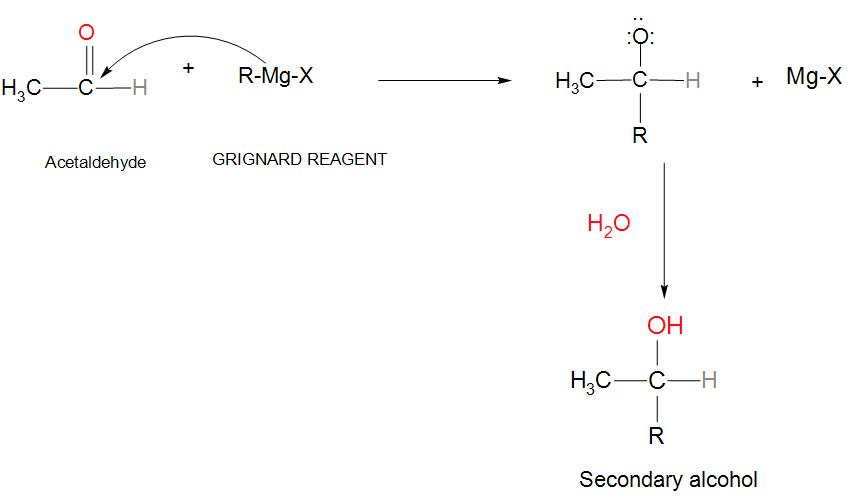
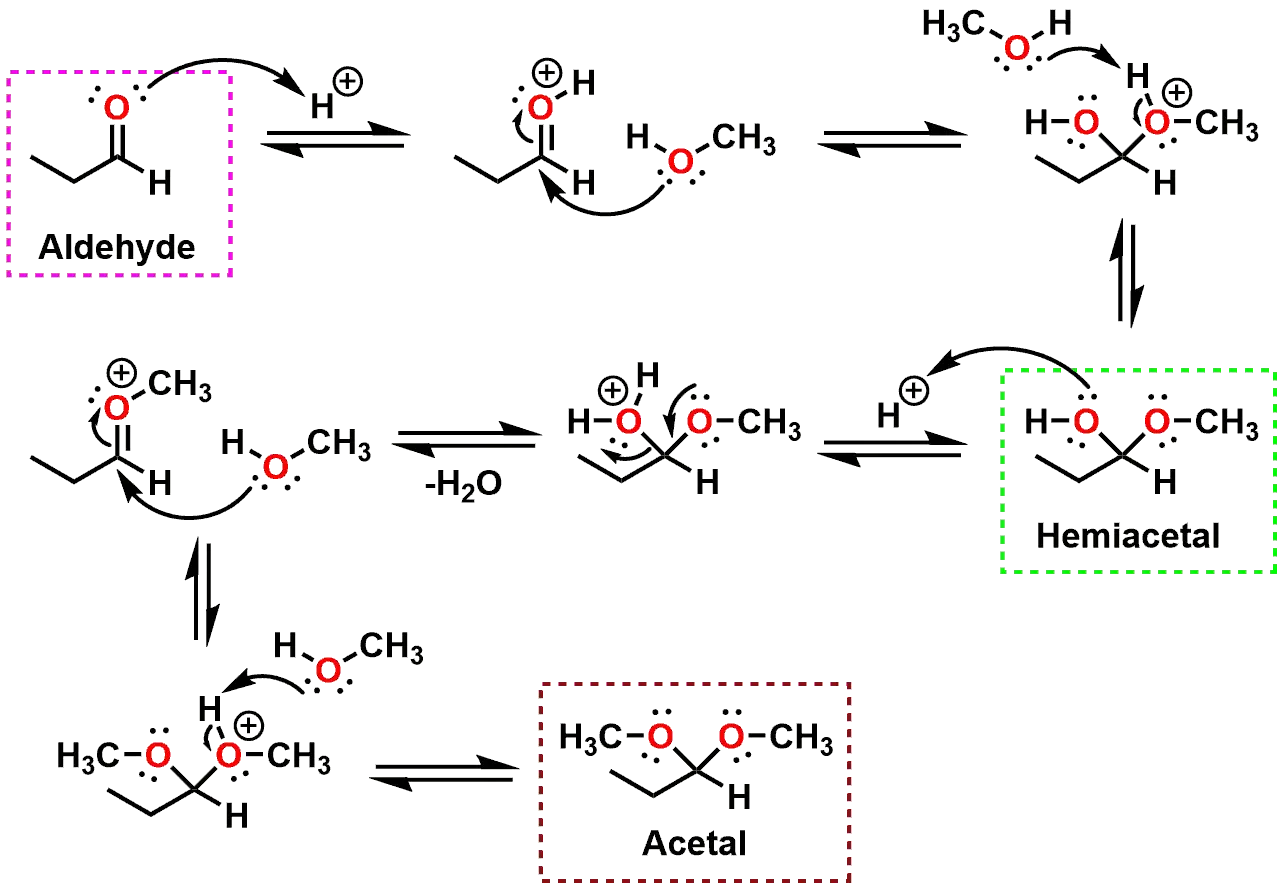
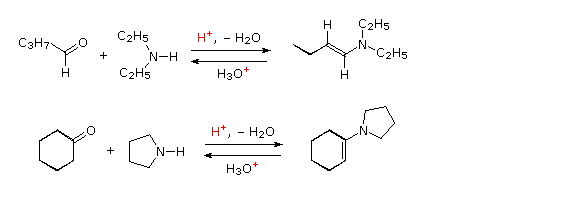
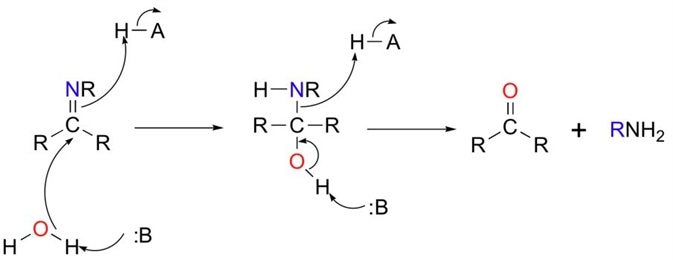
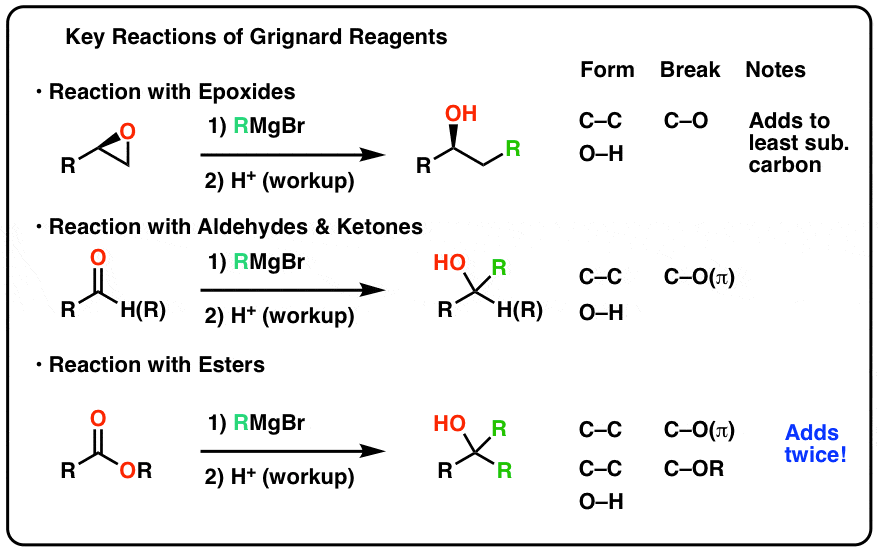
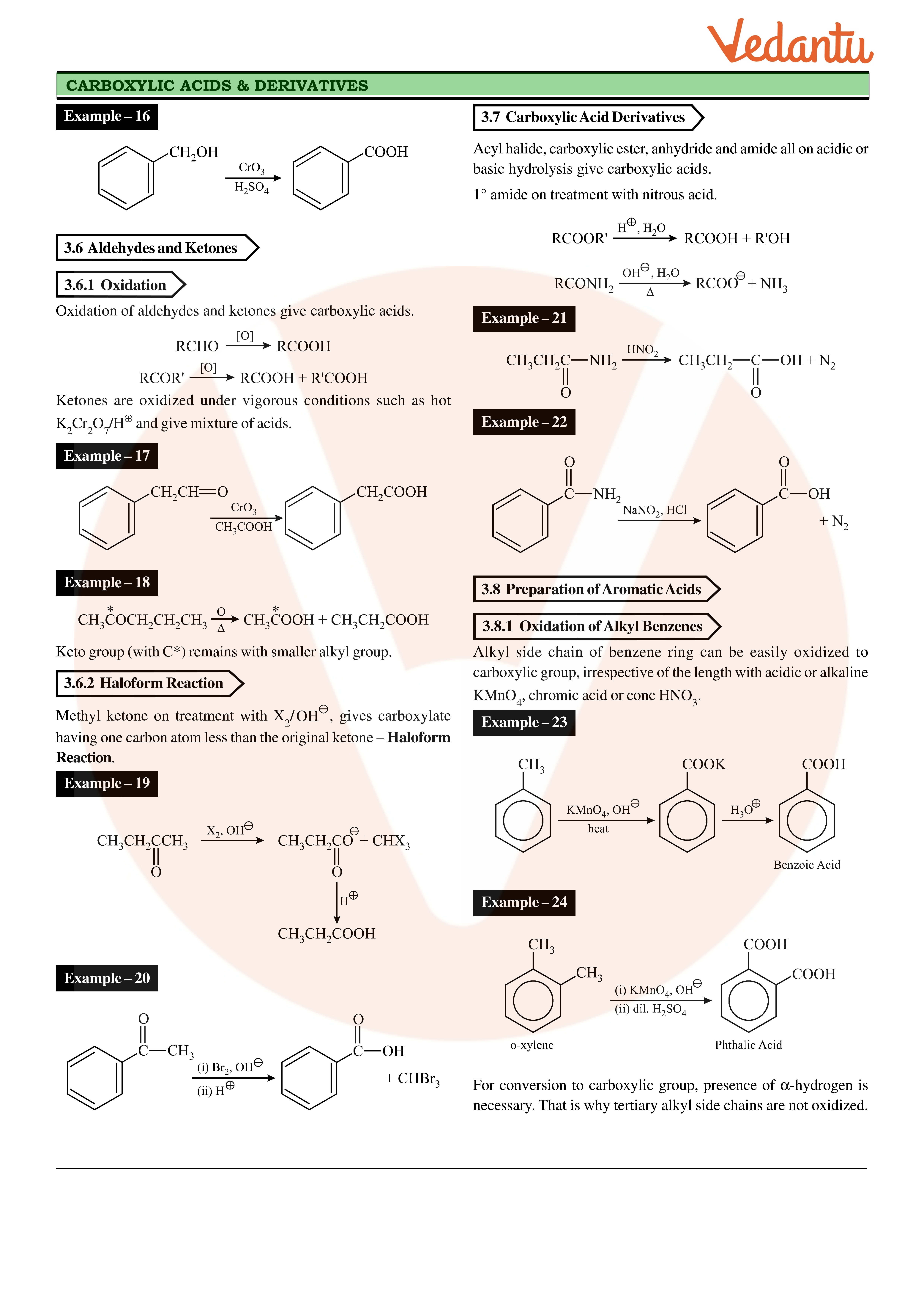
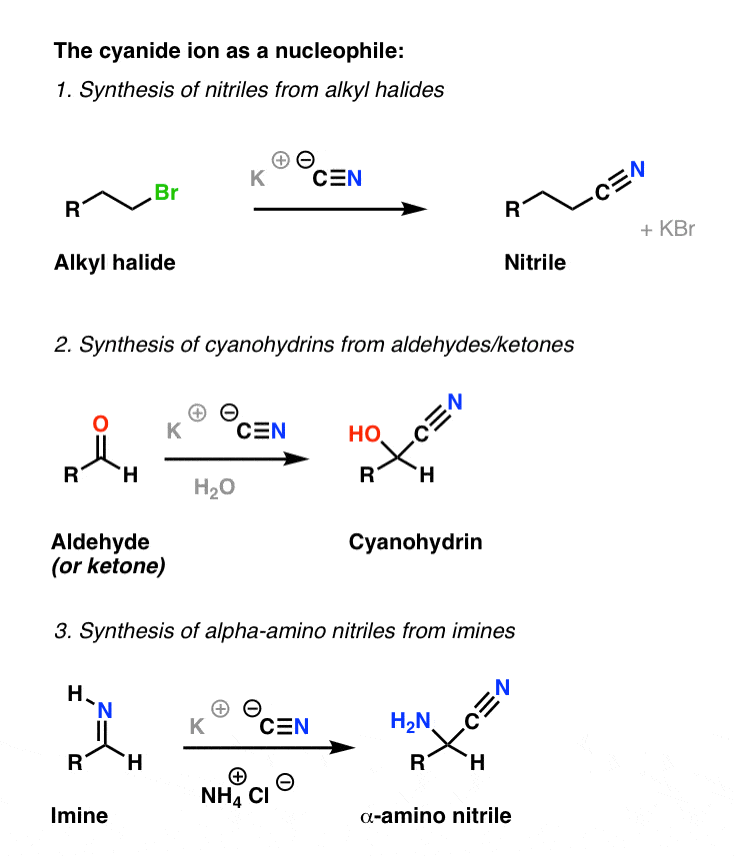
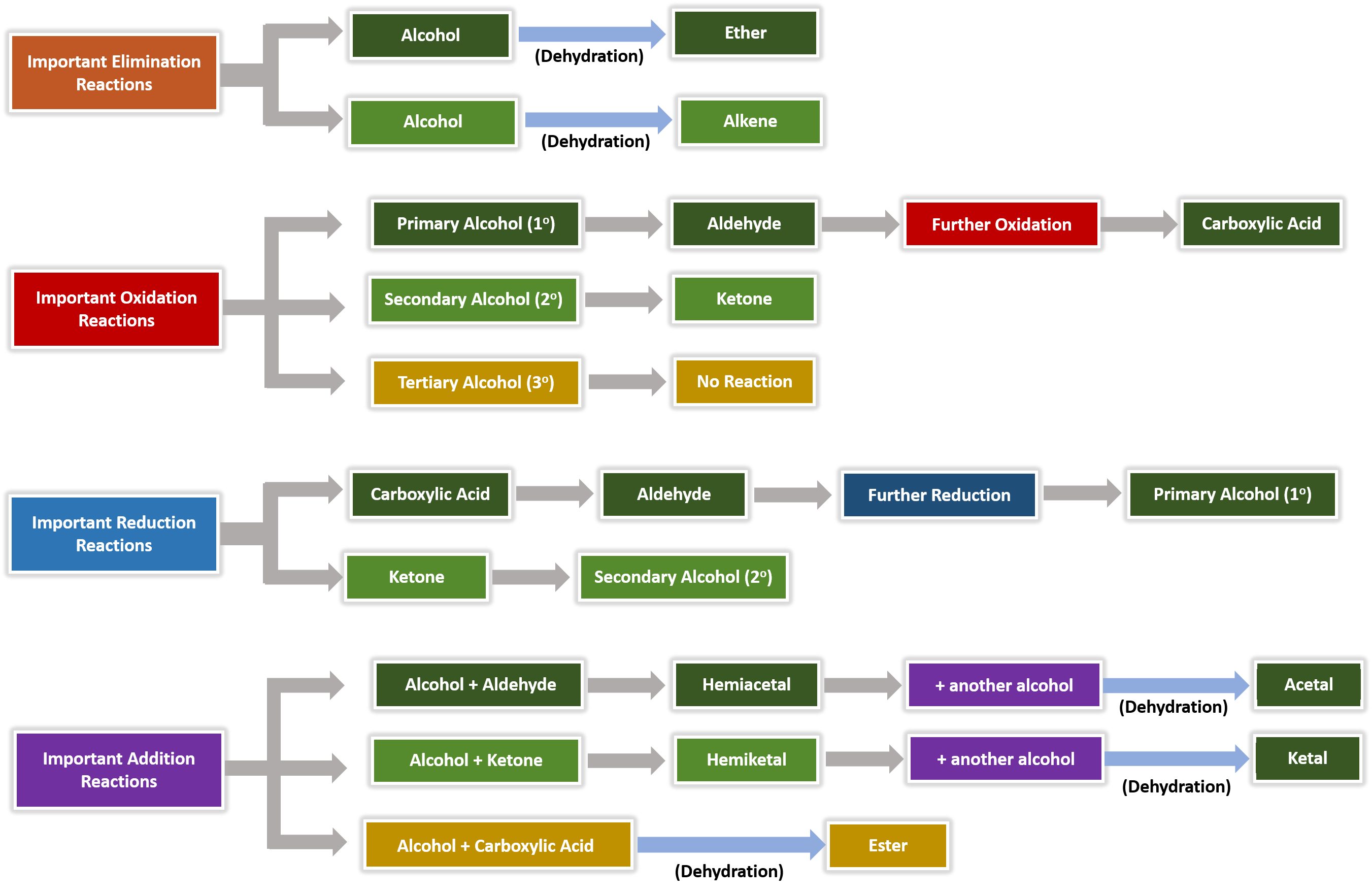
aldehyde hydrolysis
