usp hand sanitizer
|
Usp-hand-sanitizer-ingredients.pdf
A Hand Sanitizer Resource. A collection of standards provided as a resource to assist with the challenges posed by COVID-19. Not official text. |
|
Supporting Quality Alcohol-Based Hand Sanitizer Production: USP
Around 183 individuals registered to participate in USP's Seminar on February 23 2021 |
|
Frequently Asked Questions: Alcohol-Based Hand Sanitizer During
28-Apr-2020 Why are the hand sanitizer formulations important? ... How were the formulations in the USP compounding hand sanitizer resource document. |
|
Hand Sanitizers and Updates on Methanol Testing
23-Feb-2021 General Background on Hand Sanitizer. ? Recent Safety Concerns and FDA Actions. ? Substitution. ? Methanol Testing Requirements for Drug ... |
|
EA993G Hand Sanitize Infographic_2022-03
Download and follow the Compounding Alcohol-Based Hand Sanitizer During COVID-19 Pandemic recommendations at usp.org/compounding. Packaging and Storage:. |
|
Supporting Quality Alcohol-Based Hand Sanitizer Production: USP
USP Seminar: Ensuring Quality Hand Sanitizer Production. During COVID-19 For Healthcare Professionals in the United States. Executive Summary. |
|
Ensuring Quality Hand Sanitizer Production During COVID-19
Sanitizer Production During. COVID-19 Seminar. Why Quality Hand. Sanitizer Matters. Nurisha Wade. Vice President Healthcare. Quality and Safety |
|
BRIEFING ?797? Pharmaceutical Compounding—Sterile
Compounding personnel perform antiseptic hand cleansing with an frequency with an approved disinfectant such as sterile 70% IPA. |
|
Ensuring Quality Hand Sanitizer Production During COVID-19
Alcohol-Based Hand Sanitizer Formulas 2018 USP https://www.papercitymag.com/culture/hand-sanitizer-how-find-compounding-pharmacies-. |
|
Ensuring Quality Hand Sanitizer Production During COVID-19
USP has services and programs to help! ? USP Ingredient Verification Program. ? USP Educational Courses. ? Documentary Standards (monographs & general |
|
Usp-hand-sanitizer-ingredientspdf
Excerpted USP-NF and FCC Standards: A Hand Sanitizer Resource A collection of standards provided as a resource to assist with the challenges posed by |
|
Supporting Quality Alcohol-Based Hand Sanitizer Production - USP
USP Seminar: Ensuring Quality Hand Sanitizer Production During COVID-19 For Manufacturers in the United States Executive Summary Alcohol-based hand |
|
Hand Sanitizers and Disinfectants Analysis of Alcohols Shimadzu
Shimadzu provides all of the analytical solutions for hand sanitizer USP/FCC Alcohol neutral spirits and disinfectants including GC FTIR HPLC and |
|
Compounding Alcohol-Based Hand Sanitizer During COVID-19
18 mar 2020 · The USP Compounding Expert Committee (CMP EC) provides the following recommendations for compounding alcohol-based hand sanitizers for use |
|
Plain Text - NLM Digital Collections
2 Alcohol-based hand sanitizer for purposes of this guidance can be prepared using alcohol or isopropyl alcohol (IPA) consistent with FDA policies outlined in |
|
HHS Letterhead ONLY Template - USP-NF
25 fév 2021 · This letter pertains to the modernization of the USP/NF Isopropyl Alcohol FDA continues to see an increasing number of hand sanitizer |
|
Illinois Department of Financial and Professional Regulation
alcohol-based hand sanitizers due to the COVID-19 outbreak The Department recommends following guidelines issued by the United States Pharmacopeia (USP): |
|
A Comparison of Measurement Methods for Alcohol-Based Hand
hand sanitizers should contain at least 60 alcohol (typically ethanol) as well as minimal quality-safety/usp-hand-sanitizer-ingredients pdf |
|
Temporary Policy for Preparation of Certain Alcohol-Based Hand
15 avr 2020 · The hand sanitizer formula should be adjusted based on the actual concentration of hydrogen peroxide used 11 If a firm wants to use or supply a |
|
FDA updates on hand sanitizers consumers should not use
FDA Warns consumers and healthcare professionals of Hand Sanitizers The test method for methanol described in the USP Alcohol Monograph can also be used |
What is the ISO standard for hand sanitizer?
ISO 9001 Certification Standard and GMP Certification for hand sanitizer.What is the specification of sanitizer?
General Description: Alcohol-based hand sanitizer, with alcohol content of 60-80%, and capacity of 75-100ml. Hand antisepsis for personal use.What is the specification for alcohol hand sanitizer?
Ethyl alcohol is the active ingredient in most hand sanitizers. A concentration of 60 % to 95 % alcohol in hand sanitizing product is recommended.- Must not exceed 95% alcohol content by volume. (The Centers for Disease Control and Prevention recommends that ABHS contain at least 60% alcohol.) 1.2 liters (41 ounces, 0.32 gal) for dispensers in rooms, corridors, and areas open to corridors.
|
Hand Sanitizer - USPorg
Excerpted USP-NF and FCC Standards: A Hand Sanitizer Resource A collection of standards provided as a resource to assist with the challenges posed by |
|
Compounding Alcohol-Based Hand Sanitizer During - USPorg
25 mar 2020 · 1 If soap and water are not available, using a hand sanitizer with a final concentration of at least 60 ethanol or 70 isopropyl alcohol |
|
USP Alcohol-Based Hand Sanitizer Infographic
Download and follow the Compounding Alcohol-Based Hand Sanitizer During COVID-19 Pandemic recommendations at usp org/compounding Packaging and |
|
USP FAQs Regarding Alcohol-Based Hand Sanitizer
28 avr 2020 · CDC recommends a hand sanitizer formulation with a final concentration of greater than 70 isopropyl alcohol to inactivate viruses that are |
|
FAQ Technical Grade Ethanol Hand Sanitizer - Alberta Health
25 jui 2020 · in hand sanitizers and other disinfectant products Question 2: What is “USP”- or “ pharmaceutical-grade” ethanol? “USP” stands for United |
|
Hand Sanitizer Analysis Using the Agilent 8860 GC Configure with a
29 jui 2020 · hand sanitizer The methods for alcohol determination, such as USP 1 and ASTM D3695,2 recommend a mid-polar or polar column |



