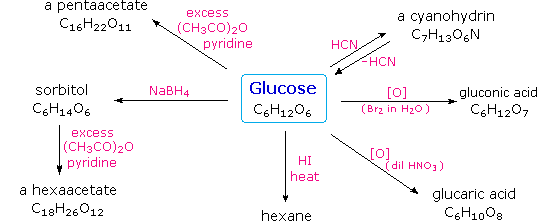
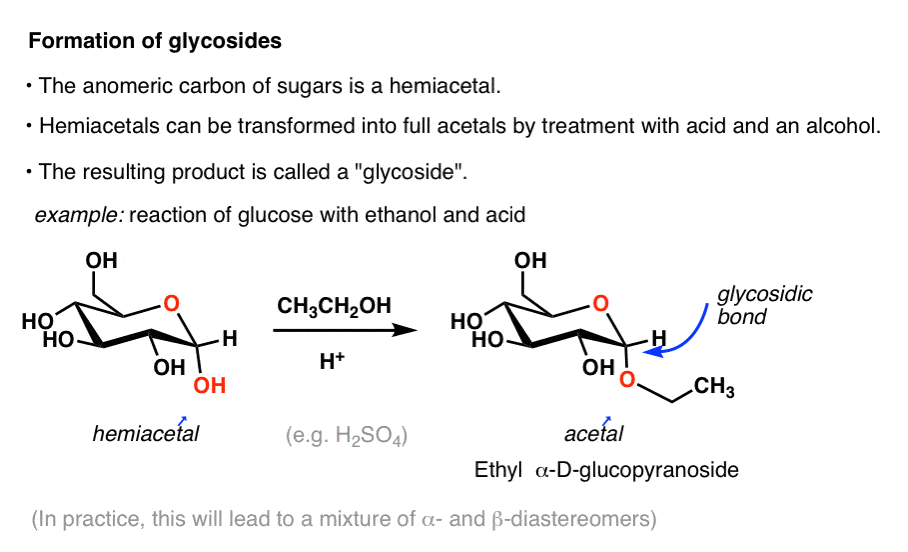
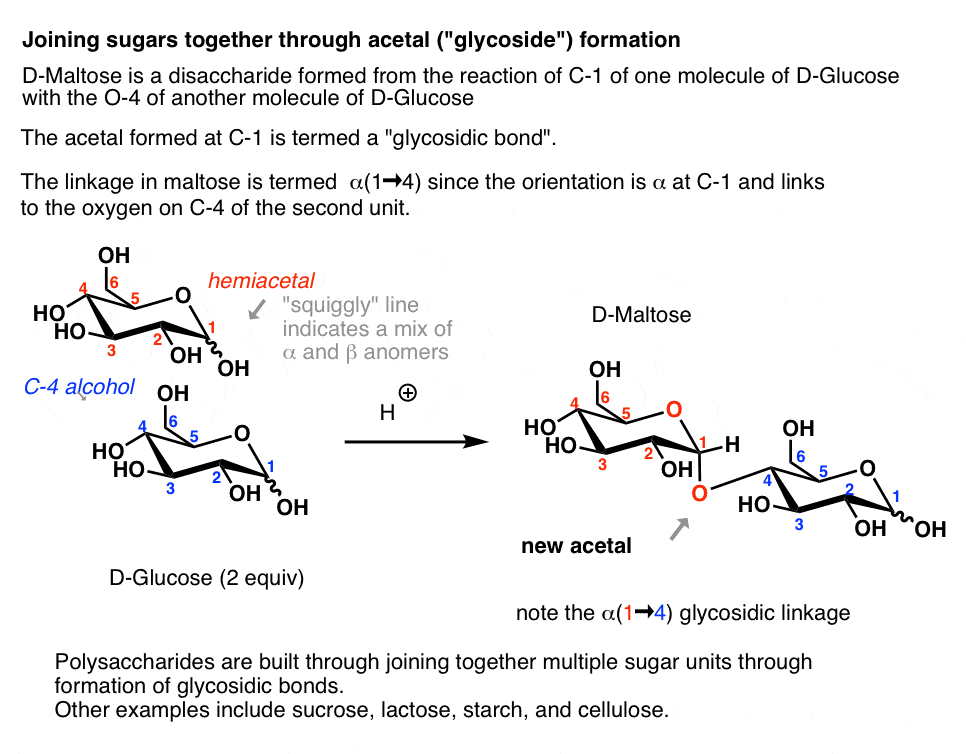
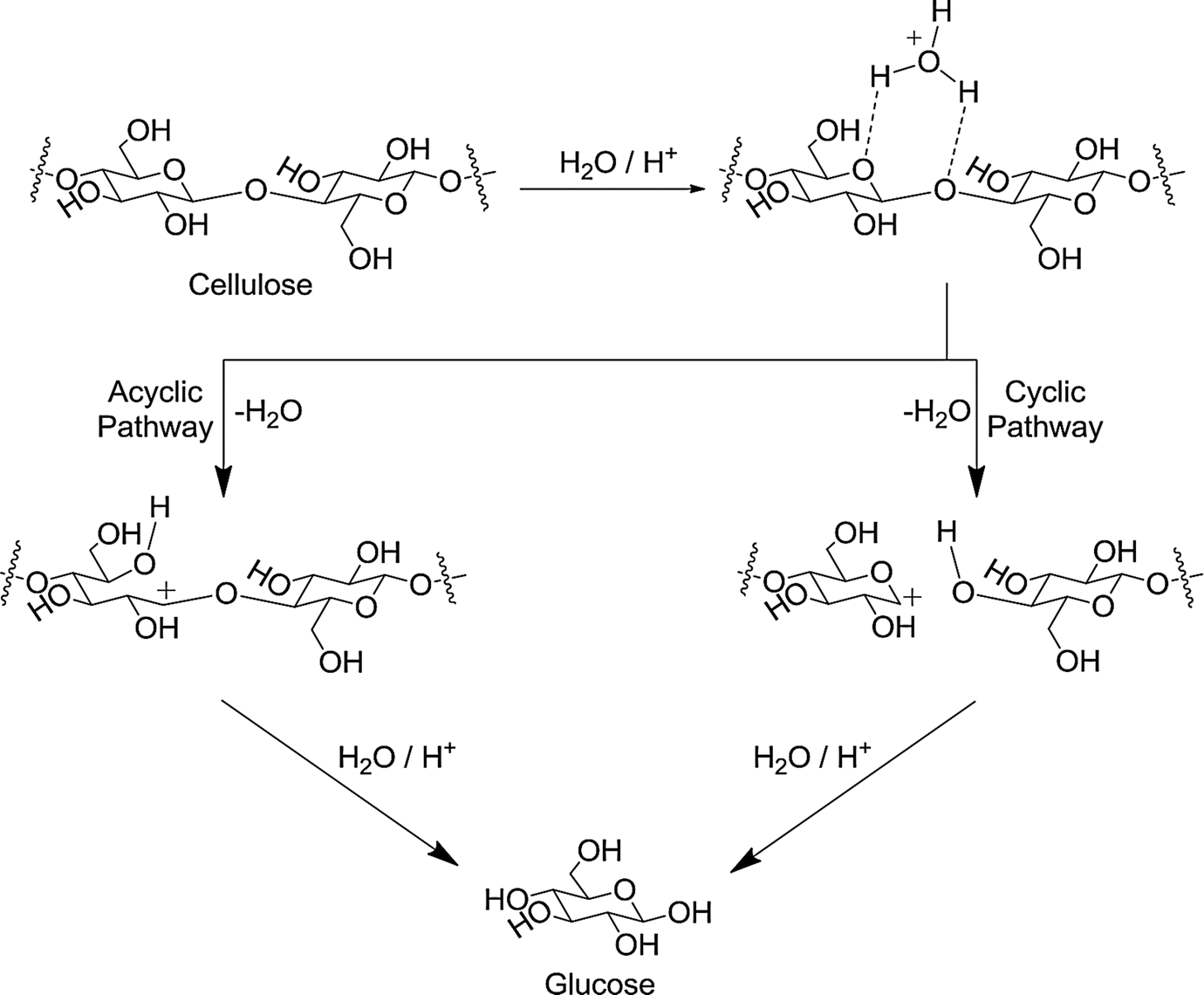
the acid catalyzed hydrolysis of sucrose occurs by the following overall reaction
|
Hydrolysis of Sucrose over Sulfonic Acid Resins
The acid splitting of sucrose as first-order reaction occurs at the fructosyl oxygen[56] and only In the following |
|
Levansucrase of Bacillus subtilis: Kinetic and
tosylase which catalyses mainly the following reaction. Sucrose + Acceptor -P acid and enzymatic catalysis of sucrose hydrolysis. The acid hydrolysis of ... |
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
There are two general mechanisms for acid-catalyzed. Although reactions which follow either mechanism can he made to follow pseudo-first order kinetics |
|
HYDROLYSIS 2016.pdf
In general hydrolysis occurs via one of two classes of mechanisms; Acid catalyzed pre-equilibrium creates more electrophilic reaction centre. |
|
Efficient Production of Acetic Acid from Nipa (Nypa fruticans) Sap by
7 janv. 2016 Keywords: Sucrose hydrolysis Oxalic acid |
|
The oxygen-18 isotope shift in carbon-13 nuclear magnetic
The acid-catalyzed hydrolysis of sucrose has a rich history. It of a carbon-oxygen bond although general acid catalysis of the hydrolysis has also been ... |
|
Fundamentals of Chemical Reaction Engineering
6 mars 1999 If this reaction would occur by Hz interacting directly with Brz to ... acid over a mercuric chloride catalyst at 500 K and 5.0 atm total ... |
|
Deuterium isotope effects in a - ? pyranose and in pyranose
same situation exists in certain other reaction systems. Examples are (a) the acid hydrolysis of sucrose [23];. (b) the base-catalyzed enolization of |
|
GENERAL ACID CATALYSIS IF ACETAL HYDROLYSIS
It seemed not unreasonable to assume that the same process occurs also in the hydrolysis reaction. Breasted and Hamnett linear free energy relatronsnips were |
What type of catalyst is used in acid catalysis?
