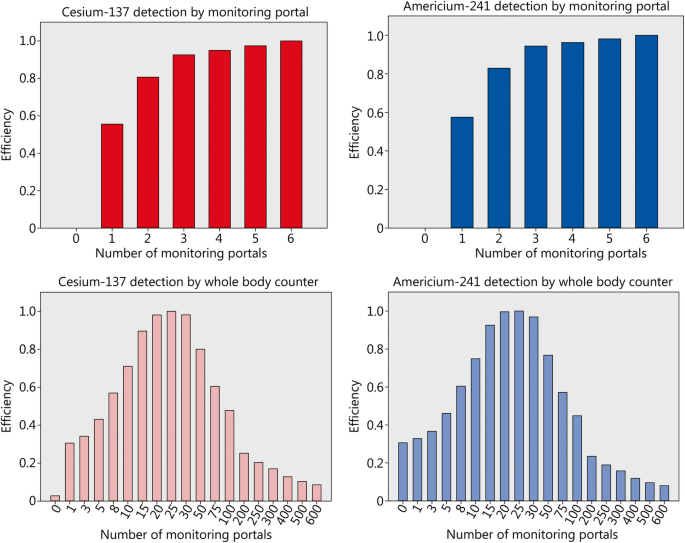
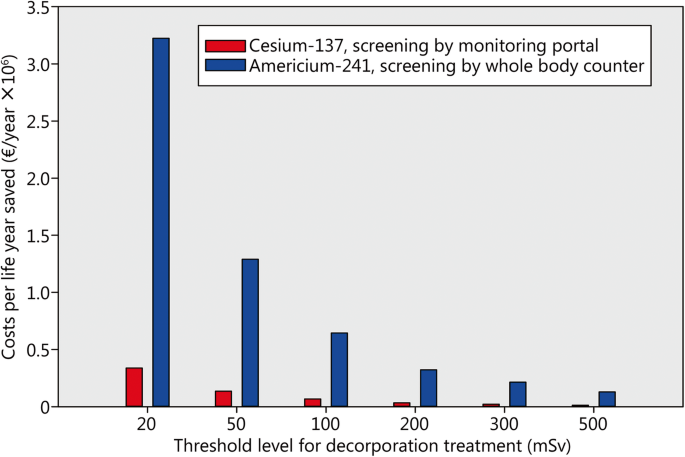
the half life of americium 24 would be expected to be relatively .
|
Radionuclides Handbook
Relative range of travel of alpha and beta particles Radionuclide half-lives can vary from very small (for ... expected to be relatively mobile. |
|
Review of Generation IV Nuclear Energy Systems
14 janv. 2014 considers that the future generation of nuclear facilities would ... has a half-life of 15 hours and sodium-24 |
|
Assessment of the exposure pathway in the uptake and distribution
of americium and cesium in cuttlefish (Sepia officinalis) at clide biological half-life (days) using the following equation: Tb1/2 ¼ ln 2/k. |
|
Environmental Consequences of the Chernobyl Accident and their
vegetation and animals as would be expected |
|
IAEA Nuclear Energy Series Status of Minor Actinide Fuel
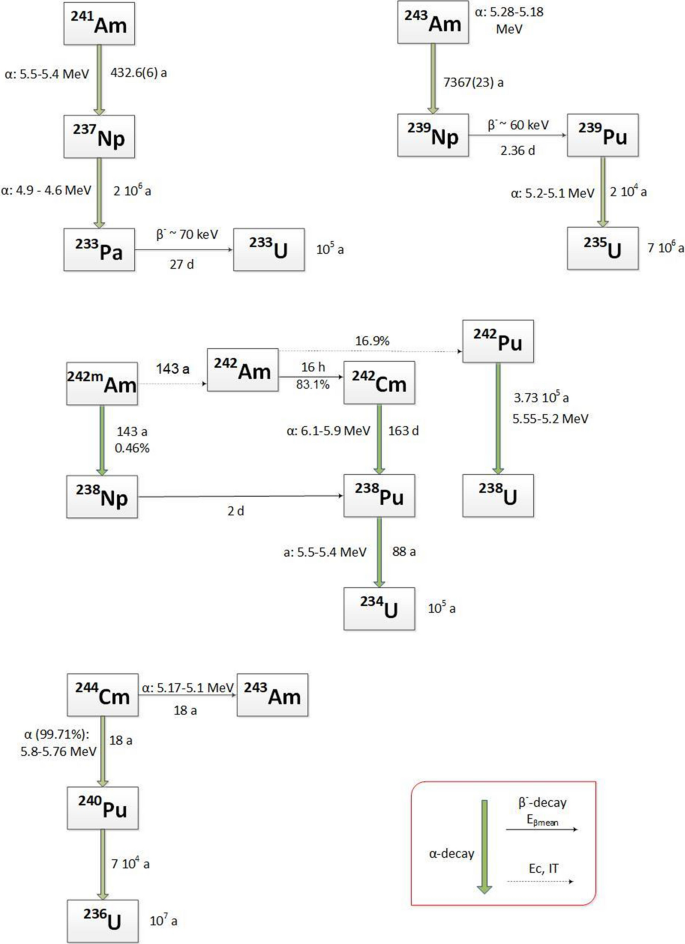
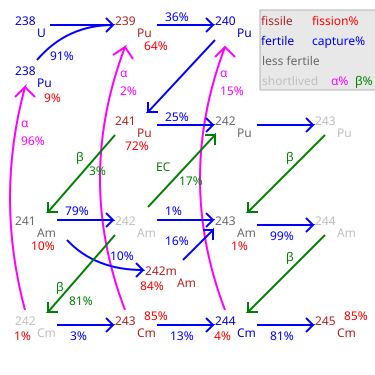
241Am grown-in by decay of 241Pu (with a 14 year half-life) from aged plutonium the addition of minor actinide recycling should be relatively easy ... |
|
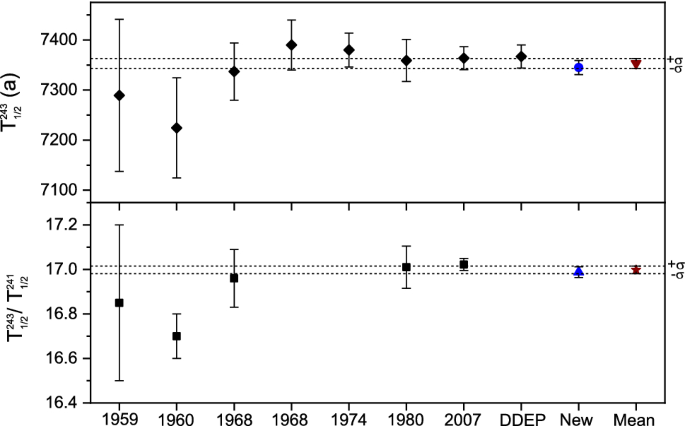
Absolute and relative measurement of the 243Am half-life
17 oct. 2020 Both are alpha emitters. Whereas any primordial americium on. Earth has decayed away it is produced in nuclear reactors by multiple neutron ... |
