carboxylate salt boiling point
|
Chapter 5 Carboxylic Acids and Esters
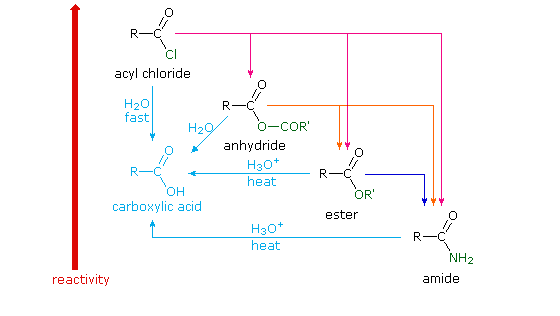
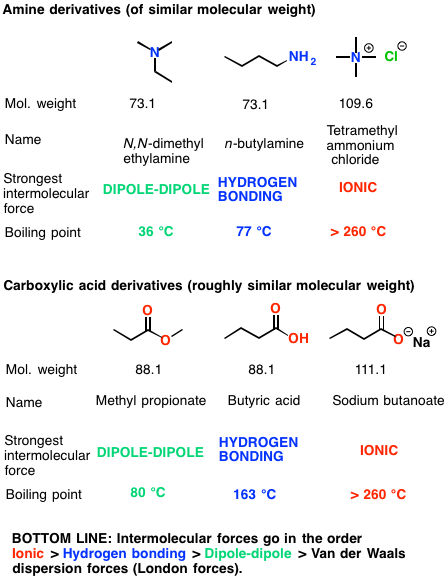
The boiling points of carboxylic acids compared to 1° alcohols aldehydes Reaction of a carboxylic acid with a base to form a carboxylate salt and water |
Are carboxylate salts soluble in water?
3.10.
Indeed, when turned into its carboxylate salt form, this alkoxyamine is fully water-soluble