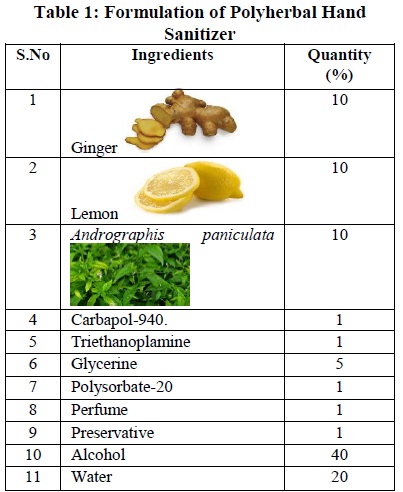






![Brochure] Fresh GEL Hand Sanitizer (70% Ethanol / Aloe Vera Extract) Brochure] Fresh GEL Hand Sanitizer (70% Ethanol / Aloe Vera Extract)](https://d3i71xaburhd42.cloudfront.net/1186afb5314d230d4bf0689e946e158a425e6595/2-Table1-1.png)
![PDF] Formulation of handsanitizer with antibacterials substance PDF] Formulation of handsanitizer with antibacterials substance](https://www.cdc.gov/handwashing/images/hand-sanitizer-factsheet.jpg)
![Read [PDF] Homemade Hand Sanitizer A Practical Guide to Make Your Ow Read [PDF] Homemade Hand Sanitizer A Practical Guide to Make Your Ow](https://i1.rgstatic.net/publication/314245369_Formulation_and_Evaluation_of_the_Effectiveness_of_a_Novel_Hand_Sanitizer_using_Pleurotus_ostreatus_Oyster_Mushroom_Extract/links/58bdaf8192851c471d5bf719/largepreview.png)


![Read [PDF] Homemade Hand Sanitizer A Practical Guide to Make Your Ow Read [PDF] Homemade Hand Sanitizer A Practical Guide to Make Your Ow](https://dtkp6g0samjql.cloudfront.net/uploads/photo/file/12598827/il_fullxfull.991155239_la57.jpg)