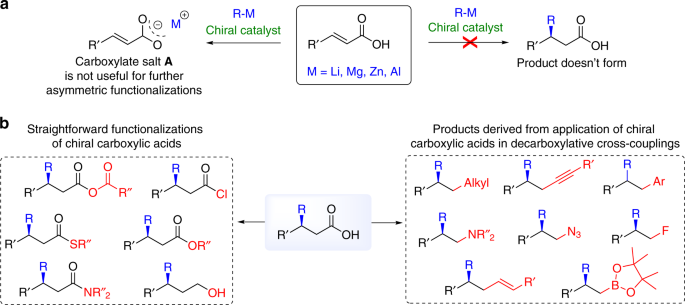
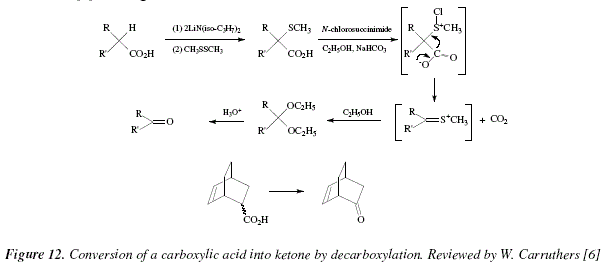
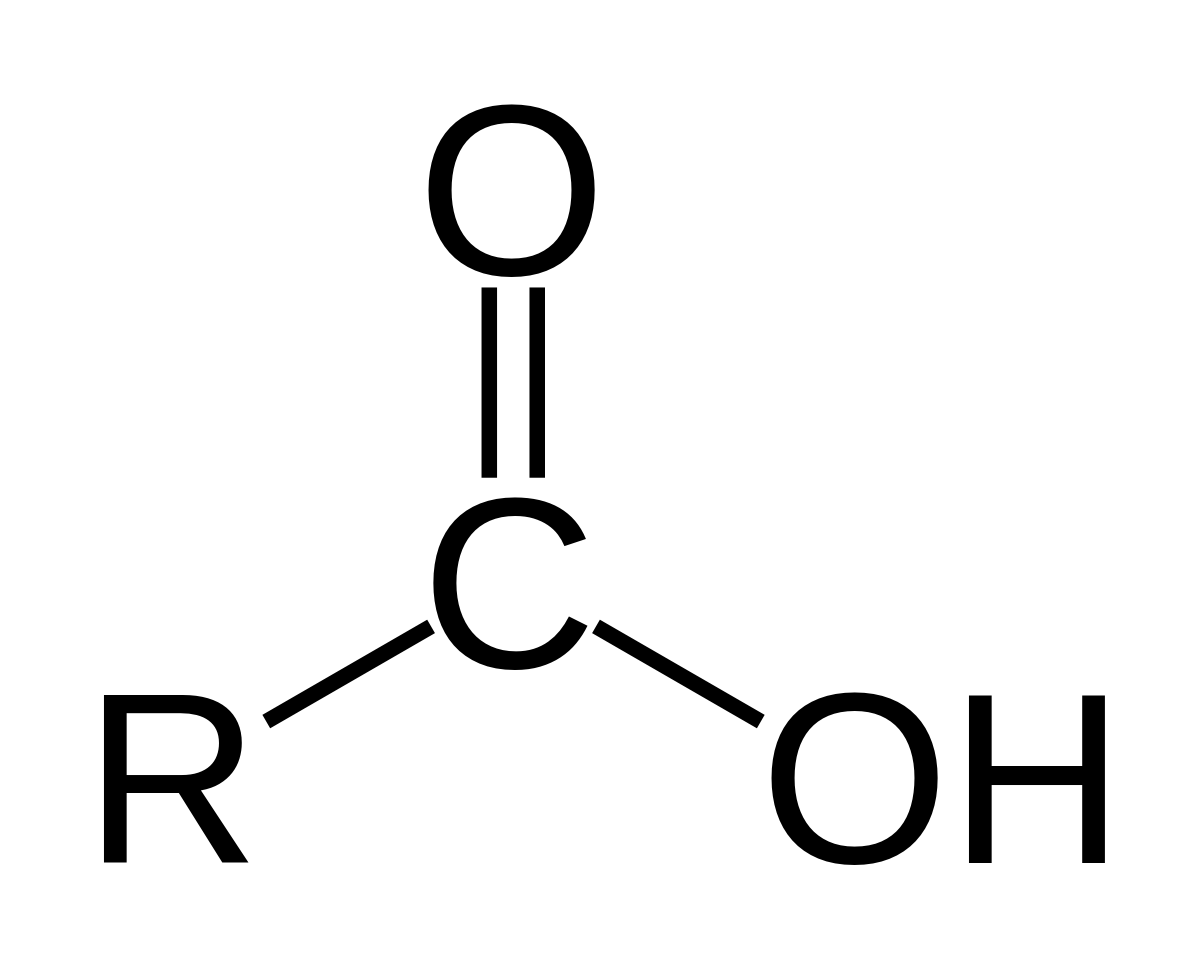
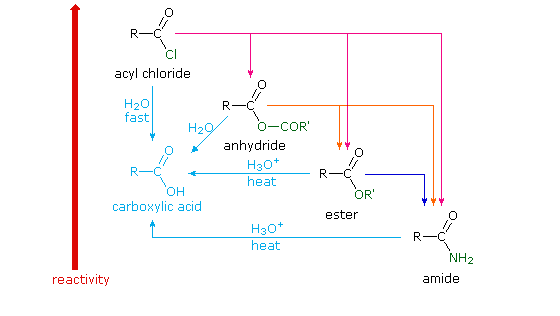
carboxylate salt examples
|
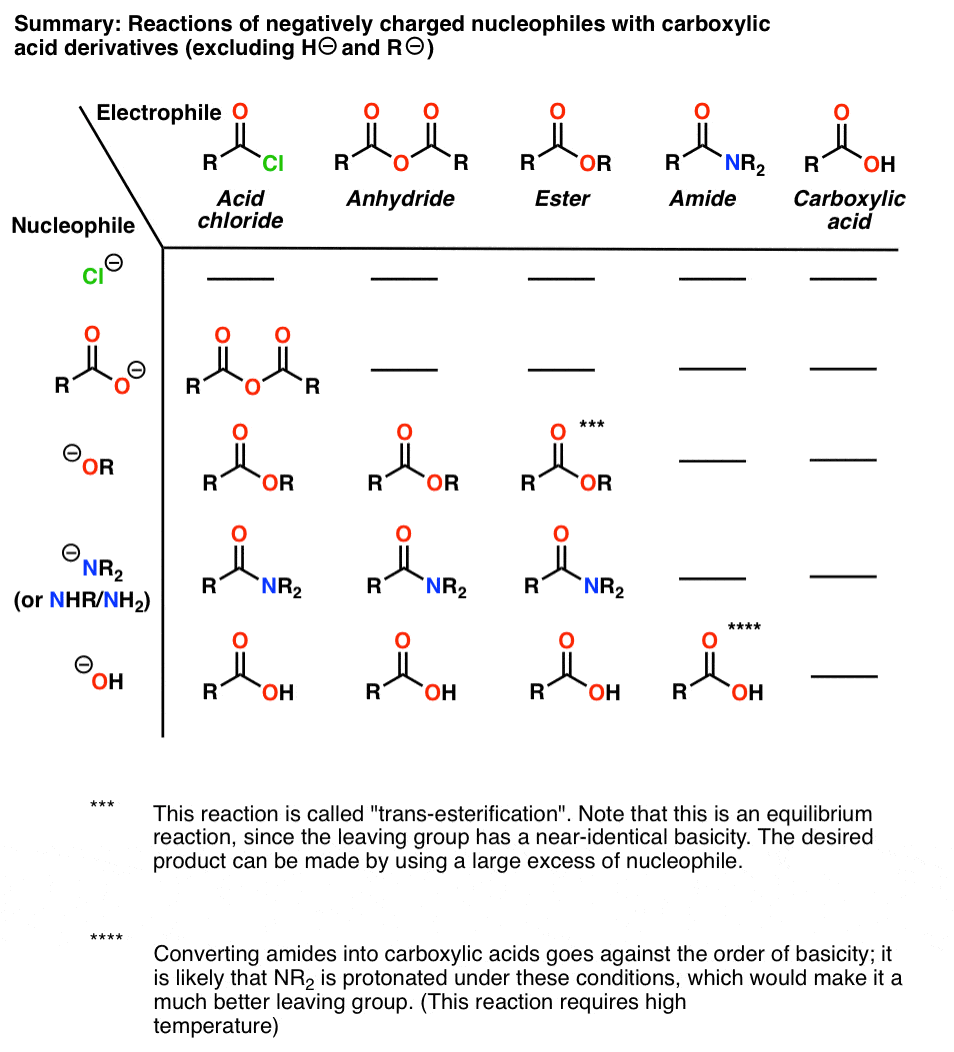
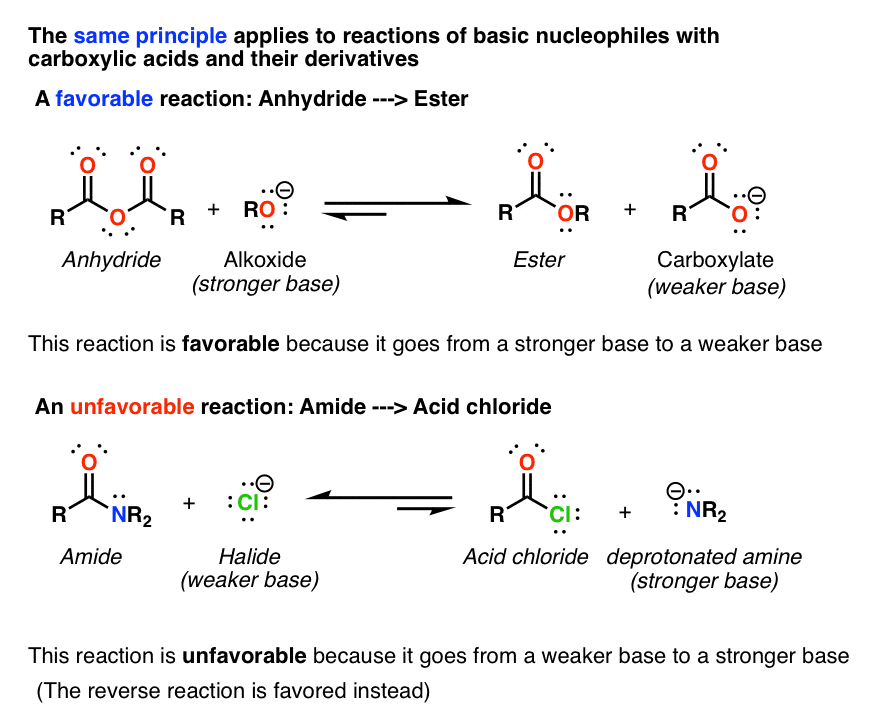
Product Class 3: Carboxylic Acid Salts
examples of carboxylic acid salts known for their broad spectrum resistance to β-lactamases [1] Scheme 1 β-Lactam Antibiotics as Carboxylic Acid Salts[1] 1 |
How do you name carboxylate salts?
Naming carboxylates
Salts of carboxylic acids are named by writing the name of the cation followed by the name of the acid with the –ic acid ending replaced by an –ate ending.
This is true for both the IUPAC and Common nomenclature systems.3.
2) Sodium carboxylate salts
As reported in reference 3, sodium carboxylate salts of alkenyl succinic acid corrosion inhibitors can form diesel injector internal deposits.
It is a natural extension to expect that other carboxylic acid-functional compounds also have the potential to form salt deposits.
What forms carboxylate salt?
Carboxylic acids have the capability to form anions and salts (carboxylates).
The carboxyl group can be connected to a simple aliphatic or aromatic radical R as well as to heterocycles.
