Insert BLOD 2016-2017
|
BEHAVIORAL SCIENCE AROUND THE WORLD Profiles of 10
and Development Unit (eMBeD) spends a lot of time thinking about how resources/2016-2017-annual-report.html. ... of youth donors for blood donation. |
|
XOLAIR Prescribing Information
XOLAIR is indicated for add-on maintenance treatment of nasal polyps in adult patients 18 reduced blood pressure with or without associated symptoms ... |
|
Mitigating the Risk of Bacterial Contamination of Platelets - Recent
Sep 30 2019 Bacterial Risk Control Strategies for Blood Collection Establishments and ... Platelet PGD Test package Insert |
|
NIH Triennial Report FY 2016-2018
NIH Research Activities in FY 2016 2017 |
|
No. 329-Canadian Contraception Consensus Part 4 of 4 Chapter 9
A blood pressure measurement is the only examination and/or investigation that is required prior to contraceptive ring has been inserted (II-2). |
|
Towards-Race-Equity-in-Education-April-2017.pdf
Apr 1 2017 students and to put into context the many issues that are occurring ... http://schottfoundation.org/blog/2016/10/24/school-prison-pipeline- ... |
|
DEPARTMENT OF PATHOLOGY AND LABORATORY MEDICINE
expanded the clinical laboratory services to include a Complete Blood Count In 2016-2017 graduate students from the program contributed authorship to a ... |
|
Digital Economy Report 2019
Mar 10 2019 Details and percentages in tables do not necessarily add up to the totals because of ... 2012 to 5.7 per cent in 2016–2017 (ABS |
|
Untitled
Add Databases / Schemas Open Table. Navicat loads all your BLOB fields (images) while opening the table. ... Insert BLOB data as hexadecimal format. |
|
OPINIONS on the safety of breast implants in relation to anaplastic
Mar 26 2021 implant sales data |
|
Package Insert - Prevnar 13 - Food and Drug Administration
Package Insert - Prevnar 13 1 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PREVNAR 13 safely and effectively See full prescribing |
|
Reference ID: 4133376 - Food and Drug Administration
blood creatine phosphokinase increased 7 DRUG INTERACTIONS Clinically relevant pharmacokinetic interactions are not expected between vismodegib and a substrate inducer or inhibitor of |
|
Reference ID: 4083804 - Food and Drug Administration
2 weeks If blood counts remain low for greater than 2 weeks upon recovery reduce dose by 100 mg and resume treatment If cytopenia recurs reduce dose by an additional 100 mg upon recovery and resume treatment Doses less than 300 mg/day have not been evaluated a Absolute Neutrophil Count 2 5 Concomitant Use With CYP3A Inhibitors |
|
AZOR (amlodipine and olmesartan medoxomil) - Food and Drug
should be individualized and should be shaped by considerations such as baseline blood pressure the target goal and the incremental likelihood of achieving goal with a combination compared to |
|
Reference ID: 3955374 - Food and Drug Administration
Administer Xolair 75 to 375 mg by subcutaneous injection every 2 or 4 weeks Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL) measured before the start of treatment and by |
|
Reference ID: 3896348
Undergoing peripheral blood progenitor cell mobilization and collection (? 5 incidence) are bone pain pyrexia and headache (6 1) With severe chronic neutropenia (SCN) (? 5 difference in |
|
HIGHLIGHTS OF PRESCRIBING INFORMATION History of severe
Revised: 3/2017 FULL PRESCRIBING INFORMATION: CONTENTS * 1 INDICATIONS AND USAGE 1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION 2 1 Preparation for Administration 2 2 Recommended Dose |
|