and can readily initiate polymerization in the aqueous phase.What is the boiling point of a carboxylic acid?
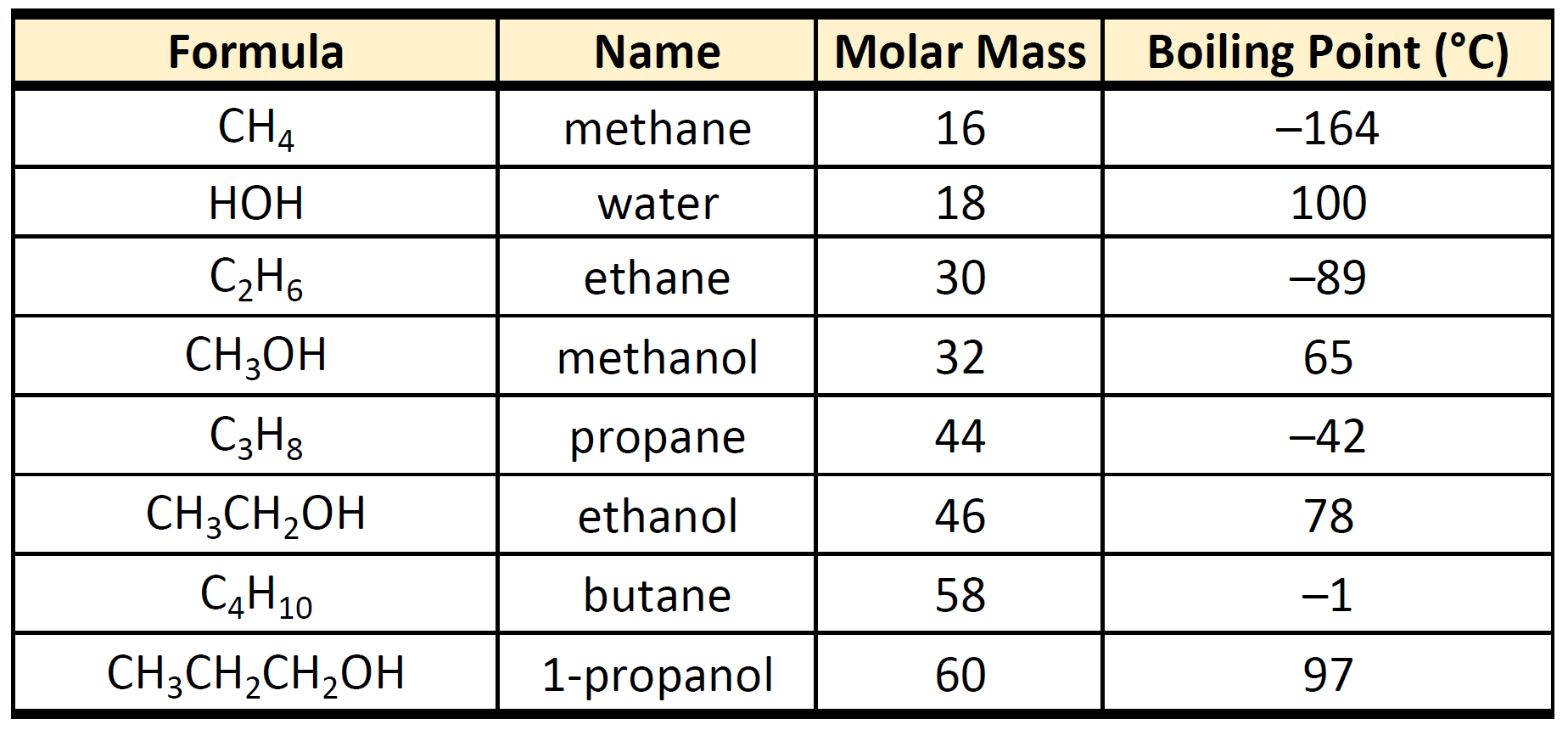
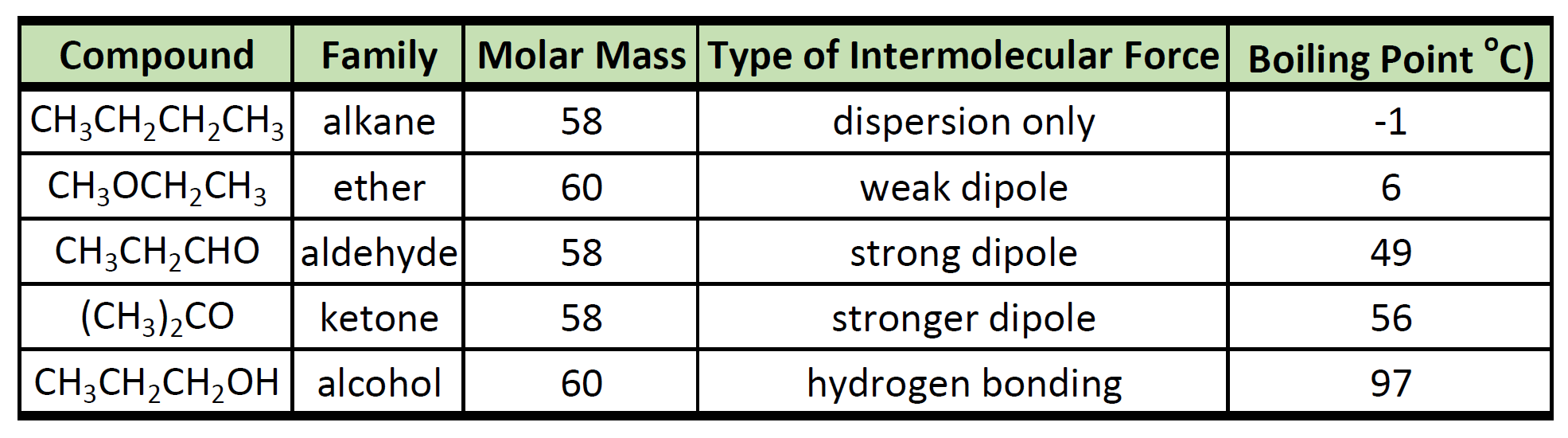
Even the simplest carboxylic acid, formic acid, boils at 101 °C (214 °F), which is considerably higher than the boiling point of ethanol (ethyl alcohol), C2H5OH, which boils at 78.5 °C (173 °F), although the two have nearly identical molecular weights.
