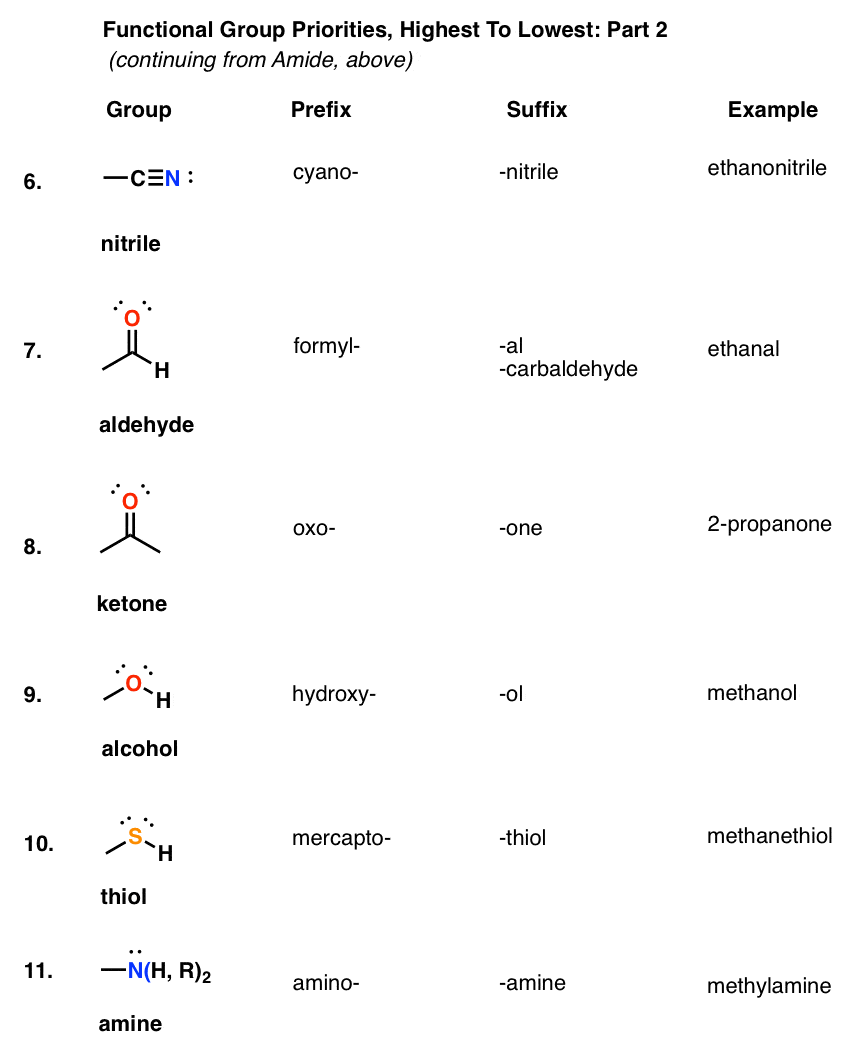
aldehyde functional group formula
What is the functional group of aldehyde?