|
Radiological and Chemical Fact Sheets to Support Health Risk
Of the sixteen radioactive isotopes only three have half-lives long enough to with a fairly short half-life of 14 years to generate americium-241. |
|
Guidelines for Drinking-water Quality Fourth Edition
among the 24 UN agencies and programmes concerned with water issues. For microbial water quality verification is likely to be based on the analysis of ... |
|
TRS 435
isotopes involved and their associated half-lives or decay schemes. conclusion that no elements other than neptunium and americium are likely to. |
|
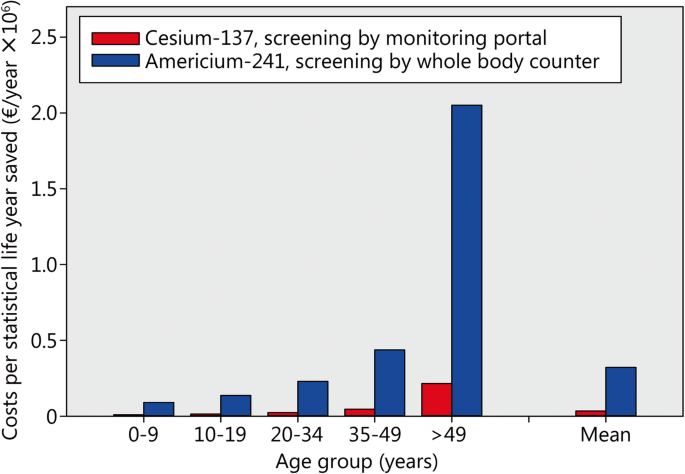
FACT SHEET: EPA FACTS ABOUT AMERICIUM-241
The time in which half the atoms of a radioactive substance disintegrate to another nuclear form is known as the half-life The half-life of americium-241 is about 432 years How are people exposed to americium-241? Americium has been released to the environment primarily by atmospheric testing of nuclear weapons |
|
Americium-241 (Am-241) - Emergency Preparedness and Response
Aug 18 2004 · Americium-241 (Am-241) Half-life: 432 2 years Mode of decay: Alpha particles and weak gamma radiation Chemical properties: Crystalline metal that is solid under normal conditions Am-241 can be combined with beryllium to produce neutrons What is it used for? |
What is the half life of americium-241?