If water is added to simple aldehydes a rise in temperature, and a change in the ultraviolet absorption spectrum, as the result of the hydration of the carbonyl group, are observed.
This hydration involves initially the donation of the lone electron pair on the hydroxyl-oxygen to the carbonyl-carbon.
What happens when aldehyde is hydrolysed?
Aldehydes and ketones react with water to give 1,1-geminal diols known as hydrates.
In general, hydrates are not stable enough to be isolated as the equilibrium shifts back to starting materials (due to Le Chatelier's principle).
What happens when an aldehyde is dissolved in water?
Aldehydes and ketones react with water to yield 1,1-diols, or geminal (gem) diols.
The hydration reaction is reversible, and a gem diol can eliminate water to regenerate an aldehyde or ketone.
The position of the equilibrium between a gem diol and an aldehyde or ketone depends on the structure of the carbonyl compound.
What does H3O+ do to aldehydes?
H3O+ 1. o Does not react with carboxylic acids, esters, amides, aldehydes, or ketones (with the exception of α,β-unsaturated aldehydes and ketones). o Reacts with α,β-unsaturated aldehydes and ketones to give β-substituted carbonyl compounds.
This process is called 1,4-addition or conjugate addition.
|
Hydration of Aldehydes and Ketones
reaction an acetal can be hydrolyzed to an aldehyde or ketone by treatment with aqueous acid. • Since the reaction is also an equilibrium process |
|
Human liver aldehyde dehydrogenase. Esterase activity.