Two types of names are used for organic acids.
What is an example of a carboxylate salt?
Carboxylic acid salt is the salt formed when a carboxylic acid reacts with a base.
For example, sodium acetate is a salt composed of sodium cations and acetate anions, but ethyl acetate is a covalently bonded ester.
|
Chapter 5 Carboxylic Acids and Esters
Examples: Carboxylate Salts. • Draw the structure of pyruvate the form of pyruvic acid which is found as an intermediate in energy. |
|
Chapter 5 Carboxylic Acids and Esters
Examples: Naming Carboxylic Acids. • Name the following compounds: Examples: Carboxylate Salts ... Carboxylate salts are ionic compounds and are. |
|
Product Class 3: Carboxylic Acid Salts
Salts of carboxylic acids are frequently encountered in organic synthesis and alosporins 2 (Scheme 1) are prominent examples of carboxylic acid salts ... |
|
Field desorption mass spectrometry of carboxylate salts
carboxylate salts and the effect on fdms of added complexing agents especially fructose. Experimental. The instrumental parameters and methods of sample |
|
Direct reversible decarboxylation from stable organic acids in
Jun 18 2020 cent to the carboxylate was tolerated |
|
Fuel Quality and Diesel Injector Deposits
Sep 10 2012 The proposed mechanism of metal carboxylate salt IID ... few examples are available for analysis |
|
Promoting carboxylate salts in the ESI source to simplify positive
Dec 31 2020 Each PU sample (a few mg) was dissolved in THF and further diluted in methanolic solutions of different salts (see text). 2.2. Mass spectrometry. |
|
Salt Hydrates: New Reversible Absorbents for Carbon Dioxide
amounts of CO2.1011 Displaying this property are hydrates of various salts which contain fluoride or carboxylate ions. Two examples are. |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
They add fragrance and flavour to nature for example |
|
Chapter 6 Amines and Amides
Examples: Nomenclature of Amines Salts of amines are named by changing “amine” to ... of the corresponding carboxylic acid to -amide. If. |
|
Nomenclature of Carboxylic Acids - Angelo State University
O Benzoic acid 5 Examples: Naming Carboxylic Acids Examples: Carboxylate Salts • Draw the Carboxylate salts are ionic compounds, and are typically |
|
Prelab 6: Carboxylic Acids
A specific example of the reaction of a carboxylic acid and water is the reaction A carboxylic acid salt formed from a carboxylate anion and a Na+ or K+ |
|
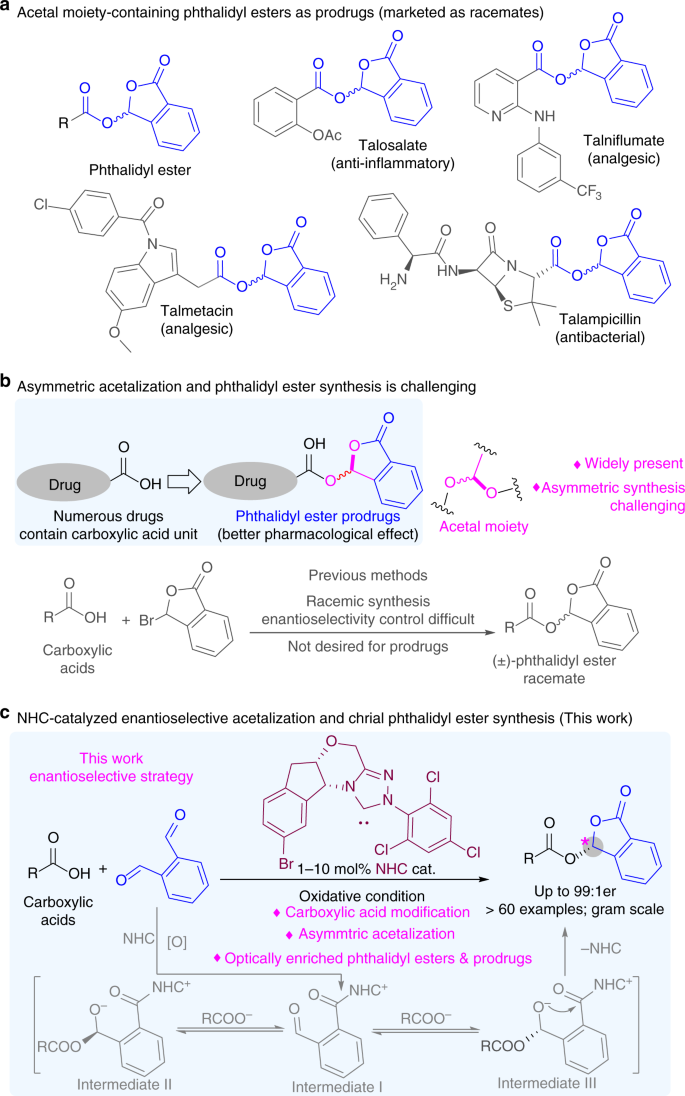
