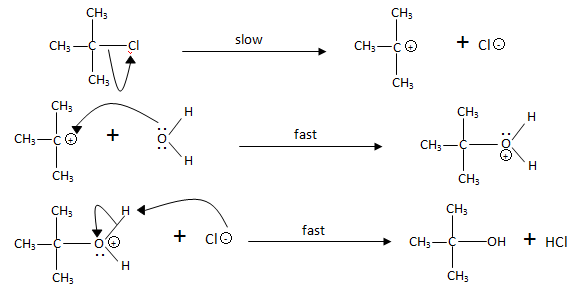
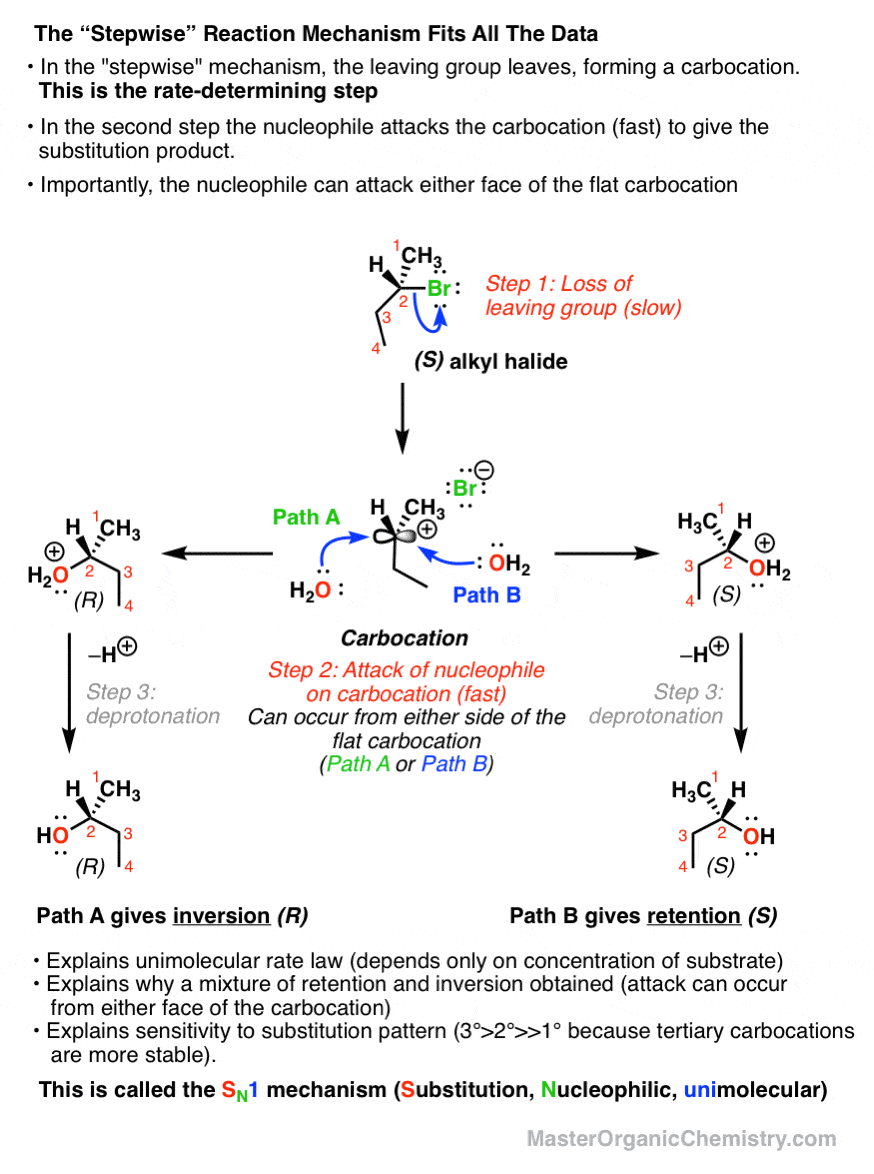
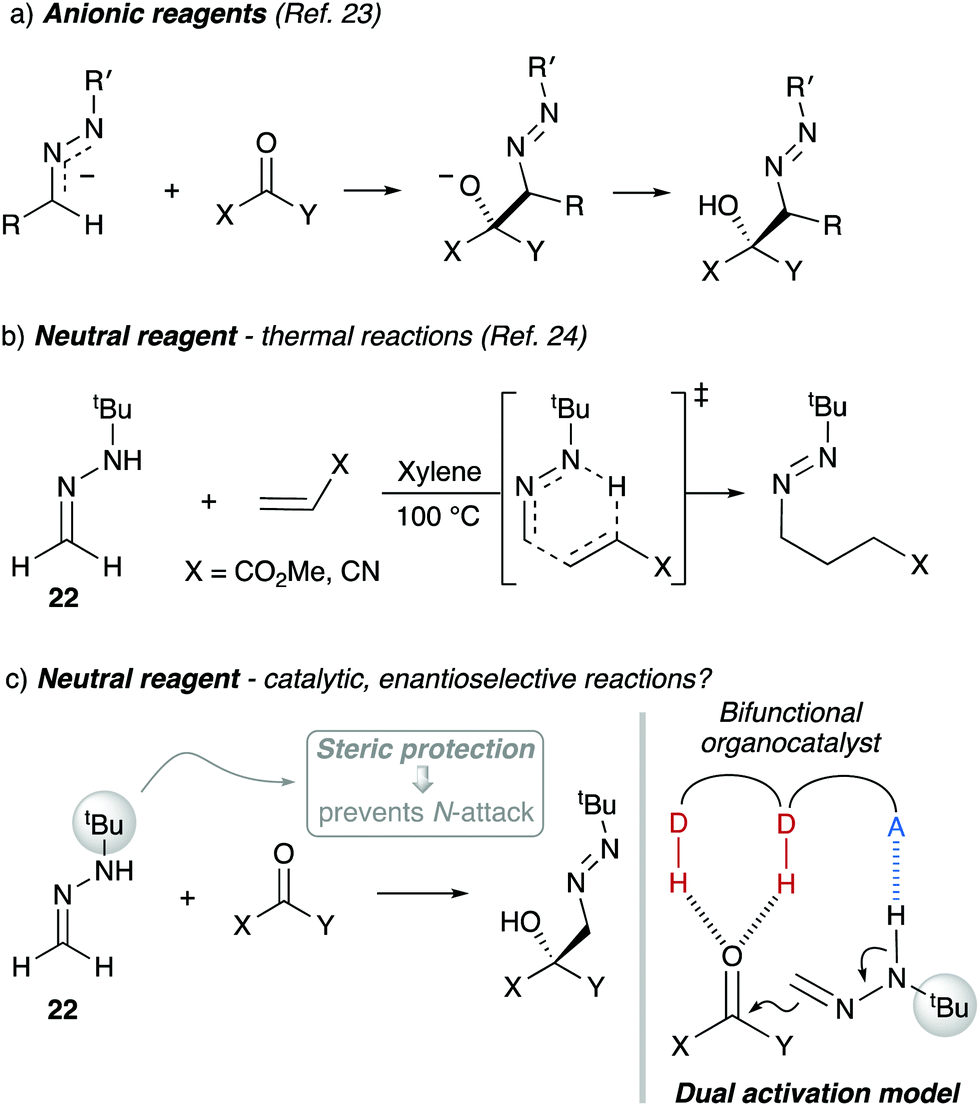
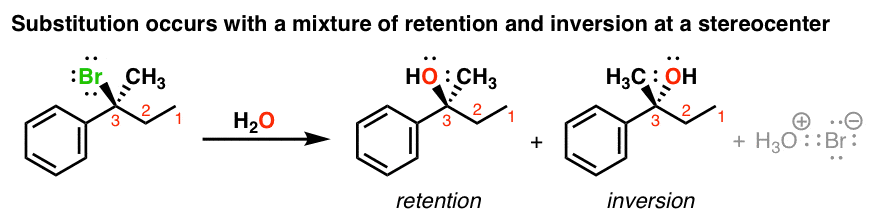
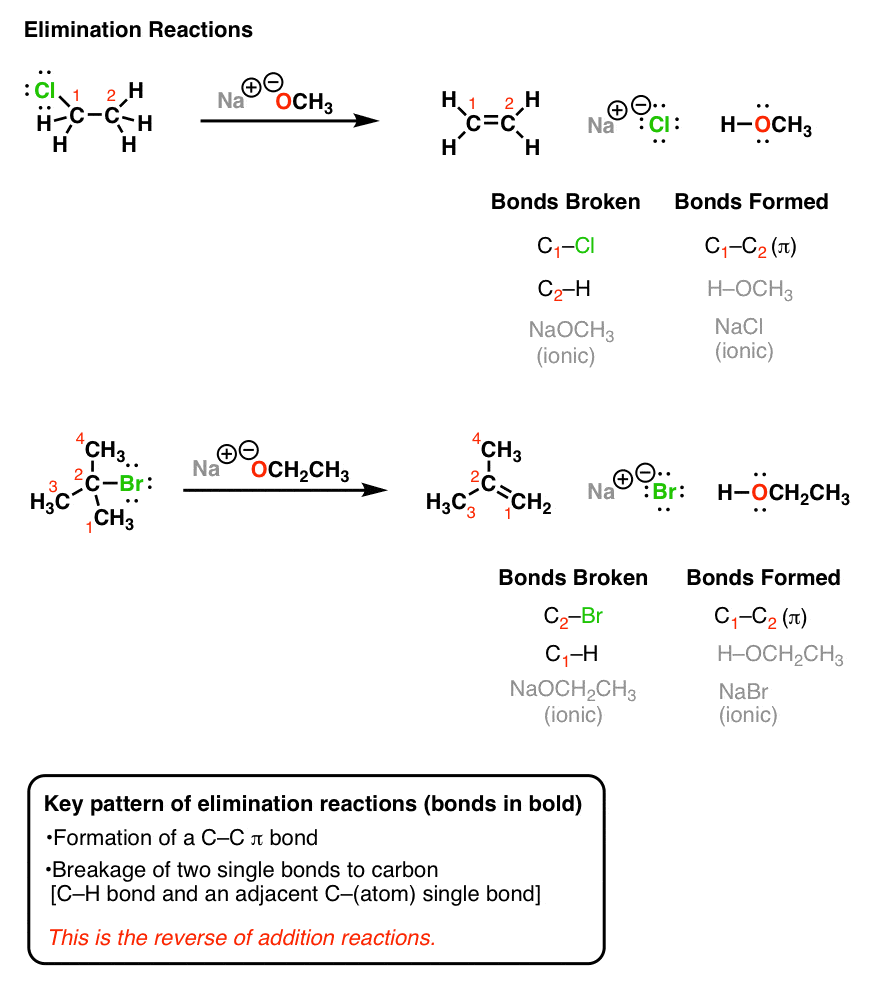
the hydrolysis of tert butyl chloride is given in the reaction below
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride
It is important to verify that students are using the stated number of equivalents. Solvent Free Conditions and Use of Brine. This experiment was run solvent- |
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride
It is important to verify that students are using the stated number of equivalents. Solvent Free Conditions and Use of Brine. This experiment was run solvent- |
|
THE HYDROLYSIS OF t-BUTYL CHLORIDE