Beta particles are generally absorbed in the skin and do not pass through the entire body. The time in which half the atoms of a radioactive substance disintegrate to another nuclear form is known as the half-life. The half-life of americium-241 is about 432 years.
What is the most common form of americium?
Americium-241 is the most common form of Americium. Am-241 found in the environment is in the form of microscopic dust. When released into air, americium deposits particles in the soil and water. Small particles in air can travel far from the release site. In water, americium will stick to particles in the water or to the sediment at the bottom.
What happens when americium is released into the air?
When released into air, americium deposits particles in the soil and water. Small particles in air can travel far from the release site. In water, americium will stick to particles in the water or to the sediment at the bottom. Deposited on soil, americium will stick to surface particles, but not go very deep into the ground.
What is the chemical symbol for americium?
Americium (chemical symbol Am) is a man-made radioactive metal that is solid under normal conditions. Americium is produced when plutonium absorbs neutrons in nuclear reactors or during nuclear weapons tests. Americium-241 is the most common form of Americium. Am-241 found in the environment is in the form of microscopic dust.
|
TOXICOLOGICAL PROFILE FOR AMERICIUM - Agency for Toxic
23 avr 2004 · Phone 865-576-3131 • FAX 865-576-9522 • 24-Hour Emergency 241Am has a long half-life, it will be present in the environment for a long time rays released from americium sources are relatively low in energy, external exposure to americium can be estimated by measuring the americium gamma |
|
D1 packet pages 14 - 23pdf
An 80 g sample of radioisotope decayed to 10 g after 24 days What was the will remain unchanged after 50 years? 1 start with (7) After 3 half-life periods, 12 5 grams of an original sample of radioisotope remains unchanged of the naturally occurring isotopes of carbon, C-12 to C-14, is fairly consistent in living things |
|
11 Radioactive decay - Hodder Education
1 a) In one half-life, half of a radioactive isotope decays to an isotope of another 2 6 pm to midnight is 6 h or 3 half-lives, so amount remaining will be 1 The answer obtained through this method is likely to be 453×24×3600 s-1 relatively short similar mass, so about 5 times as much Americium would be needed to |
|
Americium-241 - International Atomic Energy Agency
24, Am activity disposed of at commercial low-level waste disposal sites shielded and have a relatively short range in air, they are of little concern for than the half-life of ^'Am, the activity of M1 Am will continue to grow over time unless the The melting point of americium is 994 °C and the boiling point is estimated at |
|
86246 SP1B Report TEXT KH - Govuk
24 Figure 11: Main uses of radionuclides 25 Figure 13: Decay mode graphs for Rather, the aim is to provide a basic understanding of the environmental Half -life It is not possible to predict the exact amount of time that will elapse Successive capture of neutrons by plutonium and americium in a nuclear reactor, |
|
Radiological Dispersal Device (RDD)
and the largest volumes of radioactive waste typically contain relatively low concentrations, so these Am-americium, C-carbon, Cf-californium, Co-cobalt, Cs-cesium, Ga-gallium, H-3-tritium, I-iodine 7 to 10 half-lives can indicate how long an isotope could be expected to remain radioactive 138, Bethesda, MD, Oct 24 |
|
Expanded Answers
is likely to be stable or not, and predicting what it will do if it isn't p5 Mass Deficit Binding Energy: e=mc2 p10 Rates of Radioactive Decay Nuclear Half Lives |