Package Insert - Flublok Quadrivalent - Immunization Info
Package Insert BLA STN 125285 Flublok Quadrivalent PI (V1 3) 2016 Page 2 of 18 FULL PRESCRIBING INFORMATION: CONTENTS* 1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION 2 1 Dosage 2 2 Administration 3 DOSAGE FORMS AND STRENGTHS 4 CONTRAINDICATIONS 5 WARNINGS AND PRECAUTIONS 5 1 Managing Allergic Reactions |
|
HIGHLIGHTS OF PRESCRIBING INFORMATION CONTRAINDICATIONS
2016-2017 Formula Initial US Approval: 2009 -----INDICATIONS AND USAGE----- Fluzone High-Dose is a vaccine indicated for active immunization for the prevention of influenza disease caused by influenza A subtype viruses and type B virus contained in the vaccine (1) |
|
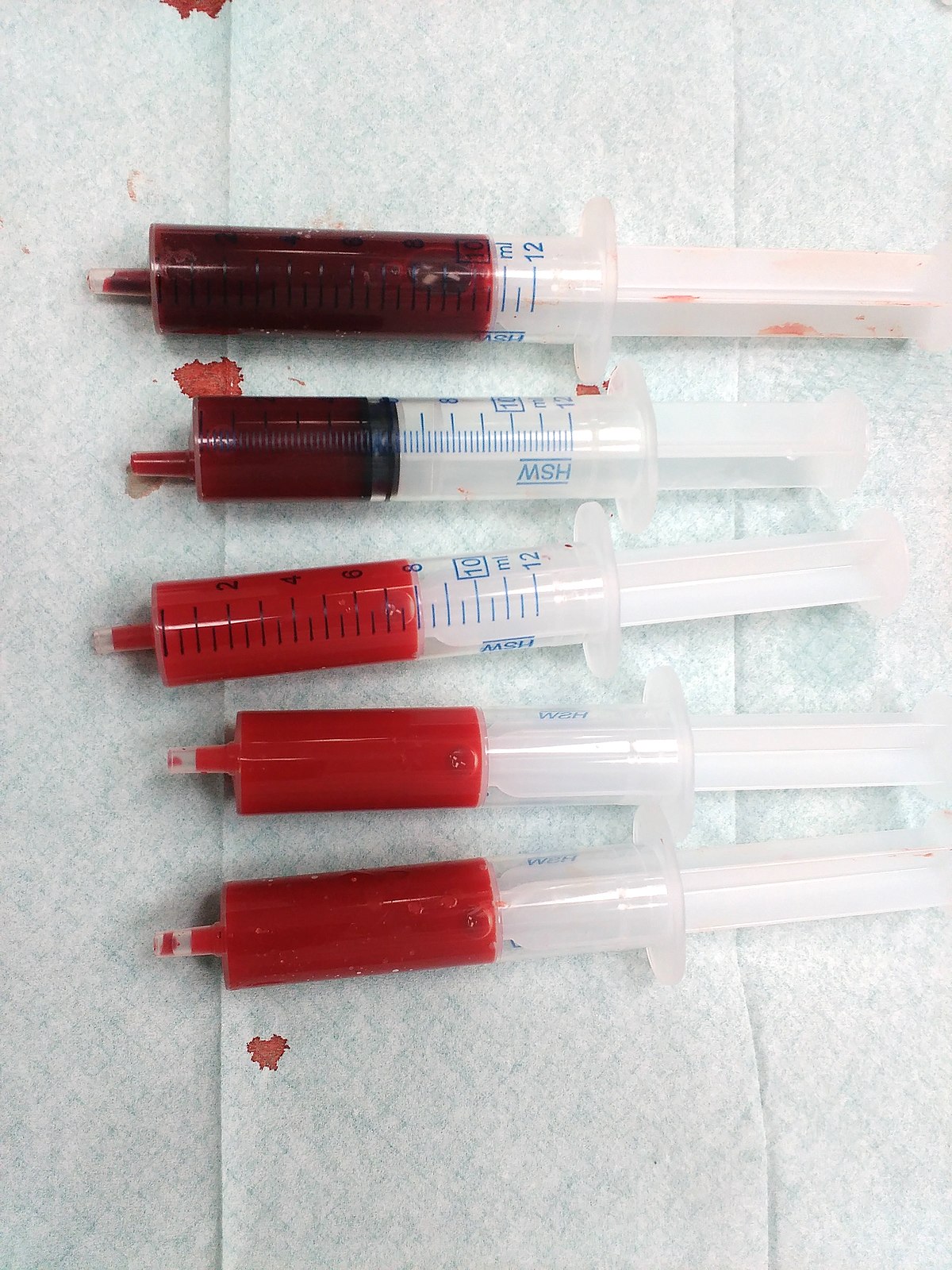
Blood Collection and Handling Training Guide - QuantiFERON
As 1 ml tubes draw blood relatively slowly keep the tube on the needle for 2 to 3 seconds once the tube appears to have completed filling to ensure that the correct volume is drawn The black mark on the side of the tubes indicates the 1 ml fill volume QFT blood collection tubes have been validated for volumes ranging from 0 8 ml to 1 2 ml |
|
RYDAPT HIGHLIGHTS OF PRESCRIBING INFORMATION RYDAPT safely
Initial U S Approval: 2017-----INDICATIONS AND USAGE-----RYDAPT is a kinase inhibitor indicated for the treatment of adult patients with: Newly diagnosed acute myeloid leukemia (AML) that is FLT3 mutation-positive as detected by an FDA-approved test in combination with |
|
Searches related to insert blod 2016 2017 filetype:pdf
%20blood%20&%20biologics/published/Package-Insert---Flucelvax.pdf |
|
Insert BLOD 2016-2017
LA FARANDOLE DU 24 AU 28 OCTOBRE 2016 LA FARANDOLE DU 10 AU 14 AVRI 2017 « Les milles et une nuit » GRIMPEZ SUR LE TAPIS VOLANT ET |
|
CENTRAL VENOUS CATHETER - Regina QuAppelle Health Region
NOTE: Physician will insert CVC and assess for adequate blood return been applied over insertion site 17 Request chest x-ray to confirm position of catheter |
|
HEALTH SERVICES CODE T3 NURSING PROCEDURE TITLE
Approved: June 7, 2017 Page 1 of 22 NURSING ALERT: • Insertion and removal is the responsibility of a physician Notify MRP if signs of malposition are present: Inability to withdraw blood, a “gurgling” sound heard when flushing |
|
Guidelines for the rational use of blood and blood products
G6PD Deficiency Patients 16 Consent Form for Blood Transfusion 105 17 Prescribing Insert central venous pressure line if available, and monitor CVP |
|
Blood Cultures - Elsevier
antibiotic effect 17 Typically, two sets of blood cultures are ordered, each with one cases, a hollow-bore needle is inserted into the lumen of a vein to obtain the |
|
HEPARIN SODIUM, for intravenous use - FDA
Anticoagulant use in blood transfusions, extracorporeal circulation, See 17 for PATIENT COUNSELING INFORMATION the spike is fully inserted 5 |
|
Covid-19 rapid test by combining a random forest based - medRxiv
12 jui 2020 · In a second exam, 17-18 days after the first symptoms, 110 to insert the results of blood tests that are commonly requested for patients |
|
BG education document - IFCC
This document serves as a general guide for Blood Gas Analysis (BGA) testing that can be useful for laboratory 17 APPENDIX A: Additional resources on blood gas pre-analytics Always enter a patient ID into the analyzer before analysis |
|
Nova Stat Strip Blood Glucose Meter Re-Certification 2018
Dated: December 2017 Reference: Policy LAB 23-A; Point of Care Nova Stat Strip Glucose Meter Collect blood in a grey top tube or add on a glucose test |