30-Jan-2007 To verify a proposed mechanism for the hydrolysis of t-Butyl Chloride. ... The reaction of t-butyl chloride with water proceeds as shown in ... |
|
SN1 Reaction: Hydrolysis of tert-butyl chloride
Discussion: This experiment demonstrates a typical SN1 reaction (shown below) between water and tert-butyl chloride. In this demonstration the initial. |
| [I 9571 N.ucleo$hilic SubstitNtion Reactions of Benzyl Halides. Part |
|
PRACTICE EXERCISE – ORGANIC CHEMISTRY I Alkynes
41) Provide the major organic product of the reaction shown below. 39) The t-butyl bromide would not undergo Sn2 when treated with the intermediate ... |
|
Mechanism of Sabstitdion at a Satwated Carbovt Atom. Part VIII
discussed in relation to recent theories of reaction kinetics in solution. 255) it was shown that the hydrolysis of tert.-butyl chloride in aqueous ... |
|
Experiment 3 Preparation of tert-Butyl Chloride
We will also use a separatory funnel the use and purpose of a drying agent and the technique of distillation. The reaction is shown in Figure 3.1. |
|
Hydrolysis of t-butyl chloride CSTR-Study
20-Oct-2013 The reaction mechanism of t-butyl chloride hydrolysis is shown in figure 1. The rate determing step of the mechanism is the formation of ... |
|
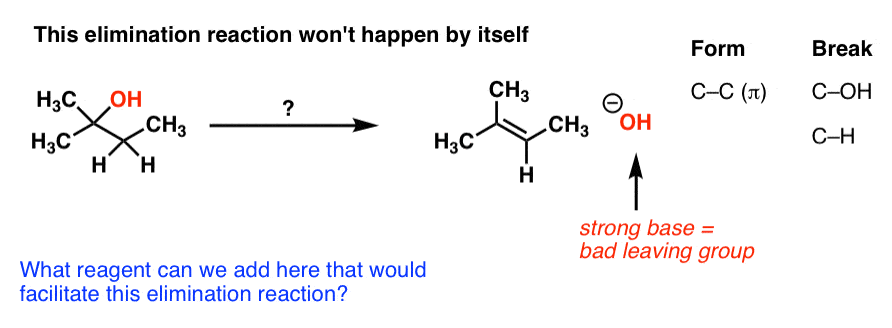
ORGANIC CHEMISTRY I – PRACTICE EXERCISE Elimination
4) Which diastereomer of 1-bromo-4-t-butylcyclohexane the cis or the trans 25) Propose a detailed |
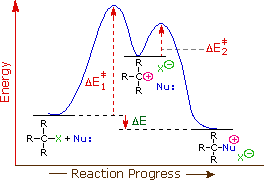
Is the hydrolysis of tert-butyl chloride an S N 2 reaction?