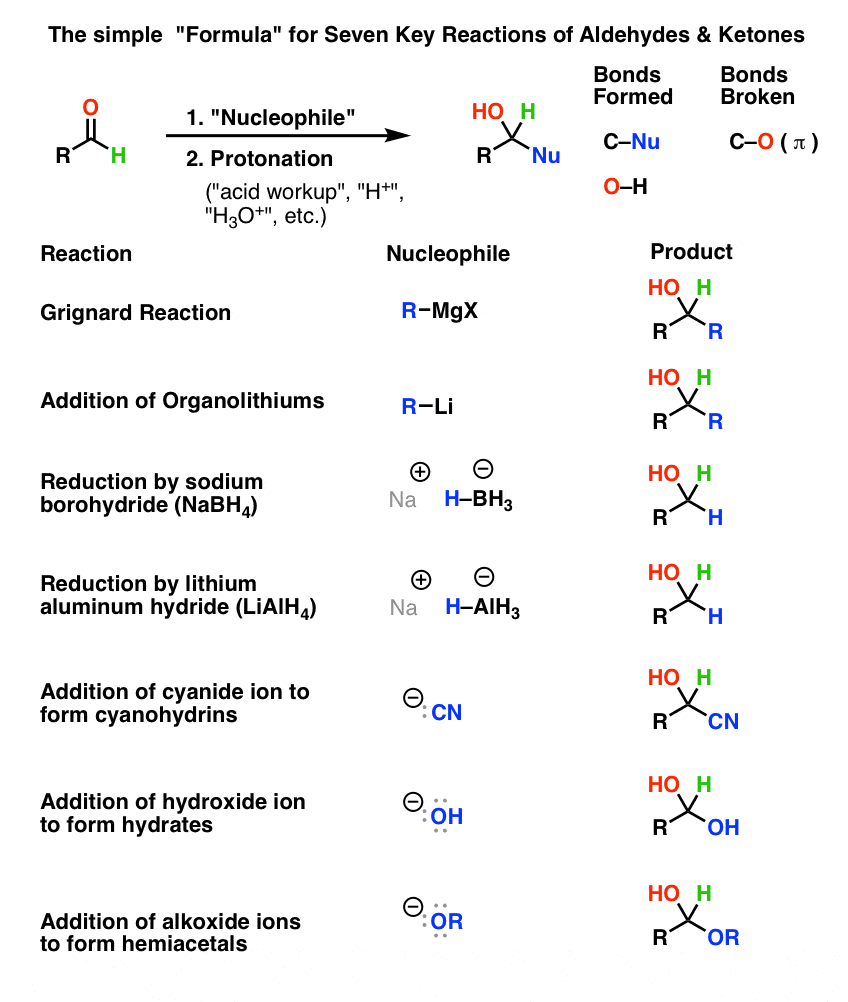
Aldehydes and ketones are organic compounds which incorporate a carbonyl functional group, C=O.
The carbon atom of this group has two remaining bonds that may be occupied by hydrogen or alkyl or aryl substituents.
If at least one of these substituents is hydrogen, the compound is an aldehyde.How do you write an aldehyde group?
Nomenclature of aldehydes
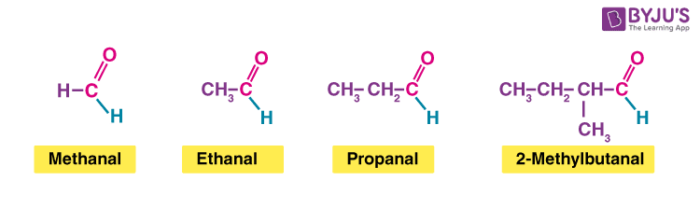
The aldehyde is shown by changing the suffix -e to -al.
Because the carbonyl group of an aldehyde can only be on the end of the parent chain and, therefore, must be carbon 1, there is no need to use a number to locate it.9 fév. 2024What is the formula of aldehyde group?
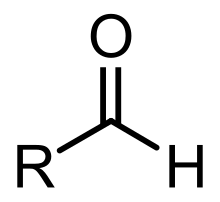
The chemical formula for an aldehyde is RCHO.
In this formula, R represents a hydrogen atom or carbon/hydrogen chain, CO represents the carbonyl, and H represents the hydrogen attached to the carbonyl chain.
