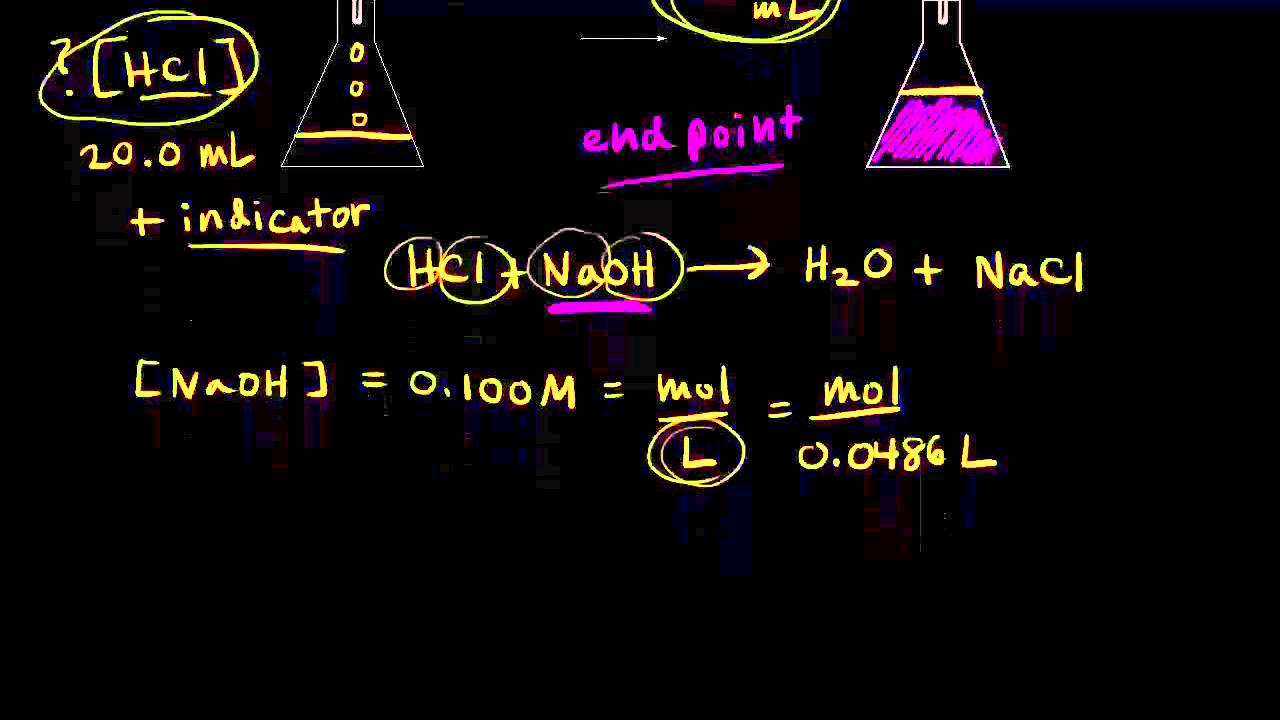
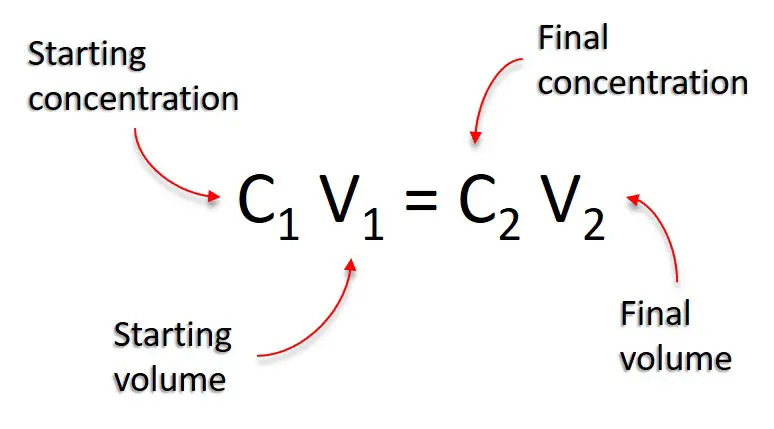
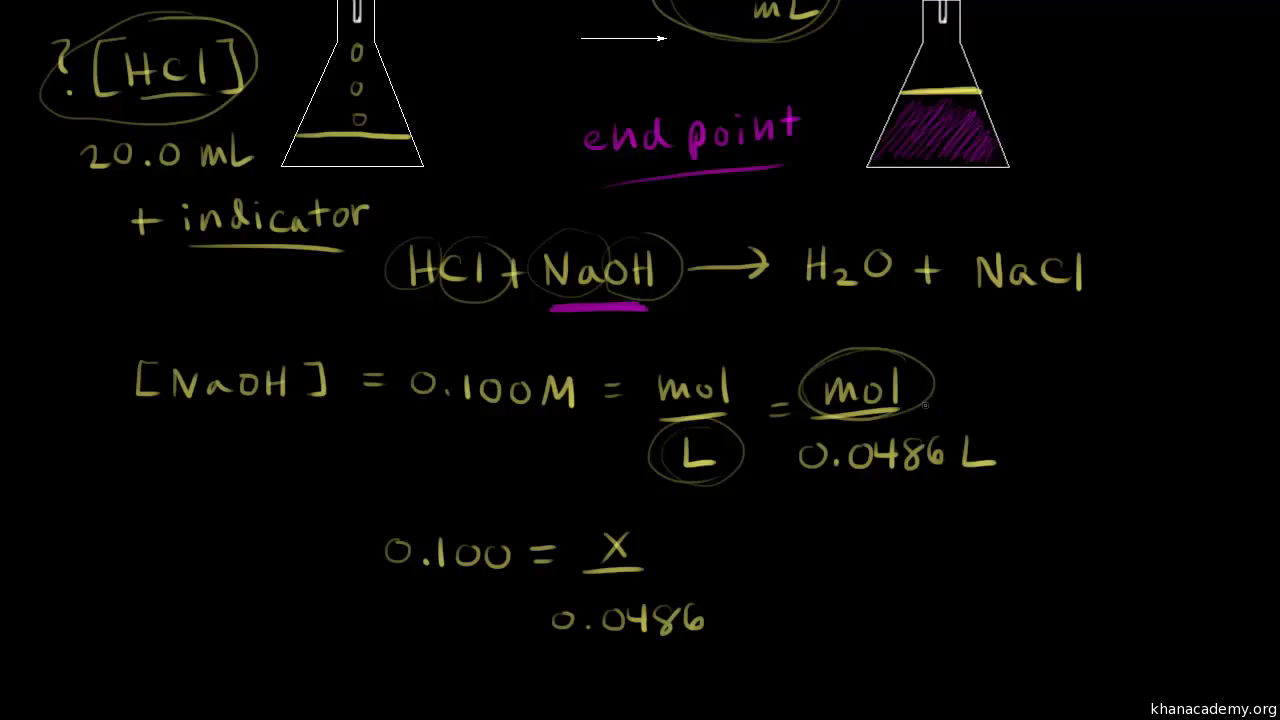c1v1=c2v2 khan academy
|
AMERICAN
Use the American Elements solution dilution calculator to determine the volume of a stock concentrate to add to achieve a specific volume and concentration. |
|
Alloprof
La dilution est utilisée pour diminuer la concentration d'une solution en y ajoutant du solvant sans changer la quantité de soluté (C1V1 = C2V2). |
|
The C1V1 = C2V2 Equation Explained
The C1V1 = C2V2 formula is a lifesaver for researchers who are wanting to do dilutions. Here I will explain what the equation means and how you can use it. |
|
Tocris Bioscience
The Tocris dilution calculator is a useful tool which allows you to calculate how to dilute a stock solution of known concentration. |
|
AT A GLANCE/ PHARMACY CALCULATIONS C V = C V
C1V1=C2V2 is used to calculate an unknown quantity where two solutions/mixtures are proportional … C1V1 = Concentration/amount (start) and Volume (start). |
|
PAAV-Ef1a-DIO C1V1 (t/t)-TS-mCherry - Addgene