Amidation Reactions from the Direct Coupling of Metal Carboxylate
19 déc 2013 · protected carboxylic acid functionality in peptide couplings 6 For example, alkaline earth metal carboxylate salts of α-amino acids have been |
|
Grindstone neutralization reactions for the preparation - MedCrave
15 mar 2019 · Salts of carboxylic acids are familiar and play an important role in so many fields, for example pharmaceutical,1 food processing,2 textile |
|
CARBOXYLIC ACIDS AND THEIR DERIVATIVES 1 Carboxylic
I Salts (a) of heavy metals (for example, silver salts), (b) of amines methylthiuronium chloride with the sodium salt of carboxylic acid in metha- nol The salts |
|
6 Synthesis of Esters
nium carboxylate or the lack of tight ion pairing in this salt might account for the example, if an equimolar mixture of a carboxylic acid, an alkyl halide and a |
|
CHAPTER 4 CARBOXYLIC ACIDS
4 1 NAMING CARBOXYLIC ACIDS AND CARBOXYLATE IONS To generate the systematic name In the examples shown below the positive ion is a Na + ion |
|
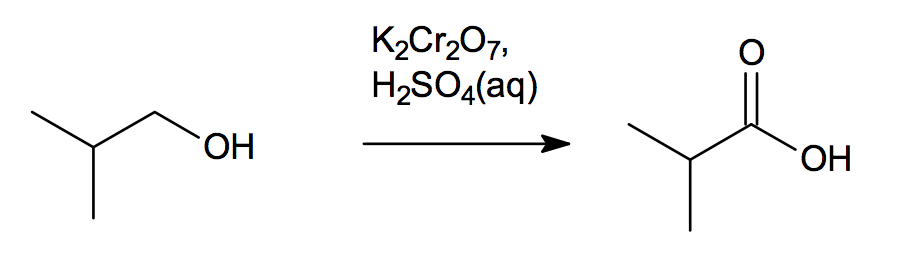
Carboxylic Acids
In the following examples, the common name of each acid is given A consequence of the water solubility of carboxylic acid salts is that we can convert |