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
most important functional groups in organic chemistry. In aldehydes the carbonyl group is bonded to a 12.1 Nomenclature and Structure of Carbonyl Group. |
|
Infrared Spectroscopy
May 15 2013 compound's structure |
|
INFRARED SPECTROSCOPY (IR)
What functional groups exist in the molecule? • If we have a specific stereoisomer? The field of organic structure determination attempts. |
|
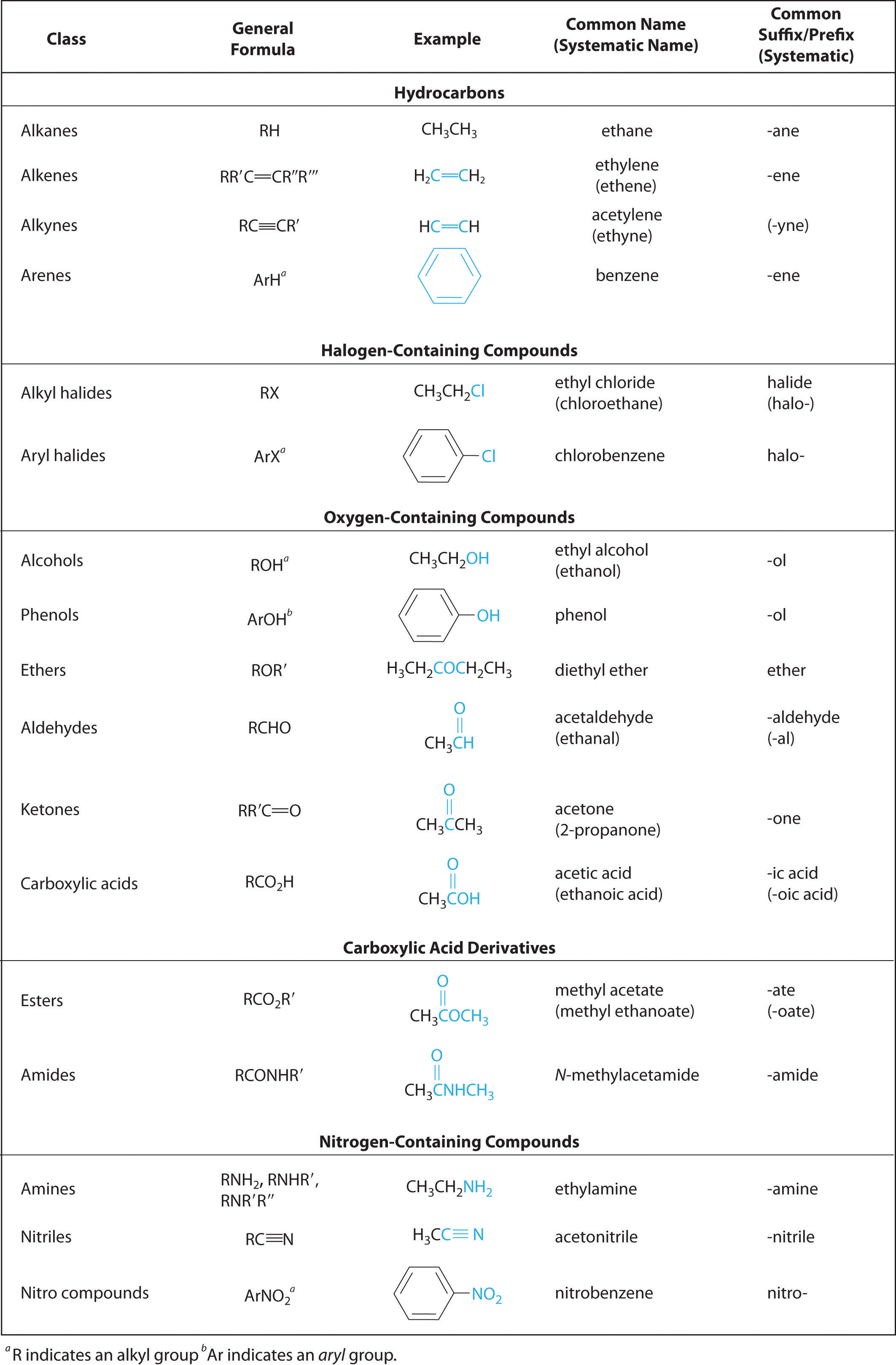
Short Summary of IUPAC Nomenclature of Organic Compounds
Nomenclature of Molecules Containing Substituents and Functional Groups hydroxy- amino-. Structure. Family of Compound. Carboxylic Acid. Aldehyde. |
|
Testsforfunctionalgroups - inorganiccompounds
To identify the functional groups present in an organic compound. I. TESTS FOR UNSATURATION These two carbonyl compounds (aldehydes and ketones) are. |