In a pure carboxylic acid, hydrogen bonding can occur between two molecules of acid to produce a dimer.
This immediately doubles the size of the molecule and so increases the van der Waals dispersion forces between one of these dimers and its neighbors - resulting in a high boiling point.
|
Chapter 5 Carboxylic Acids and Esters
becomes greater the boiling point increases and the Boiling Points of Various Functional Groups ... Carboxylate salts are named by naming the metal. |
|
Chapter 6 Amines and Amides
1° and 2° amines have lower boiling points than Salts of amines are named by changing “amine” to ... of the corresponding carboxylic acid to -amide. If. |
|
Chapter 5 Carboxylic Acids and Esters
becomes greater the boiling point increases and the Boiling Points of Various Functional Groups ... Carboxylate salts are named by naming the metal. |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
The boiling points of aldehydes and ketones are higher than hydrocarbons and ethers of alcohol while another is oxidised to carboxylic acid salt. |
|
Carboxylic Acids and Their Derivatives (pp. 971-978)
For example compare the boiling points of acetic acid and ethanol. Carboxylate salts are ionic and are therefore more soluble in water than their ... |
|
DIRECT CONVERSION OF CARBOXYLATE SALTS TO
This research focuses on converting fermentation broth carboxylate salts into 2-1 Apparatus for determine boiling point elevation and ammonium. |
|
DPD Free Chlorine Reagent
27 jun 2016 Chemical Family. Mixture. Chemical Name. CAS No. Weight-%. HMRIC #. Carboxylate Salt. -. 60.479. -. |
|
The Dissociation Constants of Acids in Salt Solutions. VI
for use. Table II gives the ratios of the dissociation con-. Table I. Purity of the. Carbocyclic Carboxylic Acids . Boiling point. 20. Analysis by. |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
The boiling points of aldehydes and ketones are higher than hydrocarbons and ethers of alcohol while another is oxidised to carboxylic acid salt. |
|
LA41Carboxylic Acids - Newcastle University
The boiling point and solubility of carboxylic acids is related to their ability to form hydrogen bonds This anion then can form a salt ionically bonding with a |
|
DIRECT CONVERSION OF CARBOXYLATE SALTS TO - CORE
Through the physical solubility of the solute in the extractant phase, the carboxylic acids form acid-amine complexes with high-molecular-weight tertiary amine |
|
Carboxylic Acids, Amines, and Amides
carboxylic acid, amine, or amide molecules to one another, and how these forces affect boiling points and melting points 3 Identify carboxylic acid salt |
|
Chapter 13 Carboxylic Acids, Esters, Amines, and Amides
A carboxylic acid contains a carboxyl group, which have higher boiling points than alcohols, ketones, and produces the salt of the carboxylic acid and an |
|
Nomenclature of Carboxylic Acids - Angelo State University
becomes greater, the boiling point increases and the Boiling Points of Various Functional Groups Carboxylate salts are named by naming the metal |
|
Carboxylic Acids
animal urine, and calcium oxalate (the calcium salt of oxalic acid) is a major The higher boiling points of carboxylic acids result from their polarity and from |
|
CARBOXYLIC ACIDS AND THEIR DERIVATIVES 1 Carboxylic
carboxylic acids, as well as their derivatives, esters, amides, and anhydrides, A Review of Melting Points of S-l-Naphthylmethylthiuronium Salts Acid Formic |
|
Physical properties of carboxylic acids - TSFX
are marketed as carboxylic acid salts (sodium or potassium salts) A brief review of boiling point trends • Boiling points are determined by attractions between |
|
FACTFILE: GCE CHEMISTRY - CCEA
4 8 1 recall the structural formulae of carboxylic acids 4 8 2 explain the boiling points and solubility of 4 8 5 recall that carboxylic acids form salts with |














![12 Aldehydes Ketones and Carboxylic acids - [PDF Document] 12 Aldehydes Ketones and Carboxylic acids - [PDF Document]](https://upload.wikimedia.org/wikipedia/commons/thumb/f/fb/Propiolic_acid_Structural_Formula_V.2.svg/1200px-Propiolic_acid_Structural_Formula_V.2.svg.png)