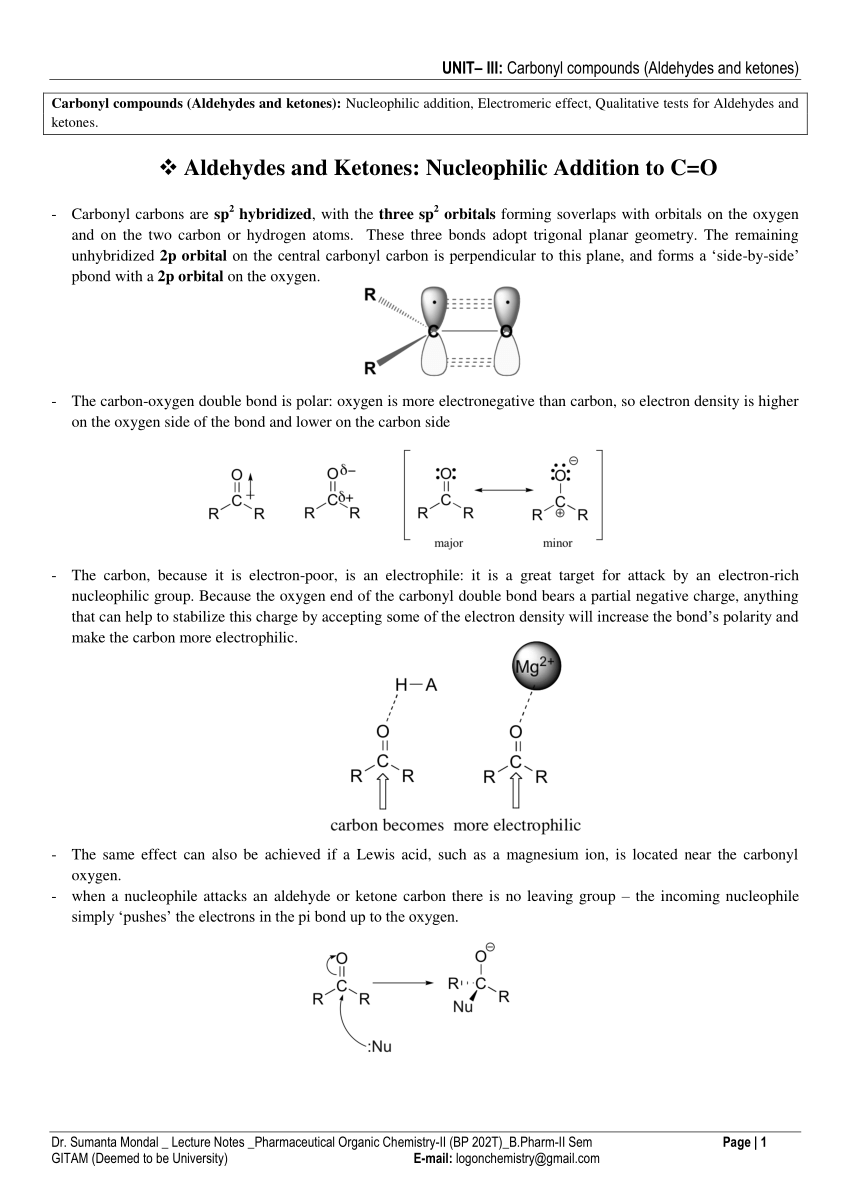
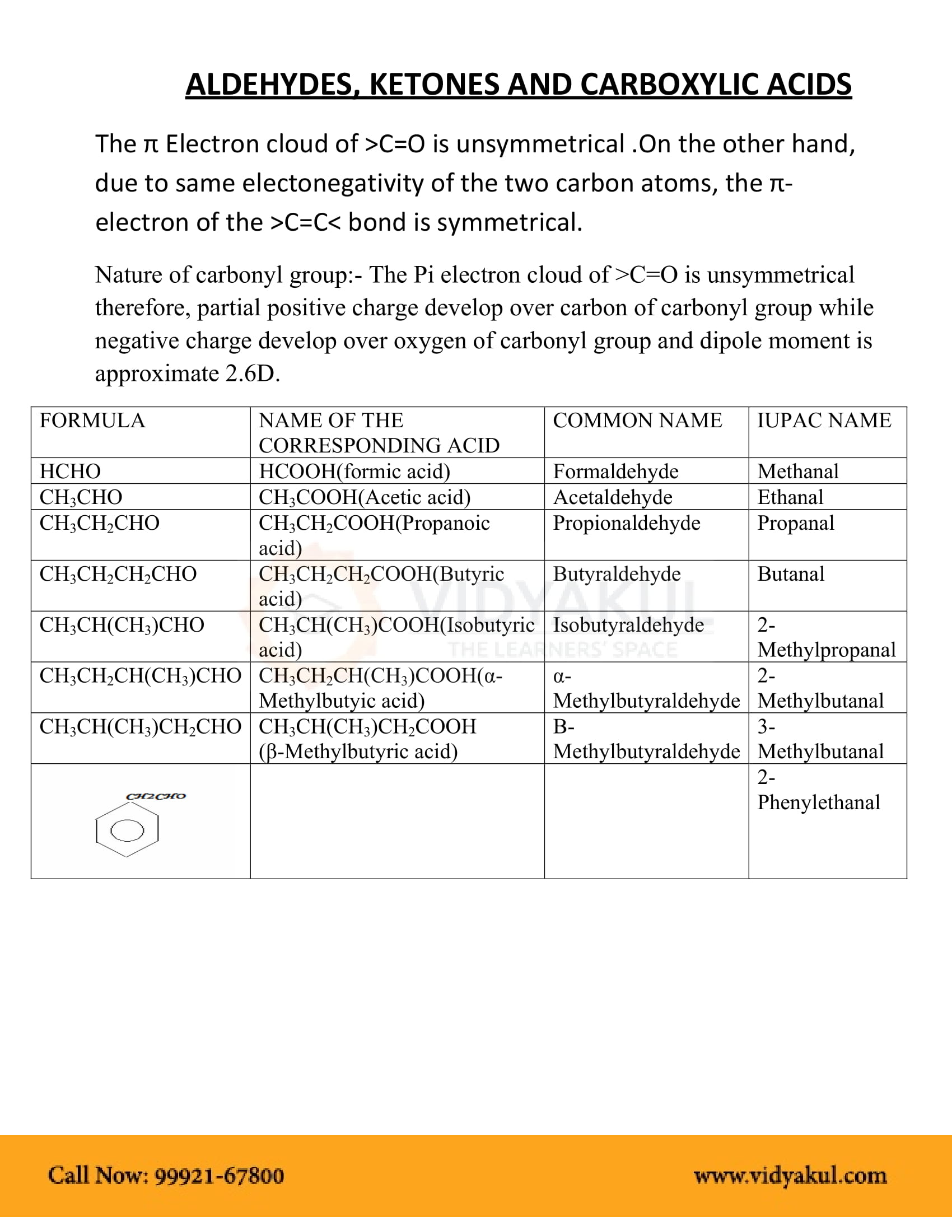
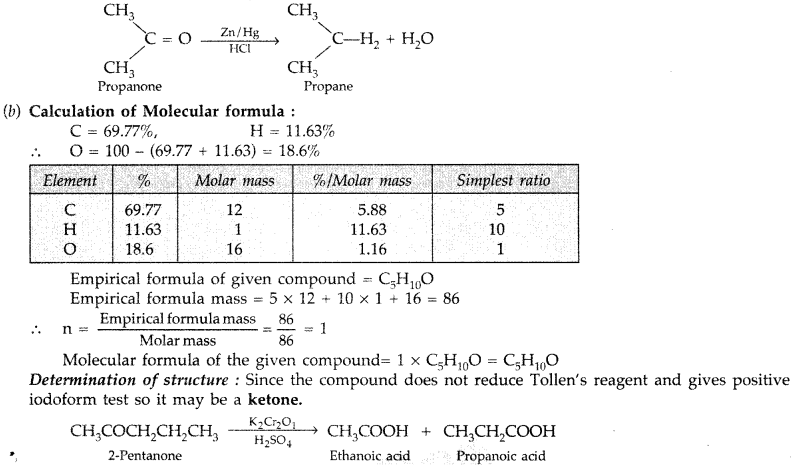
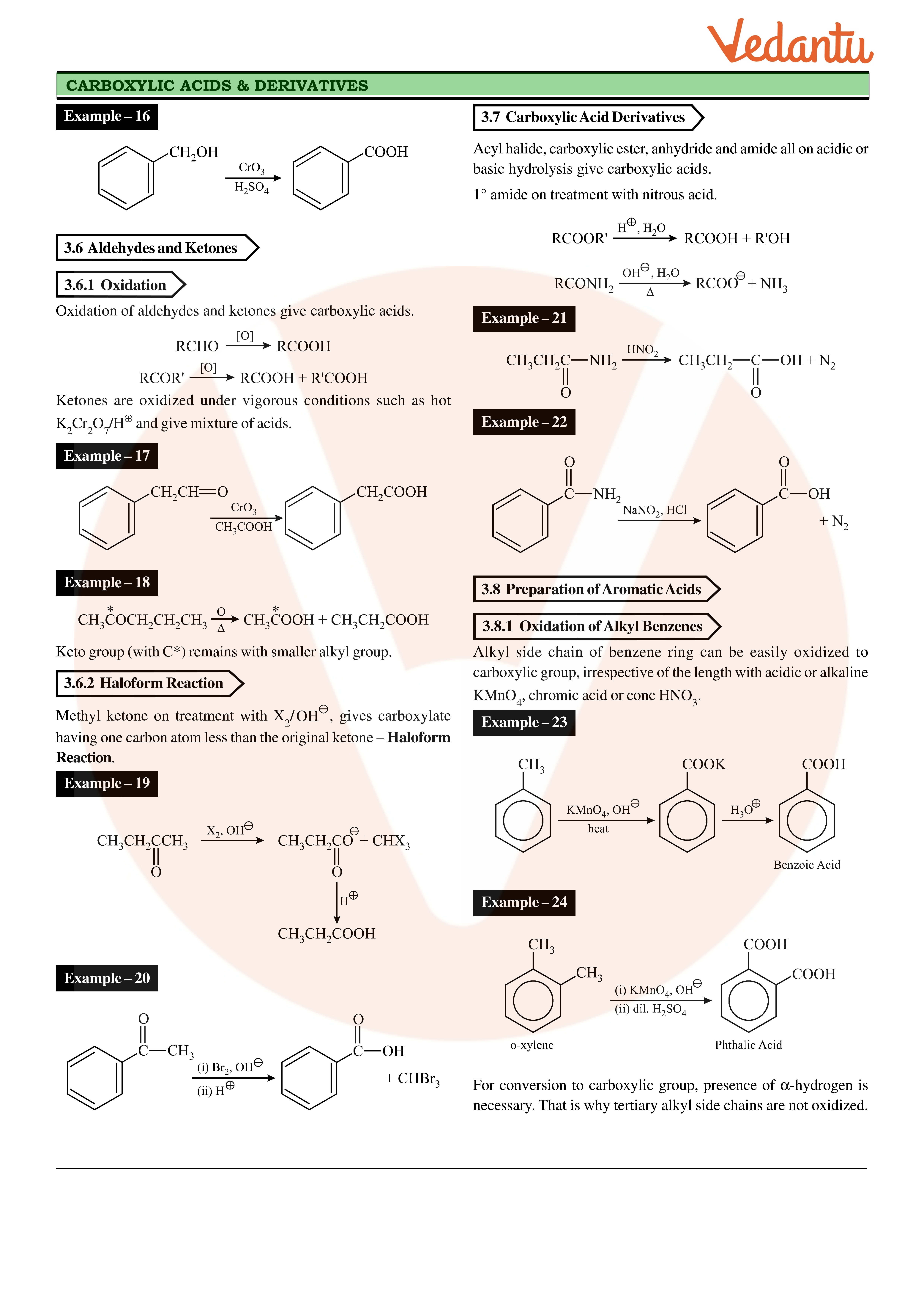
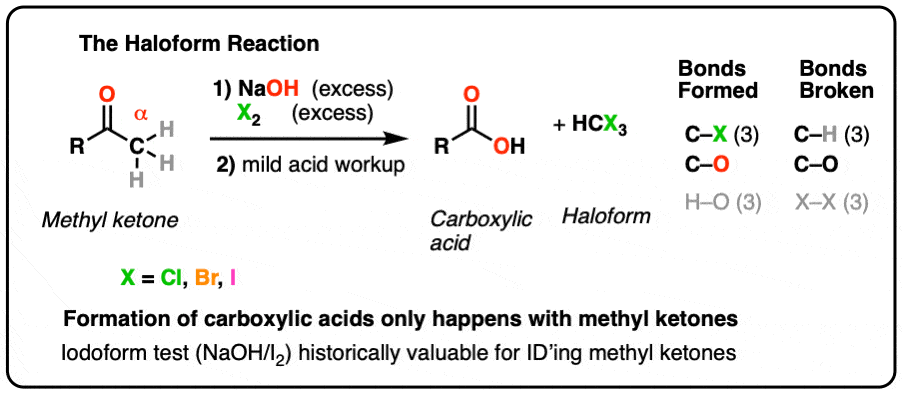
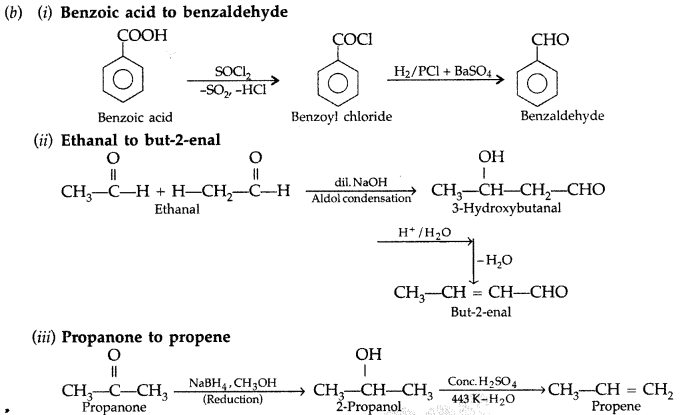
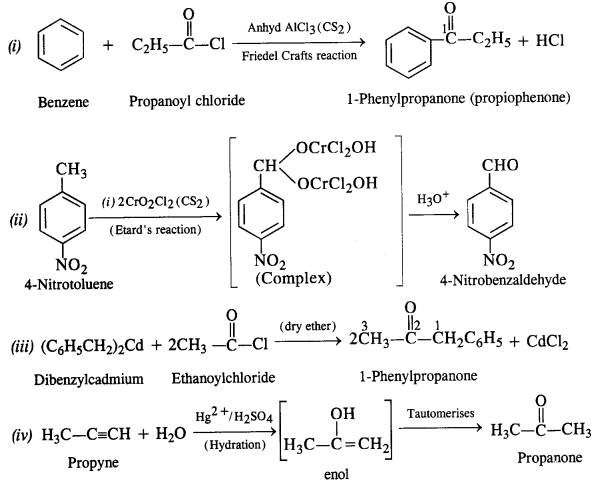
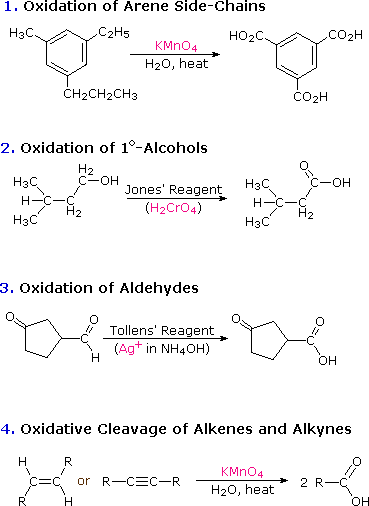
test to distinguish between carboxylic acid and ketone
|
Chemical tests to distinguish carbonyl compounds
can be used to help identify the specific aldehyde or ketone after purifying |
|
Testsforfunctionalgroups - inorganiccompounds
