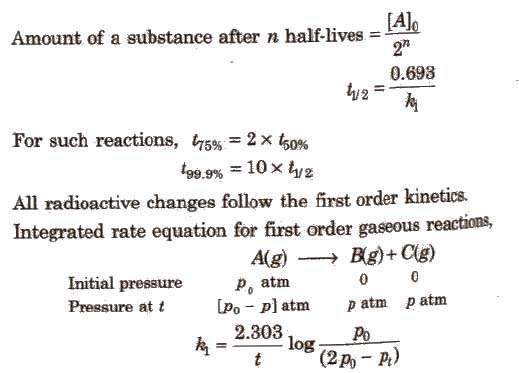all formulas of chemical kinetics class 12
|
Kinetics formulas copy
Chemical Kinetics - Formulas conc [A] All rates written as time or t Instantaneous rate is the slope of a concentration vs time plot and is d[A] shown by the differential equation: dt Overall rates for forward reactions are shown as POSITIVE rates d[A] therefore all reactants (which have negative rate of change) must have their rates |
How to memorize simple formulas on chemical kinetics?
Try to memorize the Simple Formulas on Chemical Kinetics provided as they form a stronger base during your study. 1. Rate of Reaction in the form of a Chemical reaction: m 1 A + m 2 B + ……… → n 1 P + n 2 Q + n 3 R + …….. 2. Unit of Rate constant: 3. First order reaction: 4. Second Order Reaction: Case: (i) When concentration of A and B taking same.
What does k mean in chemistry?
The constant k is the rate constant of the reaction, and is a measure of how fast or slow the reaction is. It depends on the reaction itself (the chemical compounds A and B) and environmental factors such as temperature. The rate constant does not depend on the concentrations of the species involved in the reaction.
Why is chemical kinetics important?
Chemical kinetics, also called reaction kinetics, helps us understand the rates of reactions and how it is influenced by certain conditions. It further helps to gather and analyse information about the mechanism of the reaction and define the characteristics of a chemical reaction.
What is the difference between chemical kinetics and rate of reaction?
Chemical kinetics is the study of the rate at which chemical reactions occur. It helps us understand the factors affecting these rates, the mechanism of the reaction, and the sequence of events that lead to the formation of products. Rate of Reaction is the change in concentration of reactants or products per unit time.
What Is Chemical Kinetics?
Chemical kinetics, also called reaction kinetics, helps us understand the rates of reactions and how it is influenced by certain conditions. It further helps to gather and analyse information about the mechanism of the reaction and define the characteristics of a chemical reaction. byjus.com
Rate of Formations and Disappearances
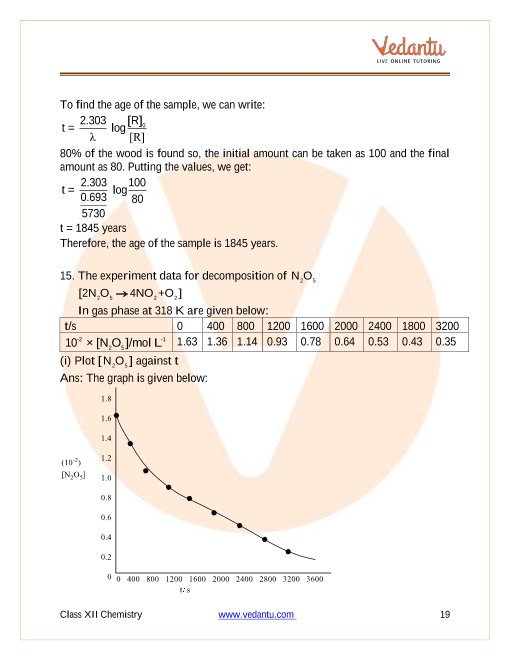
In any chemical reaction, as the reaction proceeds, the amount of reactants decreases, whereas the amount of products increases. One has to understand that the rate of the overall reaction depends on the rate at which reactants are consumed or the rate at which the products are formed. If a graph is plotted between the concentration of reactants an
Average and Instantaneous Rate
The rate of reaction can be classified into average and instantaneous rates depending on the amount of time period. If the time period taken is finite, then it’s called the average rate and is represented as, Δl → change in concentration Δt → change in time r avg → average rate The average rate doesn’t give exact information in most cases about the