|
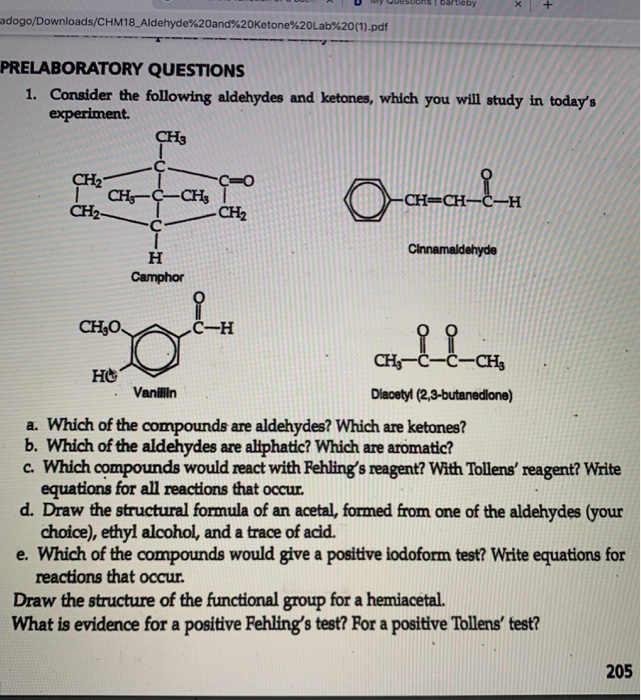
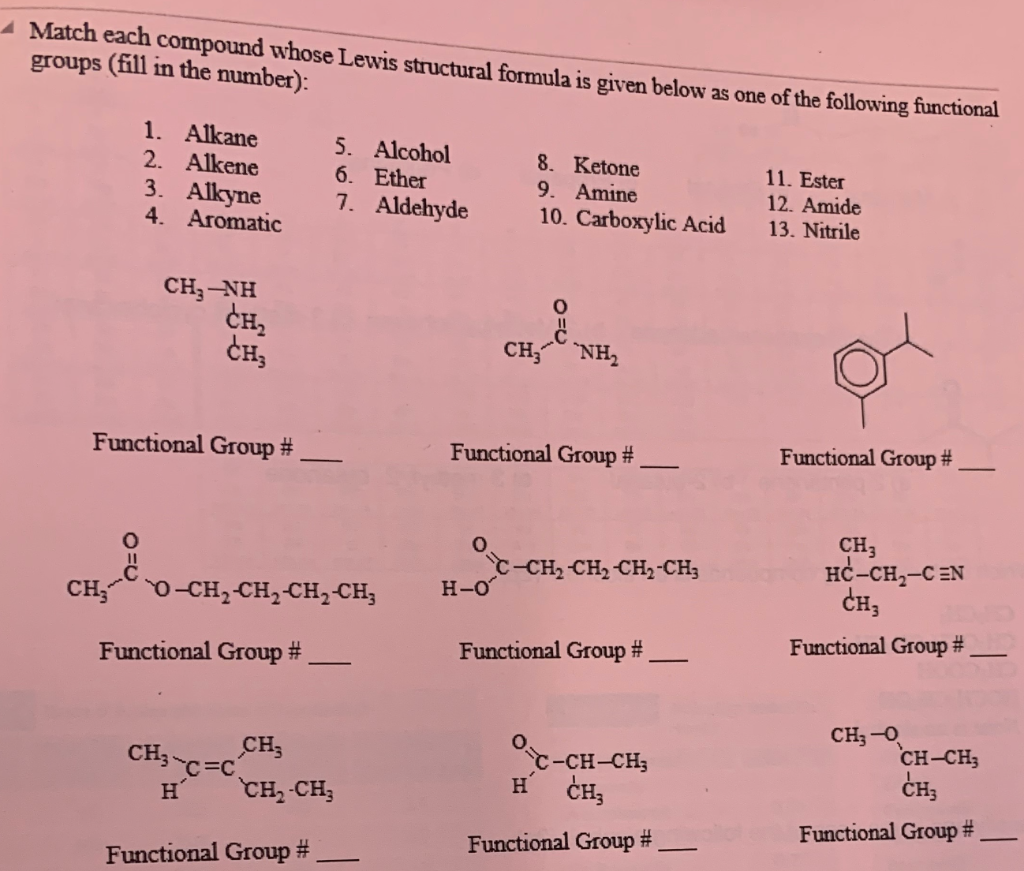
1. Identify the functional groups on the following organic molecules.
Apr 12 2018 H-C-C-0-C-C-H. H H. H H. Which type of compound is represented by the structural formula shown? A. a ketone. B. an aldehyde. C. an ester. |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
most important functional groups in organic chemistry. In aldehydes the carbonyl group is bonded to a 12.1 Nomenclature and Structure of Carbonyl Group. |
|
A Ru(II) Polypyridyl Complex Bearing Aldehyde Functions as a
May 10 2019 two aldehyde functional groups (complex 3 |
|
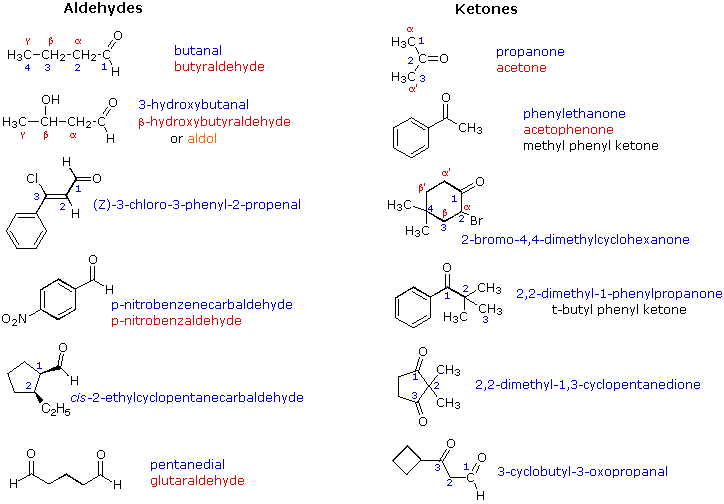
Aldehydes and Ketones