Although the hydrolysis of tert-butyl chloride, as shown above, might be interpreted as an S N 2 reaction in which the high and constant concentration of solvent water does not show up in the rate equation, there is good evidence this is not the case.
What factors affect the reaction rate of tert-butyl chloride?
The reaction rate can be affected by certain factors such as the addition of catalysts, concentration of reactants, physical states of reactants, and temperature such as in the reaction of the hydrolysis of tert-butyl chloride in this experiment. One method of studying chemical kinetics is by
What happens if the tert-butyl chloride solution is uncorked?
d. If the flask is uncorked while the tert-butyl chloride solution is being heated to complete the reaction, so that some of the tert-butyl chloride is lost by evaporation, this would affect the results we obtained for the rate constant. They would probably be precise but not accurate.
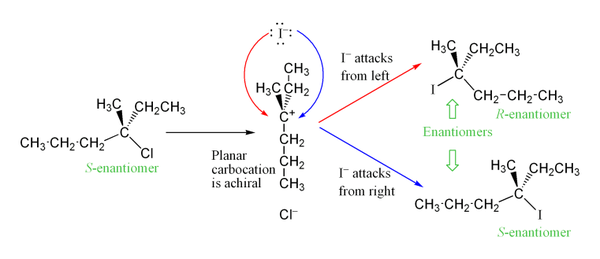
What are the characteristics of chiral 3o-alkyl bromide reaction?
It not only shows first order kinetics, but the chiral 3º-alkyl bromide reactant undergoes substitution by the modest nucleophile water with extensive racemization. In all of these features this reaction fails to meet the characteristics of the S N 2 mechanism. A similar example is found in the hydrolysis of tert-butyl chloride, shown below.
|
SN1 Reaction: Hydrolysis of tert-butyl chloride - NC State University
Discussion: This experiment demonstrates a typical SN1 reaction (shown below) between water and tert-butyl chloride In this demonstration the initial solution is |
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride - The Royal Society
ion tert-Butyl chloride Scheme SM 2 1 1 1 – Mechanism for the formation of tert- Butyl Chloride from tert-Butyl calculated, making note of the limiting reagent in this specific reaction Below is the pre-laboratory assignment that students were |
|
THE HYDROLYSIS OF t-BUTYL CHLORIDE
30 jan 2007 · The reaction of t-butyl chloride with water proceeds as shown in the two step reaction below: CH3 CH3 k STEP ONE: CH3 C Cl CH3 C + |
|
Physical Chemistry - School of Chemistry University of Leeds
Kinetics of a first order reaction (hydrolysis of t-butyl chloride) by measurements of conductivity radiation Atomic states are defined by the arrangement of electrons in atomic orbitals An the spectrometer slit, see picture below Switch it on |
|
Hydrolysis of t-butyl chloride CSTR-Study
20 oct 2013 · 1 Theory The reaction mechanism of t-butyl chloride hydrolysis is shown in figure 1 The rate determing step of the mechanism is the formation |
|
CSTR study: The reaction of t-butyl chloride and water
5 nov 2011 · Figure 1: Reaction mechanism for the hydrolysis of t-butyl chloride The formation of The experimental setup is shown and explained in Fig 3 |
|
Hydrolysis of t-butyl chloride - at NTNU
The reaction between tert-Butyl chloride (t-butyl chloride) and water is investigated at different conditions each other Figure 2 demonstrates the mechanism of t-butyl chloride hydrolysis Sample calculation to be shown in the appendix |
|
Experiment 8 — Kinetics of SN1 Solvolysis
reaction In this lab we will be measuring the rate of solvolysis of tert-butyl chloride as a function The rate of this 1st-order reaction can be expressed as the decrease in concentration of shown below and is yellow; in basic solution it is blue |