Chemical Kinetics

CHEMICAL KINETICS Class 12

Integrated Rate Laws
|
Chemical Kinetics - Formulas
Half-life (t1/2) : The time it takes for the concentration to drop to one half its current value during the course of the reaction. Note that the “current value |
|
Formulae For CHEMICAL KINETICS
chemical equation. It can be experimentally determined. XII Chemistry. CHAPTER 4 - CHEMICAL KINETICS. If rate law expression for a reaction is. |
|
Chemical Kinetics Formula Sheet
Gradeup CSIR-NET. Super Subscription. Features: 1. Memory Based Test Series of the actual exam paper. 2. All the CSIR NET Test Series based on the latest |
|
Grade 12 Chemistry: A Foundation for Implementation
21 ene 2011 Grade 12 Chemistry. Topic 1: Reactions in Aqueous Solutions 1. Topic 2: Atomic Structure 1. Topic 3: Chemical Kinetics 1. |
|
Chapter 14 Chemical Kinetics
For a first order reaction equation (1) or (2) can be used to determine a. the concentration of a reactant at any time after the reaction has started b. the |
|
TOPIC 3: CHEMICAL KINETICS APPENDICES
Appendix 3.4B: Chemical Kinetics Problems (Answer Key) 18 GrAde 12 ChemisTry • Topic 3 Appendices ... Using the average rate of reaction formula. |
|
CHEMICAL KINETICS
(i) where k=Arrhenius factor |
|
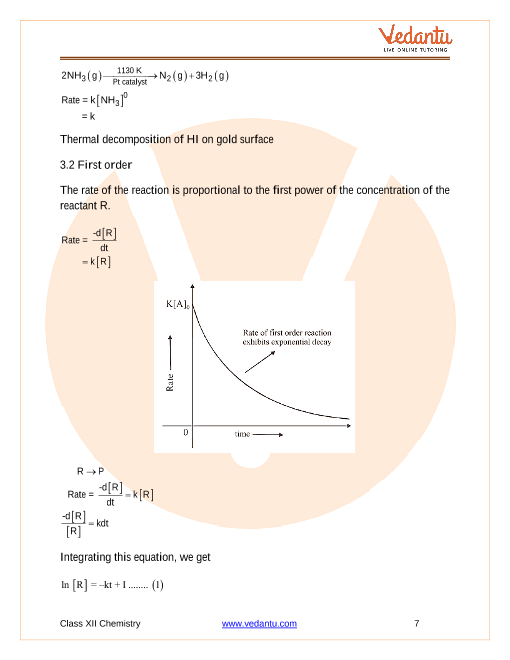
Chemical Kinetics What are Chemical Kinetics?
reactions occur kinetics also sheds light on the reaction stoichiometric coefficient in a balanced chemical equation. ... for all time intervals? |
|
Everything You Always Wanted to Know About Kinetics But Were
Chemical Kinetics But Were Afraid to Ask… Overall Reactions. An overall chemical reaction indicates reactants and products in the appropriate. |
|
Lech104.pdf
All these questions can be answered by the branch of chemistry which deals with the study of reaction rates and their mechanisms |
|
Chemical Kinetics
Chemical Kinetics - Formulas Chemical Kinetics - Formulas All rates written as ∆conc ∆time or ∆[A] ∆t Instantaneous rate is the slope of a concentration vs time plot and is shown by the differential equation: d[A] dt −d[A] dt In general for the overall reation: aA + bB → cC + dD overall rxn rate |
|
Formulae For CHEMICAL KINETICS - WordPresscom
chemical equation It can be experimentally determined XII Chemistry CHAPTER 4 - CHEMICAL KINETICS If rate law expression for a reaction is Rate = k [A] |
|
Chemical Kinetics - NCERT
reactant is the power of its concentration which appears in the rate law equation The order of a reaction is the sum of all such powers of concentration of terms for |
|
CHEMISTRY CLASS-XII - edudel
Analysis shows that a metal oxide has the empirical formula M 0 98 O 1 00 (b ) Atoms B occupy all the octahedral voids and half of the tetrahedral voids Chemical Kinetics : The branch of physical chemistry which deals with the study |
|
Physical Chemistry 3: — Chemical Kinetics — - Uni Kiel
26 jui 2019 · 5 4 1 Fundamental equation for reactive scattering in crossed molecular beams 117 Figure 1: Physical chemistry courses at CAU Kiel i e all red-ox reactions) are also limited by nuclear motions, because they usually |
|
Chapter 14 Chemical Kinetics
balanced chemical equation for the reaction Chemical kinetics is the study of how fast chemical Reactions may occur all at once or through several discrete |
|
1) Chemical kinetics - VG Vaze College
1) Chemical kinetics: It is a branch in physical chemistry which deals with the study of rate 5)Rate law: It is defined as an experimentally determined equation that course of the reaction are called zero order reactions the concentration of all the reactants except one(isolated) are taken in large excess so that the |
|
Chemical Kinetics
Notice that for every 1 mole of O2 that appears, 4 x as many moles of NO2 will also appear stoichiometric coefficient in a balanced chemical equation 1 Chung (Peter) Chieh broken and formed during the course of a reaction Elementary |
|
Reaction Rates: Chemical Kinetics
To determine the half-life for this reaction, we substitute the initial concentration of NOBr and the rate constant for the reaction into the equation for the half-life of a |
|
Unit-4 CHEMICAL KINETICS - PUE
Ans: Sum of the powers of the concentration of the reactants in the rate equation is called order of reaction Q 9) Calculate the overall order of a reaction which has |