Aldehyde A compound containing a carbonyl group bonded to hydrogen (a CHO group) Ketone A compound containing a carbonyl group bonded to two carbons In benzaldehyde it is written as a line-angle formula, and in cinnamaldehyde it is abbreviated C6H5 J |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
most important functional groups in organic chemistry In aldehydes, the The physical properties of aldehydes and ketones are described as follows Methanal |
|
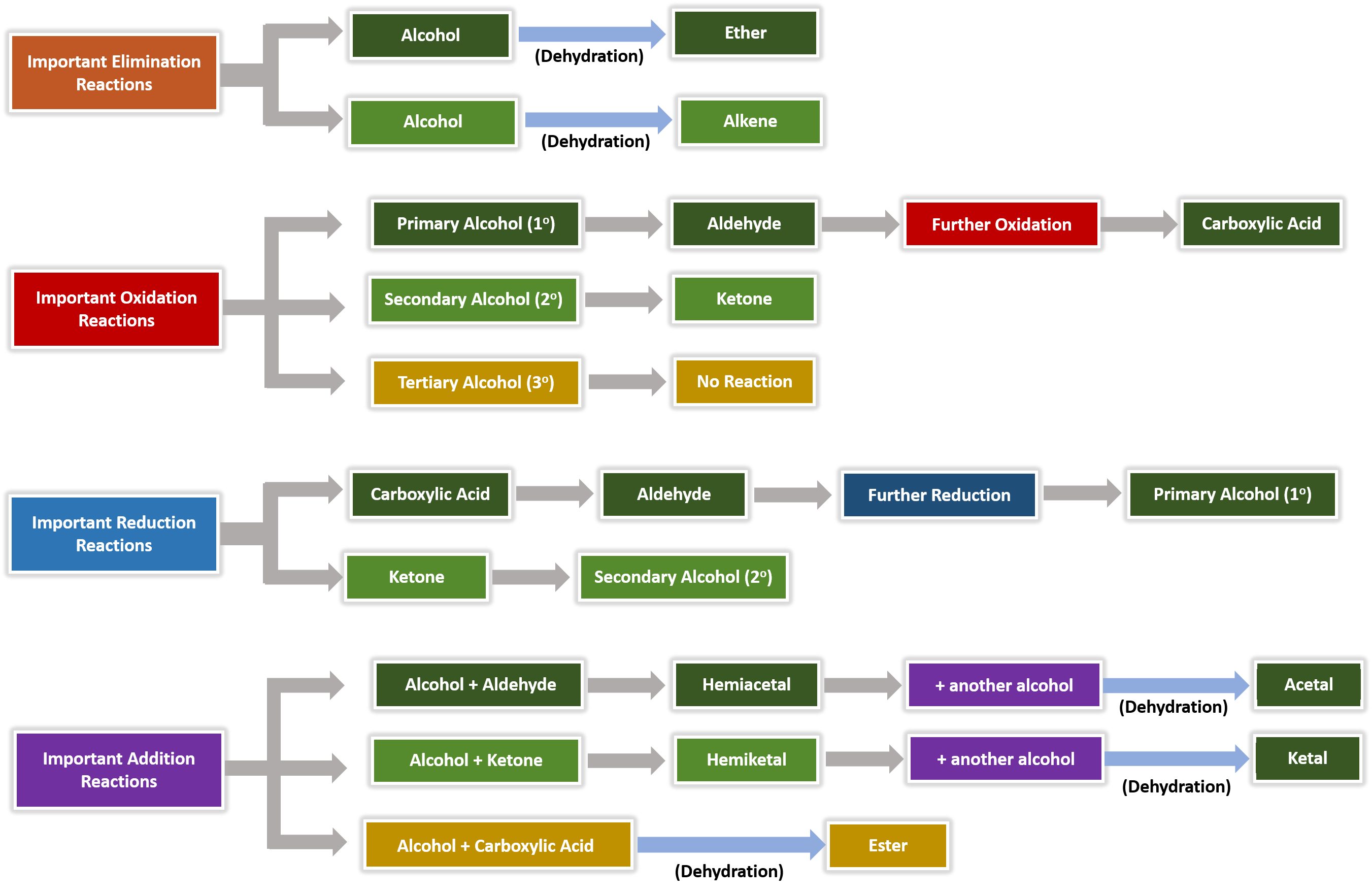
Alcohols, Ethers, Aldehydes, and Ketones
Predict the products of reactions involving alcohols and aldehydes 6 Explain what An alcohol is a compound with an –OH bonded Properties of Alcohols |