To identify the functional groups present in an organic compound. I. TESTS FOR to carboxylic acids while ketones require relatively stronger. |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
Aldehydes ketones and carboxylic acids are widespread in plants 12.13 Give simple chemical tests to distinguish between the following pairs of ... |
|
S42.CHP:Corel VENTURA
We tested the ability of human subjects to distinguish between aliphatic odorants 1-Alcohols n-aldehydes |
|
Lab 14: Qualitative Organic Analysis
Organic chemists often must identify unknown compounds. one major functional group (alcohol ketone |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
Aldehydes ketones and carboxylic acids are widespread in plants 12.13 Give simple chemical tests to distinguish between the following pairs of ... |
|
Topic 13
DISTINGUISHING BETWEEN DIFFERENT ORGANIC COMPOUNDS test for carboxylic acids ... distinguishing tests for primary secondary and tertiary alcohols. |
|
Tests for functional groups
Once you have confirmed an alcohol is present you may need to identify it There is no single test for a carboxylic acid |
|
Identifying an Unknown Compound by Solubility Functional Group
In some cases these tests will be sufficient to identify the functional group(s) such as alcohols |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn carbon chain in between the two carboxyl groups ... and ketones |
|
Identifying an Unknown Compound by Solubility Functional
A pH of 4 or lower indicates a carboxylic acid A pH of 8 or higher indicates an amine Water-soluble compounds are tested with 5 sodium hydrogen carbonate (NaHCO3) to determine whether or not they are carboxylic acids Carboxylic acids react with NaHCO3 to produce carbon dioxide bubbles as shown below in Equation 3 |
|
Searches related to test to distinguish between carboxylic acid and ketone PDF
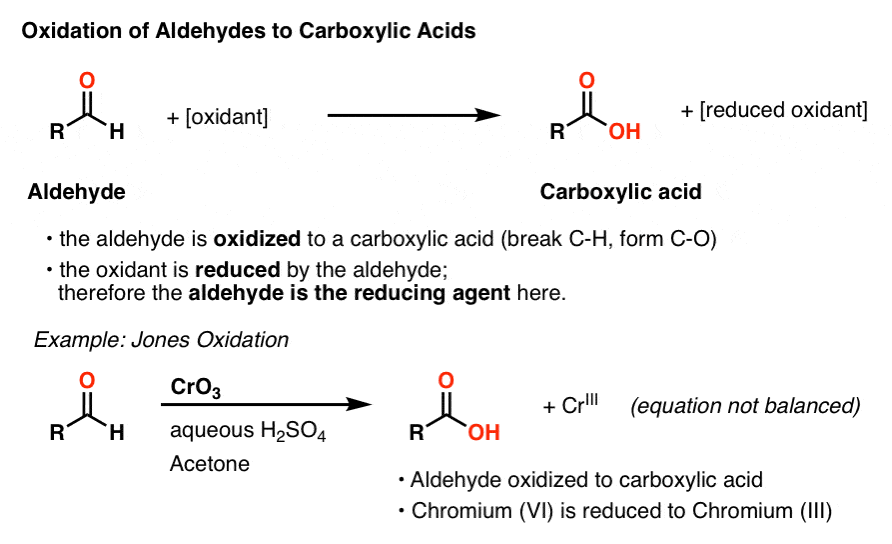
Sep 7 2014 · ketones are somewhat polar but not extremely polar they are good solvents for organic reactions Oxidation Aldehydes can be oxidized to carboxylic acids by almost any oxidizing agent Some common oxidizing agents are chromic acid Benedict’s reagent and Fehling’s reagent |
Common Misconceptions
Be aware of misconceptions learners may have around this practical, for example: 1. Learners often think molecules only contain one functional group and don’t appreciate that in fact many organic molecules contain more than one functional group. To overcome this belief, learners should be exposed to compounds that have more than one functional grou...