In acid catalysis, the catalyst is usually a ext {H}^+ H+ ion. In base catalysis, the catalyst is usually an ext {OH}^- OH? ion. An example of a reaction that can be catalyzed by acid is the hydrolysis of sucrose, also known as table sugar. Sucrose is a combination of two simpler sugars (or monosaccharides ), glucose and fructose.
Is sucrose a reducing sugar?
There are no hemiacetals remaining in the sucrose and therefore sucrose is a non-reducing sugar. Sugar or more specifically sucrose is a carbohydrate that occurs naturally in every fruit and vegetable. It is the major product of photosynthesis, the process by which plants transform the sun's energy into food.
How does water break the acetal bond in hydrolysis?
In the hydrolysis of any di- or poly saccharide, a water molecule helps to break the acetal bond as shown in red. The acetal bond is broken, the H from the water is added to the oxygen on the glucose. The -OH is then added to the carbon on the fructose.
How do you know if sucrose is glucose or fructose?
Sucrose is made from glucose and fructose units. The glucose and fructose units are joined by an acetal oxygen bridge in the alpha orientation. The structure is easy to recognize because it contains the six member ring of glucose and the five member ring of fructose. To recognize glucose look for the horizontal projection of the -OH on carbon #4.
Past day
Types of catalysts (article) | Kinetics | Khan Academy
The acid-catalyzed reaction to form glucose and fructose from sucrose, which is also known as table sugar In the first step, sucrose reversibly reacts with text H^+ H+ (in red), to form protonated sucrose. The protonated sucrose reversibly reacts with water (in blue) to give text H^+ H+, one molecule of glucose, and one molecule of fructose. lgo algo-sr relsrch fst richAlgo" data-67c="645fa5a99edb3">www.khanacademy.org › science › chemistryTypes of catalysts (article) | Kinetics | Khan Academy www.khanacademy.org › science › chemistry Cached
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
There are two general mechanisms for acid-catalyzed Although reactions which follow either mechanism can he made to follow pseudo-first order kinetics, the |
|
Inversion of Sucrose
Purpose: The rate of reaction between sucrose and water catalyzed by hydrogen ion is followed by measuring the used as the catalysts and the acids will be compared hydrolysis of sucrose into single sugars is necessary before the sugars can be metabolized In In the equations that follow we will set [sucrose] = c |
|
Acid-Catalyzed Inversion of Sucrose in the Amorphous State at Very
Factors affecting the solid-state acid-catalyzed inversion of That such a reaction can occur in the presence of acidic amorphous sucrose to glucose and fructose |
|
Sucrose Inversion: An Experiment on Heterogeneous Catalysis* - IJEE
The experiment deals with the acid-catalyzed sucrose inversion, performed in Sucrose under- goes acid hydrolysis into glucose and fructose Moreover, no change in the overall reaction rate was noted as present The following expression, for the ratio of occurs due to axial and radial dispersion in the coiled tube |
|
HYDROLYSIS
In general, hydrolysis occurs via one of two classes of Hydrolysis reactions are generally enhanced by both acids and bases and three independent The following data can be interpreted in terms of these factors and Epoxides undergo hydrolysis by neutral and acid catalyzed mechanisms under environmentally |
|
Sucrose and Corn Fiber Hydrolysis using a Succinic Acid Catalyst
The hydrolysis of sucrose by succinic acid was investigated in a preliminary study It was found with general E coli Figure 2- takes place at different rates and during the reaction the sugars can be degraded into by- products acid production include the following operations: centrifugation, evaporation, crystallization |
|
Rate of reaction Details of process from reactants to products
Study of how rapidly reactions proceed - rate of reaction Details of process For a general reaction: aA + bB --> xC + The following mechanisms have been proposed Based on For a reaction which occurs in a single elementary step NO + O3 NO2 for catalyzed acid hydrolysis of sucrose: 36 kJ/mol Rate increase of |
|
Complete sucrose hydrolysis by heat-killed recombinant - CORE
18 jui 2014 · Commercial invertase from the yeast Saccharomyces cerevisiae is thermolabile and suffers from substrate inhibition Thermotoga maritima β- |


![PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a](https://lightcat-files.s3.amazonaws.com/problem_images/d54a89c18f9e6e44-16.98.PNG)