Plasmid pAAV-Ef1a-DIO C1V1 (t/t)-TS-mCherry from Dr. Karl Deisseroth's lab contains the insert ChR1-VChR1 Chimera and is published in Nature. |
|
Surviving C1V1 = C2V2
C2 = final concentration of the solution after dilution. V1 = volume about to be diluted. V2 = fianl volume after dilution. |
|
PAAV-CaMKIIa-C1V1 (t/t)-TS-EYFP - Addgene
Plasmid pAAV-CaMKIIa-C1V1 (t/t)-TS-EYFP from Dr. Karl Deisseroth's lab contains the insert ChR1-VChR1 Chimera and is published in Nature. |
|
PAAV-CaMKIIa-C1V1 (t/t)-TS-mCherry - Addgene
Plasmid pAAV-CaMKIIa-C1V1 (t/t)-TS-mCherry from Dr. Karl Deisseroth's lab contains the insert ChR1-VChR1 Chimera and is published in Nature. |
|
Solutions - Who came up with C1V1 = C2V2? - Chemistry Stack
Sept 6 2020 tl;dr It's so simple it was likely never published as a "research result" |
|
C1V1=C2V2 - Mathcentre
C1V1=C2V2 is used to calculate an unknown quantity where two solutions/ mixtures are proportional C1V1 = Concentration/amount (start) and Volume ( start) |
|
Lab 3 - B75 Lab manual_ss_pt1
The C1V1 = C2V2 equation may be used with any concentration units (i e mass/ volume, , or molar) as long as the units are the same on each side of the |