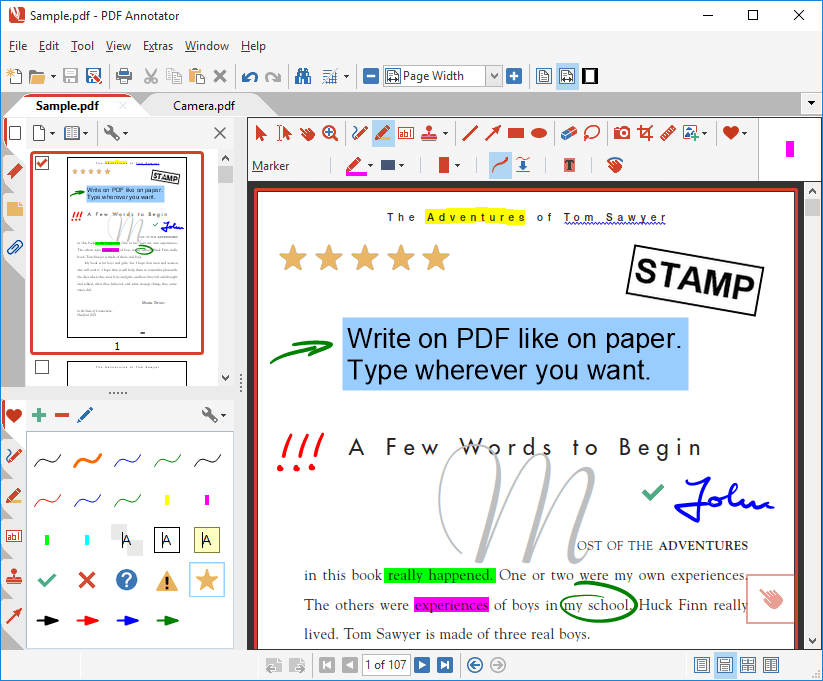
cm = c x m
|
CONVERSION TABLE FOR METRIC UNITS OF LENGTH
CONVERSION TABLE FOR METRIC UNITS OF LENGTH centimeter (cm) = 0 01 m millimeter (mm) = 0 001 m millimeter (mm) = 0 1 cm micrometer (μm) = 0 001 mm nanometer (nm) = 0 001 μm Perform the following conversions: meter = 100 centimeters meter = 1000 millimeters |
|
Conversions in the Metric System
PART A – Ratio & Proportion Method Example 1: Convert 1 74 m into cm To convert meters to centimeters we need to first identify the larger unit Looking at the Metric System chart we notice that a centimeter is 100 times smaller than a meter Thus 1 m = 100 cm Knowing this we can set up a proportion to convert 1 74 m into cm Remember! |
|
METRIC MEASURES METRIC EQUIVALENTS
METRIC MEASURES LINEAR MEASURE 10 millimeters 1 centimeter 10 centimeters 1 decimeter 10 decimeters 1 meter |
|
Measuring units Worksheet
Title: Measuring units Worksheet Author: Maria Miller Subject: measuring units worksheet Keywords: measurement measuring units worksheet Created Date |
What is the full form of cm in physics?
1. Full Form of CM 2. Importance of center of mass 3. Center of gravity In physics, CM stands for the center of mass distribution in space (sometimes called the equilibrium point) and is the distinct point where the weighted relative positions of the distributed masses sum to zero.
How do you convert m to mm?
Converting m to mm requires multiplying by 1,000, because there are 1,000 mm in one m. It follows from these two facts that converting from km to mm requires multiplying by 1,000*1,000=1,000,000. Have a blessed, wonderful day!
How do you convert meters to centimeters?
Since the metric system of measurement is based on powers of ten (10) converting between units is a snap! To convert meters to centimeters we need to first identify the larger unit. Looking at the Metric System chart we notice that a centimeter is 100 times smaller than a meter.
Is a centimeter 100 times smaller than a meter?
Looking at the Metric System chart we notice that a centimeter is 100 times smaller than a meter. Remember! A proportion is a comparison of two equal ratios in which order matters. On the Left Hand Side (L.H.S.) of the proportion, list the ratio we know. On the Right Hand Side (R.H.S), list the ratio we are trying to find out.
|
Ch1 LA MOLE
a) L'éthanol a une masse volumique ?= 0.78 g.cm-3 et une masse molaire de 46 g.mol-1. Donner la définition de la masse Cm = CxM =0.50x254 = 127 g.L-1. |
|
Application Notes for CXM 5.3 with Avaya Aura® Communication
4 févr. 2019 CXM starts the call recording by using the Single Step ... Restart the CXM Avaya CM Recorder and the CXM Voice Interface DMCC services. |
|
Le cristal de Condy
Cm=CxM=0.436g/L donc pour la fiole de 2L le sachet devait contenir 087g de cristaux. A=0 |
|
Rek.Nro Vanha kauppanimi Vahvuus LM Rekisteröinnin haltija
C6C12 |
|
Rek.Nro Vanha kauppanimi H451FI ACONITUM NAPELLUS
H594FI AESCULUS HIPPOCASTANUM HELIOS C 6-200CM |
|
Norms for Clinical Use of CXM a Real-Time Marker of Height Velocity
HV (cm/yr). 97th. 90th. 50th. 10th. 3rd. Figure 2. Collagen X biomarker (CXM) values by age for females. CXM percentile reference curves from the third to |
|
TP Bouillie Bordelaise
Avec le laser rouge car Lambda la plus proche en fonction du max d'absorbance du spectre. C=0152mol.L-1 et Cm=CxM=0 |
|
CONCENTRATIONS MOLAIRE ET MASSIQUE V nC = M mn = MV
Cm×= Relation entre concentration molaire et pourcentage massique. Exemple : sur l'étiquette d'un litre de solution d'acide chlorhydrique il est indiqué :. |
|
Ch 11 lénergie interne
La capacité thermique représente la capacité du système à absorber l'énergie thermique. C = c x m avec c la capacité thermique massique en J.K-1.kg-1. C = Cm |
|
Cefuroxime Imprinted p(HEMATrp) Cryogels: Preparation
28 févr. 2022 Finally the antimicrobial potentials of CXM-p(HEMATrp) cryogels were evaluated according to the growth inhibition zones measured in cm. The an-. |
|
CONCENTRATIONS MOLAIRE ET MASSIQUE V nC = M mn
elation entre concentration molaire et pourcentage massique Exemple : sur l'étiquette d'un litre |
|
Ch1 LA MOLE
|
|
TP dosage agneauodt - Sgen-CFDT Midi-Pyrénées
7,55 10-2x 53,5=4,04g L-1 ou Cm=7,65 10-2x 53,5=4,09g L-1 L' éleveur a donné 4,04 g (ou 4 |
|
Chimie analytique: base et CM1 - 2ATP
entration massique: ➢Relation entre concentration molaire et massique : ➢Cm=CxM ➢ |
|
Indust_Proc_Corpdf
BACPDF |
|
Correction-livre-phys-boisson-et-combustionpdf
M 4 L'ion sodium a la même masse molaire que l'atome de sodium MN = 23,0 g mol-1 Cm |
|
Sujet_bp_coif_2015pdf - SBSSA - Versailles
concentration molaire C de cette solution Arrondir le résultat au dixième On rappelle que Cm=CxM |