shown to hydrolyze p-nitrophenyl acetate. The hydrolysis of this ester by glyceraldehyde-3-phosphate dehydrogenase and by horse liver aldehyde dehydrogenase |
|
Synthesis of Coumarin 3?aldehyde Derivatives via Photocatalytic
New synthetic method of coumarins 3-aldehyde via visible light stimulation and oxidation promotion cascade radical cycliza- tion/hydrolysis process is |
|
CATALYSIS BY AROMATIC ALDEHYDES AND CARBON DIOXIDE
The aromatic aldehyde and bicarbonate catalysed hydrolyses of the p-nitro- catalyst for the hydrolysis of the p-nitrophenyl ester of leucine with a ... |
|
The Hydrolysis of Methyl o-Formylbenzoate. Participation of the
THE HYDROLYSIS OF METHYL o-FORMYLBENZOATE. PARTICIPATION OF THE. NEIGHBORING ALDEHYDE GROUP IN THE. HYDROXIDE ION AND MORPHOLINE-. CATALYZED REACTIONS1. |
|
Horse Liver Aldehyde Dehydrogenase
competitive inhibition of nitrophenyl acetate hydrolysis by an enzyme sulfhydryl to the aldehyde substrate would lead to formation of a thiohemiacetal ... |
|
ON THE ORIGIN OF THE HUMIN FORMED BY THE ACID
of a protein in the presence of an aldehyde markedly altered the nitrogen Hydrolysis in the Presence of Carbohydrates and of Aldehydes” J. |
|
Preparation of 1-Acyl-12-dihydroquinaldonitriles and their
Preparation of l-Acyl-l2-dihydroquinaldonitriles and their Hydrolysis to Aldehydes* sulfuric acid |
|
Hydrolysis and Surface-Active Properties of Alkylaldehyde-Bisulfite
Aldehyde-bisulfite derivatives are frequently used for protection of aldehydes in the field of synthetic chemistry due to their stabilities in solid state |
|
THE ORIGIN OF THE HUMIN FORMED BY THE ACID
acid and^that when a protein is hydrolyzed in the presence of an appro- priate aldehyde |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
Carbonyl groups in aldehydes and ketones may be oxidized to form compounds at the next “oxidation level”, that of carboxylic acids Addition of water to an aldehyde or ketone gives a product called a hydrate or a gem-diol (two -OH groups on the same carbon) The reaction is both acid-catalyzed and base-catalyzed |
|
Hydration of Aldehydes and Ketones
Because conversion of an aldehyde or ketone to an acetal is a reversible reaction, an acetal can be hydrolyzed to an aldehyde or ketone by treatment with aqueous acid Since the reaction is also an equilibrium process, it is driven to the right by using a large excess of water for hydrolysis |
|
Organic Chemistry - University of California, Riverside
Proton Shifts Amide Hydrolysis as an Example "Uncatalyzed" Addition of HOH to Carbonyl Compounds (16 3D) 16-17 Uncatalyzed Aldehyde Hydration |
|
Ketones and Aldehydes
The simplest carbonyl compounds are aldehydes and ketones A ketone has two alkyl The hydrolysis of a thioacetal generates an aldehyde Alternatively, the |
|
Synthesis of Ketones and Aldehydes
When water is scarce or removed, and alcohol is abundant, things drive to the right • Use H2O/H+ to hydrolyze an acetal back to an aldehyde or ketone • Use |
|
Ch 19 Answers for In-Class Handouts
E New Syntheses of Ketones/Aldehydes: Handout Reactions 10-18 (Sections 2 Br(na R 18 8 THOTTSS HAB Hydrolysis H Aldehyde 1 Buli 2 R-Br (10) |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
Aldehydes and ketones are the simplest and most important carbonyl carbon of the aldehyde group while in case of ketones the hydrolysis to aldehydes: |
|
Hydration of Ketones and Aldehydes
pKa of H2O: approx 16 pKa of alkoxide: 16-20 A hydrated ketone O Me Me Starting material MeO Na O Me Me OH Me Me OMe O Me Me Na |
|
Hydrolysis and Surface-Active Properties of Alkylaldehyde - CORE
in water and then separated it as a precipitate after alkali treatment Keywords: Cleavable Surfactant; Anionic Surfactant; Hydrolysis; Aldehyde-bisulfite Adduct |