|
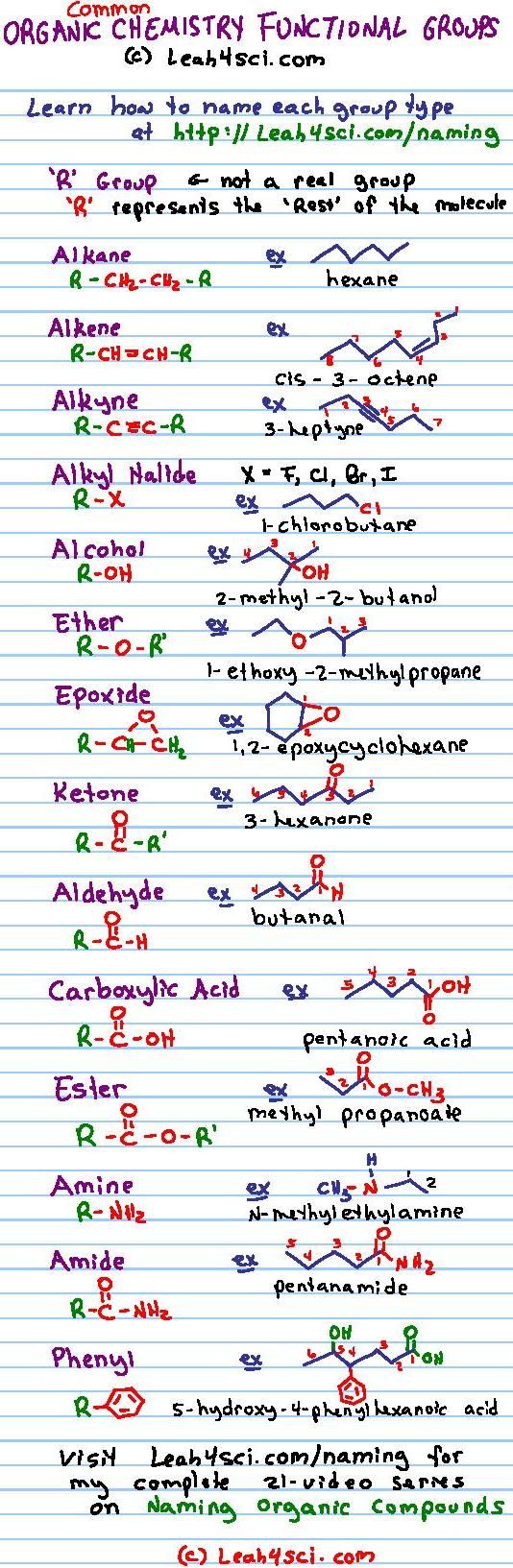
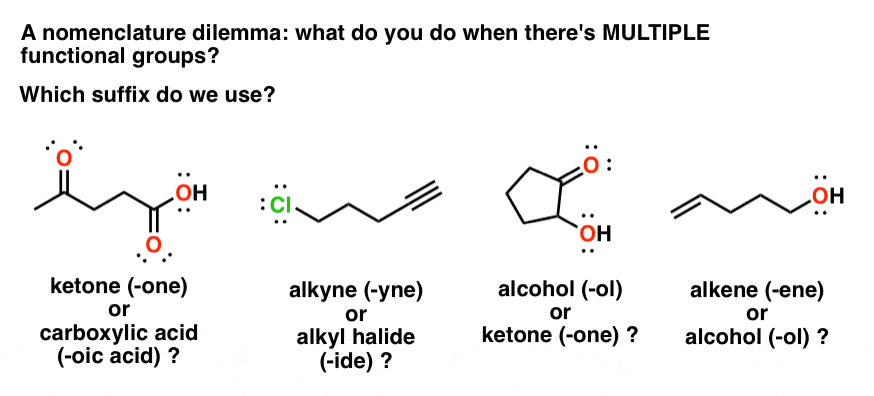
In this handout, all of our functional groups are presented as
Some functional groups have a suffix name when they are the highest priority functional group condensed line formula H3C CH3 carboxylic acid (3D) |
|
Functional Groups
4 jui 2018 · A functional group is a particular aldehyde CHACHAC-H -C-H R-C-H arrangement of atoms, which gives propanal characteristic properties to |
|
Organic chemistry functional groups keypdf
20 mai 2018 · 1 Which class of organic compounds contains nitrogen? IC: methy I A) aldehyde B) alcohol 3 Given the formula for an organic compound: |
|
Functional Groups
individual functional groups dictates the reactivity of the molecule of which note that in condensed structural formulas, the aldehyde group may be written as |
|
Functional Group Identification WS Keypdf
Identify the functional groups on the following organic molecules Acid OH Carboxylic Ester Which is the general formula for an aldehyde? 8 Which formula |
|
Alcohols, Aldehydes, and Ketones - De Boeck Supérieur
The oxidation-reduction reaction that takes place is described by the equation In the reaction the aldehyde functional group, R-CHO, is oxidized to a carboxylic |











/beta-methylamino-l-alanine-molecule-687786567-58b5bb9d5f9b586046c53d3d.jpg)