How do you identify a carboxylic acid from aldehydes and ketones?
This test is used to identify a carboxylic acid from aldehydes and ketones. Carboxylic acids undergo acid-base reaction with carbonate and hydrogen carbonates to produce salt, water and carbon dioxide. No acid and base reactions occur between an aldehyde/ketone and carbonates.
Which reagent is used to detect aldehydes and ketones?
e) use of Tollens’ reagent (aammoniacal silver nitrate) to: detect the presence of an aldehyde group ; distinguish between aldehydes and ketones, explained in terms of the oxidation of aldehydes to carboxylic acids with reduction of silver ions to silver. 6.3 Analysis 6.3.1 Chromatography and qualitative analysis
Does permanganate react with ketone?
Permanganate will oxidise an aldehyde into a carboxylic acid but does not react with ketone. This test is used to identify a carboxylic acid from aldehydes and ketones. Carboxylic acids undergo acid-base reaction with carbonate and hydrogen carbonates to produce salt, water and carbon dioxide.
What is a positive test for carboxylic acid?
A positive test for carboxylic acids is the formation of bubbles or frothing (Figure 6.52). Figure 6.52: a) Negative result (acetone), b) Positive result (lactic acid), c) Positive result (octanoic acid). A solution of bromine in CH 2Cl 2 is a test for unsaturation (alkenes and alkynes) and in some cases the ability to be oxidized (aldehydes).
|
Chemical tests to distinguish carbonyl compounds - WordPresscom
1: Detecting an aldehyde or ketone Aldehydes and ketones react with 2,4-dinitrophenylhydrazine (2,4-DNP or 2,4-DNPH) to form an orange or yellow precipitate No precipitate is formed with other carbonyl compounds such as carboxylic acids or esters |
|
Tests for functional groups
i) Methyl ketones (RCOCH3) – the iodoform (triiodomethane) reaction Methyl ketones undergo the iodoform reaction (for experimental details see methyl secondary alcohols) This distinguishes carboxylic acids from weaker acids, such as phenol, which do not react with sodium carbonate |
|
Lab 14: Qualitative Organic Analysis - California State University
identify a solvent system that will separate your unknown from any impurities After you have Both strong and weak acids (Carboxylic acids and phenols) will be C-14 Tests for the presence of esters Ketones 2,4-Dinitrophenylhydrazine |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
Aldehydes, ketones and carboxylic acids are widespread in plants 12 13 Give simple chemical tests to distinguish between the following pairs of compounds |
|
7-Aldehydes, Ketones, C Acids
Experiment 7 – Aldehydes, Ketones, and Carboxylic Acids Aldehydes of each compound in its own separate test tube the stirring rod between solutions |
|
Organic Chemistry
Name alkyl halides, alcohols, phenols, aldehydes, ketones, carboxylic acids and Perform reactions typical of alcohols and distinguish between aliphatic Use basic diagnostic tests and prepare synthetic derivatives for the purpose of |
|
Identification of an Unknown – Alcohols, Aldehydes, and Ketones
precipitates, can be used to differentiate alcohols, aldehydes, and ketones, and also to provide (Aldehydes oxidize to carboxylic acids in the presence of |
|
27 ALDEHYDES, KETONES AND CARBOXYLIC ACIDS - NIOS
distinguish between aldehydes and ketones on the basis of certain reactions and tests based on them; ○ give IUPAC names of carboxylic acids; ○ |
|
Identification of Aldehydes and Ketones
General test (2,4-Dinitrophenylhydrazine reagent) Both aldehydes Differentiation between aldehydes and ketones is achieved by taking the advantage carboxylic acid with an accompanying reduction of the silver ions from this reagent to |
|
Chemical Tests for Unknowns - Chemistry
Lucas test 2o, 3o, and benzylic alcohols cloudy solution or separate layer Tollen's carboxylic acids and secondary alcohols to ketones containing an oxygen (alcohol, aldehyde, ketone, etc) so it isn't useful for distinguishing ethers from |